Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles Using Iron Catalysis for Commercial API Production
Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles Using Iron Catalysis for Commercial API Production
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazole derivatives, due to their pervasive presence in bioactive molecular frameworks. As highlighted in recent intellectual property developments, specifically patent CN111978265B, a novel preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives has emerged as a significant technological advancement. This patent discloses a streamlined synthetic pathway that leverages inexpensive starting materials and a facile iron-catalyzed cyclization strategy. The introduction of trifluoromethyl groups into these heterocyclic systems is critical, as it significantly enhances electronegativity, metabolic stability, and lipophilicity, properties essential for modern drug design. This report analyzes the technical merits of this invention, providing strategic insights for R&D directors, procurement managers, and supply chain leaders looking to optimize their intermediate sourcing strategies.
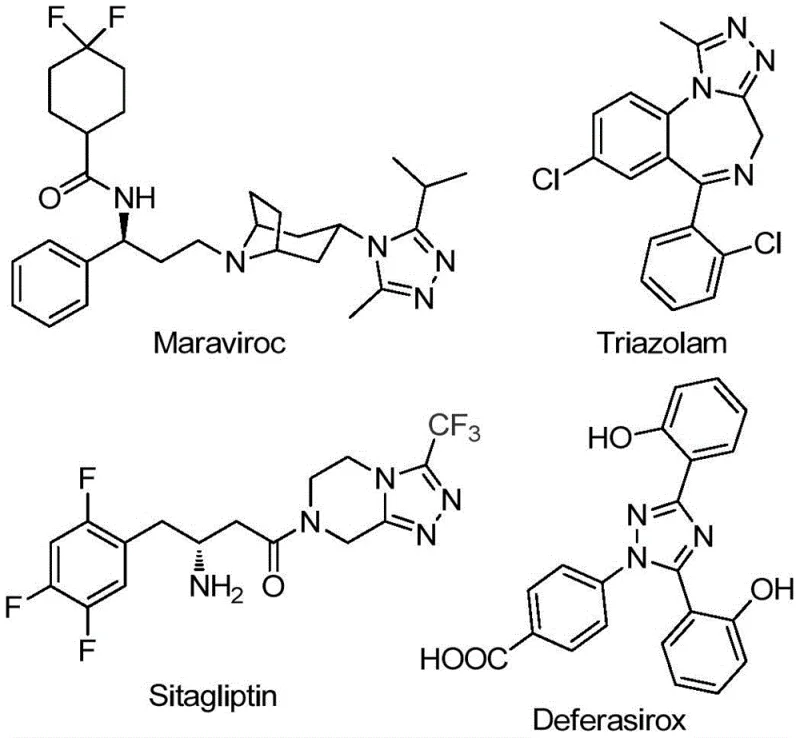
The structural versatility of the 1,2,4-triazole core is exemplified by its presence in major commercial pharmaceuticals. As illustrated in the provided chemical structures, prominent drugs such as Maraviroc, Triazolam, Sitagliptin, and Deferasirox all rely on this specific heterocyclic scaffold. The ability to efficiently synthesize 5-trifluoromethyl variants of these structures opens new avenues for medicinal chemistry optimization. The patent CN111978265B addresses the historical challenges associated with constructing these motifs, offering a solution that balances chemical efficiency with economic viability. For organizations acting as a reliable pharmaceutical intermediate supplier, adopting such methodologies can drastically improve the cost structure of complex molecule manufacturing while ensuring high purity standards required for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles was fraught with significant operational and economic hurdles. Literature reviews cited in the background of the patent identify at least five conventional approaches, each suffering from distinct drawbacks that hinder industrial adoption. Traditional methods often involve the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines. These pathways are frequently characterized by harsh reaction conditions, requiring extreme temperatures or pressures that pose safety risks and increase energy consumption. Furthermore, many existing protocols suffer from narrow substrate scopes, failing to accommodate diverse functional groups necessary for generating chemical libraries. Perhaps most critically for commercial operations, these legacy methods often yield poor reaction outputs, necessitating extensive purification steps that erode profit margins and extend lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method described in patent CN111978265B introduces a remarkably simple and efficient catalytic system. The core innovation lies in the use of cheap and readily available acyl hydrazides and trifluoroethylimidoyl chlorides as starting materials, promoted by ferric chloride (FeCl3). This approach eliminates the need for stringent anhydrous or oxygen-free environments, a common requirement in many organometallic couplings that complicates reactor setup and operation. The reaction proceeds smoothly in common organic solvents like 1,4-dioxane, demonstrating excellent functional group tolerance across a wide range of substrates. By shifting from precious metal catalysts or exotic reagents to abundant iron salts, this novel approach not only simplifies the chemical process but also aligns with green chemistry principles by reducing the reliance on scarce resources. This transition represents a paradigm shift in cost reduction in API manufacturing, allowing producers to achieve high conversion rates with minimal operational complexity.
