Scalable Palladium-Catalyzed Synthesis of Polysubstituted Oxazoles for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, which serve as the backbone for countless bioactive molecules. Patent CN110156710B introduces a significant advancement in this domain by disclosing a novel preparation method for polysubstituted oxazole compounds. This technology addresses the longstanding challenges associated with synthesizing these valuable scaffolds, which are prevalent in natural products and therapeutic agents such as the anti-inflammatory drug Oxaprozine and the antidiabetic candidate AD-5061. By leveraging a palladium-catalyzed cyclization strategy, this invention provides a streamlined pathway that transforms simple benzamide derivatives and propargyl carbonates into complex oxazole structures with high efficiency. For R&D directors and procurement specialists, this represents a pivotal shift towards more accessible and operationally simple synthetic routes that can be seamlessly integrated into existing manufacturing workflows for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-substituted oxazole compounds has been fraught with significant technical hurdles that impede efficient commercial production. Traditional literature methods predominantly rely on the oxidation of oxazolines or intricate intramolecular and intermolecular cyclization reactions. These legacy processes often suffer from severe drawbacks, including the requirement for harsh reaction conditions that can degrade sensitive functional groups, leading to lower overall yields and compromised product purity. Furthermore, the starting materials required for these conventional pathways are frequently difficult to prepare or commercially unavailable, necessitating multi-step precursor syntheses that drastically inflate costs and extend lead times. The lack of broad substrate applicability in older methods also restricts the ability of chemists to rapidly iterate on molecular designs, slowing down the development of new drug candidates and limiting the structural diversity available for biological screening programs.
The Novel Approach
In stark contrast to these cumbersome traditional techniques, the methodology described in patent CN110156710B offers a transformative solution through a direct palladium-catalyzed coupling and cyclization sequence. This innovative approach utilizes readily available benzamide compounds and propargyl carbonates as building blocks, which are heated in the presence of a zero-valent palladium catalyst, a specialized phosphine ligand, and a base within a polar organic solvent. The reaction proceeds smoothly at temperatures between 100°C and 140°C, eliminating the need for extreme conditions while maintaining high conversion rates. As illustrated in the general reaction scheme below, this method allows for the direct construction of the oxazole core with excellent regioselectivity and functional group tolerance.

The operational simplicity of this novel route cannot be overstated; the post-reaction processing involves standard filtration, extraction, and column chromatography, making it highly amenable to both laboratory-scale optimization and industrial scale-up. This ease of purification directly translates to reduced processing time and lower solvent consumption, addressing key pain points for supply chain managers focused on efficiency and environmental compliance in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Cyclization
From a mechanistic perspective, this transformation relies on a sophisticated catalytic cycle initiated by the zero-valent palladium species, likely generated in situ from precursors such as tris(dibenzylideneacetone)dipalladium or bistriphenylphosphine palladium dichloride. The catalytic cycle begins with the oxidative addition or coordination of the palladium center to the propargyl carbonate, facilitating the formation of a pi-allyl palladium intermediate. Concurrently, the benzamide substrate coordinates to the metal center, positioning the nucleophilic nitrogen or oxygen atoms for subsequent attack. The presence of bulky and electron-rich phosphine ligands, specifically 4,6-bis(diphenylphosphino)dibenzofuran or bis(2-diphenylphosphino)phenyl ether, plays a critical role in stabilizing the active catalytic species and promoting the reductive elimination step that closes the oxazole ring. This ligand acceleration effect ensures that the reaction proceeds with high turnover numbers, minimizing the amount of expensive palladium required and reducing the risk of metal contamination in the final product.
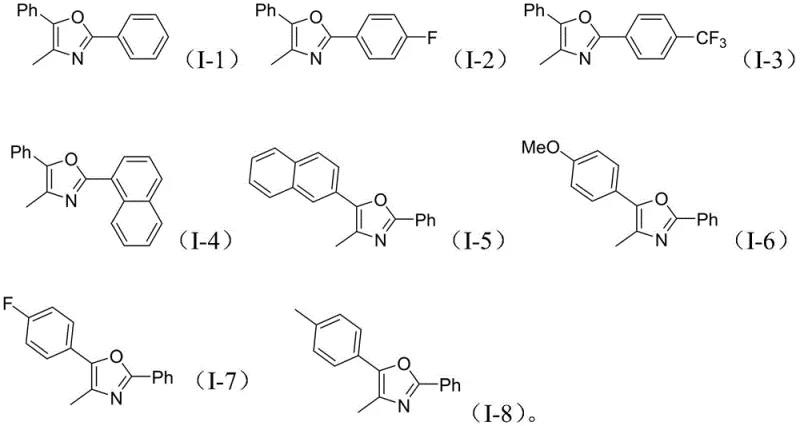
Impurity control is another critical aspect where this mechanism excels, particularly for R&D teams focused on strict regulatory standards. The use of specific bases like potassium carbonate or cesium carbonate helps to neutralize acidic byproducts generated during the decarboxylation of the propargyl carbonate, preventing side reactions such as polymerization or hydrolysis. The reaction conditions are optimized to favor the formation of the desired 2,4,5-trisubstituted oxazole scaffold over potential isomers, as evidenced by the clean NMR spectra reported for compounds (I-1) through (I-8). The versatility of the system is further demonstrated by its tolerance to diverse electronic environments on the aromatic rings, accommodating electron-withdrawing groups like trifluoromethyl and fluoro, as well as electron-donating groups like methoxy and methyl, without significant loss in yield. This broad substrate scope is visually represented by the array of successfully synthesized derivatives shown below.
