Revolutionizing Amide Production: A Metal-Free Photocatalytic Strategy for Commercial Scale-Up
Revolutionizing Amide Production: A Metal-Free Photocatalytic Strategy for Commercial Scale-Up
The synthesis of amide compounds represents a cornerstone operation in modern organic chemistry, underpinning the production of countless active pharmaceutical ingredients and fine chemicals. As detailed in patent CN113943199A, a groundbreaking methodology has emerged that utilizes nitriles and diarylmethanes as primary raw materials to construct these vital bonds through a photocatalytic oxidation pathway. This innovative approach addresses critical limitations found in legacy synthetic routes by employing a dual-catalyst system comprising 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) and tert-butyl nitrite (TBN) under blue light irradiation. By leveraging molecular oxygen as a green terminal oxidant, this process not only achieves high efficiency but also aligns with the rigorous sustainability goals of contemporary chemical manufacturing. For industry leaders seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic depth and operational simplicity of this technology is essential for securing a competitive edge in the global market.
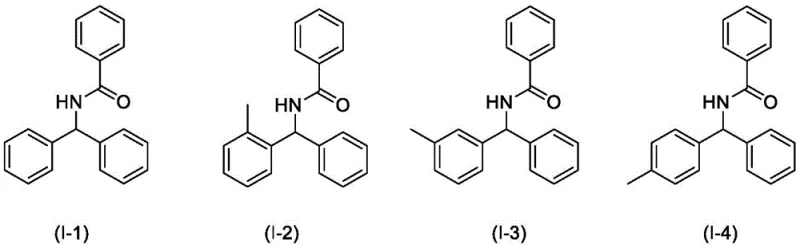
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of amide bonds has relied heavily on the activation of carboxylic acids or the utilization of transition metal catalysts to facilitate C-H bond functionalization. Traditional pathways often necessitate the use of stoichiometric amounts of hazardous oxidants or expensive metal complexes, which introduce significant complications regarding product purity and waste management. The presence of transition metals, such as copper or manganese, frequently leads to residual contamination that requires extensive and costly purification steps to meet regulatory standards for pharmaceutical applications. Furthermore, many conventional methods demand harsh reaction conditions, including elevated temperatures and pressures, which increase energy consumption and pose safety risks during large-scale operations. These inherent inefficiencies create substantial bottlenecks in cost reduction in amide manufacturing, forcing procurement teams to absorb higher operational expenditures and longer lead times.
The Novel Approach
In stark contrast, the novel photocatalytic strategy described in the patent data offers a transformative solution by operating under mild, ambient conditions driven by visible light energy. This method eliminates the need for transition metal catalysts entirely, relying instead on an organic photocatalytic system that activates C-H bonds directly without directing groups. The use of nitriles as both the nitrogen source and the solvent streamlines the reaction setup, reducing the volume of auxiliary chemicals required and simplifying the workup procedure. By utilizing oxygen from the air as the ultimate oxidant, the process generates water as the primary byproduct, dramatically improving the atom economy and environmental footprint of the synthesis. This paradigm shift enables the production of high-purity amide compounds with minimal downstream processing, providing a robust foundation for sustainable and economically viable chemical production.
Mechanistic Insights into DDQ/TBN-Catalyzed Photocatalytic Oxidation
The core of this technological advancement lies in the synergistic interaction between DDQ and TBN under blue light irradiation, which facilitates the generation of reactive radical species necessary for C-H bond activation. Upon exposure to visible light, the photocatalyst system initiates a single-electron transfer process that oxidizes the diarylmethane substrate to form a stabilized benzylic radical intermediate. This radical species subsequently reacts with the nitrile functionality, leading to the formation of a nitrilium ion intermediate that is crucial for the subsequent hydrolysis or rearrangement steps. The presence of trifluoroacetic acid acts as a vital promoter, enhancing the electrophilicity of the intermediates and accelerating the overall reaction kinetics without being consumed in the process. This intricate dance of electron transfer and radical recombination ensures high selectivity for the desired amide product while suppressing the formation of unwanted side products or over-oxidized species.
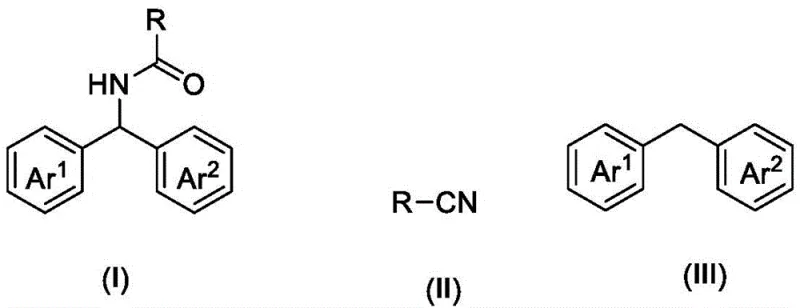
Controlling the impurity profile is paramount for any synthesis intended for pharmaceutical use, and this mechanism offers inherent advantages in that regard. The mild oxidative conditions prevent the degradation of sensitive functional groups that might otherwise be compromised under harsher thermal or chemical stress. Since the reaction proceeds at room temperature, thermal decomposition pathways are effectively shut down, ensuring that the structural integrity of complex substituents on the aromatic rings is maintained throughout the transformation. The absence of metal ions removes the risk of metal-catalyzed side reactions, such as homocoupling or polymerization, which are common pitfalls in traditional cross-coupling methodologies. Consequently, the resulting crude reaction mixture is significantly cleaner, allowing for more efficient isolation of the target molecule and reducing the burden on quality control laboratories to detect trace contaminants.