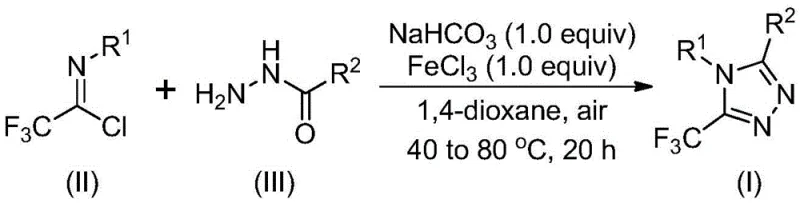
Mechanistic Insights into FeCl3-Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for R&D teams aiming to replicate or optimize the process. The reaction likely proceeds through a sequential mechanism initiated by base-promoted intermolecular carbon-nitrogen bond formation. In the first stage, sodium bicarbonate facilitates the coupling between the trifluoroethylimide chloride and the hydrazide to generate a trifluoroacetamidine intermediate. This step is crucial as it establishes the foundational connectivity required for ring closure. Following this initial assembly, the addition of the metal Lewis acid, ferric chloride, triggers the second phase of the reaction. The Lewis acid coordinates with the intermediate, promoting an intramolecular dehydration condensation that drives the cyclization to form the final 5-trifluoromethyl substituted 1,2,4-triazole derivative. This two-stage thermal profile allows for precise control over reaction kinetics, minimizing side reactions and maximizing the yield of the desired heterocycle.
From an impurity control perspective, the use of FeCl3 offers distinct advantages over stronger or less selective catalysts. The mild Lewis acidity of iron(III) ensures that sensitive functional groups on the aromatic rings, such as halogens or alkoxy groups, remain intact during the cyclization process. This chemoselectivity is paramount for maintaining the integrity of complex molecular architectures often found in late-stage intermediates. Moreover, the reaction conditions specified—initially reacting at 30 to 50°C followed by heating to 70 to 90°C—provide a thermal window that balances reaction rate with stability. The patent data indicates that this specific temperature gradient is optimal for driving the dehydration step to completion without degrading the trifluoromethyl moiety. Such mechanistic clarity provides confidence in the reproducibility of the process, a key factor for any entity seeking commercial scale-up of complex polymer additives or pharmaceutical precursors.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Derivatives Efficiently
The practical execution of this synthesis is designed for ease of operation, making it highly accessible for laboratory and pilot plant settings. The protocol utilizes a straightforward addition sequence where sodium bicarbonate, the imidoyl chloride, and the hydrazide are combined in an organic solvent. The reaction is allowed to proceed at a moderate temperature before the catalyst is introduced for the final cyclization step. This separation of steps ensures that the initial coupling occurs cleanly before the more vigorous cyclization conditions are applied. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and thermal profiles required to achieve the high yields reported in the patent examples. This level of procedural detail supports rapid technology transfer and minimizes the risk of batch-to-batch variability.
- Mix sodium bicarbonate, trifluoroethylimide chloride, and hydrazide in 1,4-dioxane and react at 30-50°C for 8-16 hours.