How to Synthesize Polysubstituted Oxazole Efficiently
Implementing this synthesis route in a production environment requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The patent specifies that the benzamide compound should be used in excess relative to the propargyl carbonate, typically in a molar ratio ranging from 2:1 to 4:1, to drive the equilibrium towards product formation and compensate for any potential decomposition of the amide. The catalyst loading is kept low, between 0.05 and 0.15 mmol per 1 mmol of carbonate, reflecting the high efficiency of the palladium system. Detailed standardized operating procedures regarding temperature ramping, solvent selection (preferably NMP or DMF), and workup protocols are essential for reproducibility. For a comprehensive guide on executing this synthesis with precision, please refer to the step-by-step instructions provided in the section below.
- Combine zero-valent palladium catalyst, phosphine ligand, benzamide compound, propargyl carbonate, and base in a polar organic solvent such as NMP or DMF.
- Heat the reaction mixture to a temperature range of 100-140°C and maintain stirring for 10 to 20 hours to ensure complete conversion.
- Upon completion, perform filtration and extraction, followed by silica gel mixing and column chromatography purification to isolate the target oxazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers substantial strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant cost reduction in pharmaceutical intermediate manufacturing achieved through the use of inexpensive and commercially abundant starting materials. Benzamides and propargyl carbonates are commodity chemicals that can be sourced from multiple suppliers, mitigating the risk of single-source dependency and price volatility. Furthermore, the elimination of complex multi-step precursor syntheses required by older methods drastically simplifies the supply chain, reducing the number of intermediates that need to be stocked, tracked, and quality-controlled. This streamlining of the material flow leads to a leaner inventory profile and faster turnaround times from order to delivery, enhancing overall supply chain agility.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of catalytic amounts of palladium rather than stoichiometric oxidants or expensive reagents often found in traditional oxazole synthesis. Although palladium is a precious metal, the low loading levels specified in the patent (0.05 to 0.15 equivalents) combined with the potential for catalyst recovery or recycling in large-scale operations result in a favorable cost profile. Additionally, the simplified purification process, which avoids tedious crystallization steps or specialized separation techniques, reduces labor costs and solvent waste disposal fees. The ability to achieve high conversion rates with minimal byproduct formation means that raw material utilization is maximized, directly contributing to a lower cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any manufacturing operation, and this method enhances reliability by utilizing robust reaction conditions that are less sensitive to minor fluctuations in temperature or reagent quality. The use of polar aprotic solvents like NMP and DMF, which are standard in the industry, ensures that solvent supply chains remain stable and unaffected by niche market shortages. Moreover, the broad substrate tolerance allows for flexibility in sourcing; if a specific substituted benzamide is temporarily unavailable, the process can often accommodate alternative analogs with minimal re-optimization, ensuring that production schedules are maintained without interruption. This resilience is vital for meeting the rigorous delivery commitments expected by global pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, but this protocol is designed with scalability in mind. The reaction operates at moderate temperatures (100-140°C) that are easily achievable with standard heating jackets and do not require specialized high-pressure equipment, reducing capital expenditure for scale-up. From an environmental perspective, the atom economy of the cyclization is favorable, and the waste stream is primarily composed of benign salts and recoverable solvents. The straightforward workup involving filtration and extraction minimizes the generation of hazardous waste, aligning with modern green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations without compromising on output volume or product quality.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is crucial for stakeholders evaluating its potential for their specific projects. The following questions address common inquiries regarding the operational parameters, substrate compatibility, and purification strategies associated with this palladium-catalyzed route. These answers are derived directly from the experimental data and embodiments detailed in the patent documentation, providing a reliable foundation for decision-making.
Q: What are the primary advantages of this Pd-catalyzed method over traditional oxazole synthesis?
A: Unlike conventional methods involving harsh oxidation of oxazolines or complex intramolecular cyclizations, this patented process utilizes readily available benzamides and propargyl carbonates under milder conditions (100-140°C), significantly simplifying operation and purification.
Q: Which substrates are compatible with this polysubstituted oxazole preparation method?
A: The method demonstrates strong substrate designability, accommodating various R1 groups such as methyl, fluoro, trifluoromethyl, and naphthyl, as well as R2 groups including phenyl and naphthyl, allowing for the synthesis of diverse bioactive scaffolds.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is highly practical for scale-up due to its simple operational steps, use of common polar solvents like DMF and NMP, and straightforward post-reaction purification via standard column chromatography, ensuring consistent quality for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Oxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has extensively evaluated the technology described in patent CN110156710B and is fully prepared to leverage this efficient palladium-catalyzed route for your custom synthesis needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted oxazole intermediate we deliver meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule. We encourage you to contact us today to discuss your project requirements,索取 specific COA data for our reference standards, and obtain detailed route feasibility assessments that will demonstrate the tangible value of our manufacturing capabilities for your next-generation pharmaceutical products.