How to Synthesize N-benzhydrylbenzamide Efficiently
Implementing this synthesis route requires precise attention to reagent ratios and reaction parameters to maximize yield and reproducibility. The protocol involves combining diarylmethane with a substantial excess of nitrile, which serves a dual role as both reactant and solvent, ensuring high concentration of the nitrogen source throughout the reaction timeline. Catalysts DDQ and TBN are added in specific mass ratios relative to the substrate, typically ranging from 5% to 30%, to maintain optimal catalytic turnover without inducing excessive background reactions. The reaction vessel must be purged with oxygen to sustain the oxidative cycle, and irradiation with an 18W blue LED lamp provides the necessary photon energy to drive the photocatalytic cycle continuously over a period of 8 to 16 hours. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining diarylmethane, nitrile (acting as both reactant and solvent), DDQ, TBN, and trifluoroacetic acid in a sealed vessel.
- Replace the air in the vessel with oxygen and irradiate the mixture with an 18W blue LED lamp at room temperature for 8 to 16 hours.
- Upon completion, remove excess nitrile under reduced pressure and purify the crude product via column chromatography to isolate the target amide compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, adopting this metal-free photocatalytic technology offers profound benefits that extend far beyond simple chemical transformation. The elimination of transition metal catalysts directly translates to a simplified purification workflow, as there is no longer a need for specialized resin columns or complex extraction protocols designed to scavenge heavy metals. This reduction in processing complexity significantly lowers the operational overhead associated with manufacturing, allowing for faster batch turnover and reduced consumption of auxiliary materials. For procurement managers, this means a more predictable cost structure and the ability to negotiate better terms based on the inherent efficiency of the production method. The reliance on oxygen as an oxidant further decouples the process from the volatility of chemical oxidant markets, stabilizing input costs and enhancing supply chain resilience against raw material shortages.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the associated metal removal steps results in substantial cost savings across the production lifecycle. By avoiding the procurement of precious metal salts and the disposal costs linked to hazardous metal waste, manufacturers can achieve a leaner cost base. Additionally, the use of nitrile as a solvent reduces the need for purchasing and recovering separate organic solvents, further driving down variable costs. These efficiencies accumulate to provide a significant competitive advantage in pricing strategies for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as DDQ, TBN, and common nitriles, are widely available commodity chemicals with stable supply chains. Unlike specialized catalysts that may have limited suppliers or long lead times, these materials can be sourced from multiple vendors globally, mitigating the risk of supply disruption. The robustness of the reaction conditions also means that production is less susceptible to delays caused by equipment failures related to high-pressure or high-temperature requirements. This reliability is crucial for reducing lead time for high-purity amide compounds and ensuring consistent delivery to downstream clients.
- Scalability and Environmental Compliance: Operating at ambient temperature and pressure significantly lowers the barrier for scaling this technology from laboratory to industrial production. The mild conditions reduce the engineering controls required for safety, making it easier to adapt existing infrastructure for this new process. Furthermore, the green chemistry attributes, specifically the use of oxygen and the generation of minimal waste, align perfectly with increasingly stringent environmental regulations. This compliance reduces the regulatory burden and potential fines, ensuring long-term operational continuity and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic amide synthesis technology. These insights are derived directly from the patent specifications and are designed to clarify the operational benefits and feasibility for industrial partners. Understanding these details is key for stakeholders evaluating the integration of this method into their existing manufacturing portfolios. The answers reflect the consensus on how this technology resolves traditional pain points in amide production.
Q: How does this photocatalytic method improve product purity compared to traditional metal-catalyzed routes?
A: This method completely avoids the use of transition metal catalysts, thereby eliminating the risk of heavy metal residues in the final product. This significantly simplifies downstream purification processes and ensures compliance with stringent pharmaceutical purity standards without requiring expensive metal scavenging steps.
Q: What are the environmental benefits of using oxygen as the terminal oxidant in this synthesis?
A: Utilizing molecular oxygen as the terminal oxidant replaces hazardous stoichiometric oxidants often used in conventional methods. This shift drastically reduces chemical waste generation and enhances the overall green chemistry profile of the manufacturing process, aligning with modern environmental compliance requirements.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the reaction operates at normal temperature and pressure using visible light irradiation, which are conditions highly favorable for scale-up. The absence of extreme thermal requirements and the use of readily available reagents suggest strong potential for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-benzhydrylbenzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic technologies in reshaping the landscape of fine chemical manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying that every batch meets the exacting standards required by the global pharmaceutical industry. We understand that the transition to new synthetic routes requires confidence in both technical capability and supply stability, which is why we invest heavily in process optimization and capacity planning.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your specific project needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free protocol for your specific product portfolio. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your development timelines. Together, we can drive efficiency, reduce costs, and secure a sustainable supply chain for your critical amide intermediates.