- Add ferric chloride (FeCl3) to the reaction mixture and increase temperature to 70-90°C for an additional 6-10 hours.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the implications of this patented technology extend far beyond the laboratory bench. The shift towards iron-catalyzed synthesis addresses several critical pain points in the global chemical supply network. By utilizing commodity chemicals like ferric chloride and sodium bicarbonate, manufacturers can decouple their production costs from the volatile pricing of precious metals like palladium or rhodium. This stability is essential for long-term contract planning and budget forecasting. Furthermore, the elimination of sensitive reaction conditions reduces the need for specialized equipment, thereby lowering capital expenditure requirements for production facilities. The robustness of the method ensures consistent output, mitigating the risk of supply disruptions that can plague more finicky synthetic routes.
- Cost Reduction in Manufacturing: The economic impact of replacing expensive catalysts with ferric chloride cannot be overstated. Iron is one of the most abundant and affordable metals on earth, leading to substantial cost savings in raw material procurement. Additionally, the simplified workup procedure, which involves basic filtration and standard column chromatography, reduces solvent consumption and waste disposal costs. The high atom economy of the reaction means less raw material is wasted as byproducts, further enhancing the overall process efficiency. These factors combine to create a highly competitive cost structure for producing high-purity OLED materials or pharmaceutical intermediates, allowing suppliers to offer more aggressive pricing without sacrificing margin.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically the acyl chlorides and hydrazides, are widely available in the global chemical market. This abundance ensures a steady flow of inputs, reducing the likelihood of stockouts that can halt production lines. The tolerance for ambient conditions (no strict anhydrous requirements) also simplifies logistics, as raw materials do not require specialized storage or transport under inert atmospheres. This resilience makes the supply chain more robust against external shocks, ensuring that delivery commitments to downstream API manufacturers are met consistently. For partners seeking a reliable agrochemical intermediate supplier, this reliability is a key differentiator in a crowded market.
- Scalability and Environmental Compliance: The patent explicitly notes the method's suitability for scaling up to gram levels and beyond, indicating strong potential for multi-ton production. The use of 1,4-dioxane, while requiring careful handling, is a well-understood solvent in industrial settings with established recovery protocols. The absence of heavy metal contaminants in the final product simplifies regulatory compliance, particularly for pharmaceutical applications where residual metal limits are strict. This environmental compatibility aligns with increasing global pressure for sustainable manufacturing practices. By adopting this greener synthetic route, companies can demonstrate their commitment to environmental stewardship while simultaneously improving their operational bottom line through reduced waste treatment costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN111978265B, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is critical for evaluating the feasibility of integrating this method into existing production workflows. The responses cover aspects ranging from substrate compatibility to purification strategies, providing a comprehensive overview of the technology's capabilities.
Q: What are the key advantages of the FeCl3-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods requiring harsh conditions or expensive reagents, this patent describes a process using cheap, readily available hydrazides and imidoyl chlorides under mild, aerobic conditions with high functional group tolerance.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is easily scalable to gram levels and beyond, utilizing common solvents like 1,4-dioxane and avoiding sensitive anhydrous or anaerobic requirements, which simplifies reactor engineering.
Q: What is the substrate scope for R1 and R2 groups in this triazole synthesis?
A: The method demonstrates broad compatibility, accepting substituted aryl groups (methyl, methoxy, halogen, trifluoromethyl) for R1 and alkyl, alkenyl, or aryl groups for R2, allowing for diverse derivative generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the FeCl3-catalyzed triazole formation. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this process, guaranteeing stringent purity specifications for every batch. With rigorous QC labs dedicated to monitoring impurity profiles and residual metals, we deliver intermediates that meet the exacting standards of the global pharmaceutical industry. Our commitment to quality ensures that your downstream synthesis remains uninterrupted and compliant with international regulations.
We invite you to explore how our expertise in heterocycle synthesis can accelerate your drug development timelines. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific molecular targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Together, we can optimize your supply chain and bring life-saving medications to market faster and more efficiently.
