Advanced Palladium-Catalyzed Synthesis of Polysubstituted Oxazoles for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Polysubstituted Oxazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, which serve as critical scaffolds in drug discovery and development. A significant breakthrough in this domain is detailed in patent CN110156710B, which discloses a highly efficient preparation method for polysubstituted oxazole compounds. This technology leverages a palladium-catalyzed annulation strategy between benzamide derivatives and propargyl carbonates, offering a streamlined pathway to complex molecular architectures. As a leading entity in the chemical sector, we recognize that the ability to access these structures reliably is paramount for the synthesis of bioactive molecules, including potential treatments for rheumatoid arthritis and diabetes. The methodology described represents a substantial leap forward in synthetic efficiency, addressing long-standing challenges regarding substrate availability and reaction severity.
This innovative approach is particularly relevant for R&D teams focused on high-purity pharmaceutical intermediates. By utilizing a zero-valent palladium catalyst system in conjunction with specialized phosphine ligands, the process achieves high conversion rates under controlled thermal conditions ranging from 100°C to 140°C. The versatility of this reaction allows for the incorporation of diverse functional groups, thereby enabling medicinal chemists to rapidly explore structure-activity relationships (SAR). For procurement and supply chain stakeholders, the reliance on commercially accessible starting materials such as substituted benzamides translates to reduced logistical complexity and enhanced supply continuity. This report delves into the technical nuances and commercial implications of adopting this synthesis route for your manufacturing pipeline.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the oxazole ring has relied on several classical transformations, each carrying inherent drawbacks that hinder large-scale application. Traditional routes often involve the oxidation of oxazolines, a process that frequently necessitates the use of stoichiometric amounts of harsh oxidizing agents, leading to significant waste generation and safety concerns. Alternatively, intramolecular cyclization strategies require pre-functionalized precursors that are often difficult and expensive to prepare, adding multiple steps to the overall synthetic sequence. These legacy methods typically suffer from narrow substrate scope, meaning that introducing specific substituents—especially electron-deficient or sterically hindered groups—can result in poor yields or complete reaction failure. Furthermore, the purification of products from these older methods can be arduous due to the formation of complex byproduct mixtures, driving up the cost of goods sold (COGS) and extending lead times for critical intermediates.
The Novel Approach
In stark contrast, the method disclosed in patent CN110156710B introduces a direct intermolecular cyclization that bypasses the need for pre-formed oxazoline intermediates. By reacting readily available benzamide compounds with propargyl carbonates in the presence of a palladium catalyst and a phosphine ligand, the process constructs the oxazole core with remarkable atom economy. The reaction conditions are notably milder compared to oxidative protocols, utilizing common polar aprotic solvents like N-methylpyrrolidone (NMP) or N,N-dimethylformamide (DMF). This shift not only simplifies the operational procedure but also significantly enhances the safety profile of the manufacturing process. The ability to tolerate a wide array of substituents, including halogens and trifluoromethyl groups, ensures that this method is applicable to a broad spectrum of target molecules, making it a superior choice for cost reduction in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Annulation
The core of this transformation lies in the sophisticated catalytic cycle mediated by zero-valent palladium species. The reaction initiates with the oxidative addition of the propargyl carbonate to the Pd(0) center, generating a reactive pi-allyl palladium intermediate. This species then undergoes nucleophilic attack by the nitrogen atom of the benzamide, facilitated by the presence of a base such as potassium carbonate or cesium carbonate. Subsequent migratory insertion and reductive elimination steps forge the carbon-oxygen and carbon-nitrogen bonds necessary to close the oxazole ring, regenerating the active Pd(0) catalyst for the next turnover. The choice of ligand, specifically bulky and electron-rich phosphines like Xantphos or DPEphos, is critical in stabilizing the palladium center and promoting the desired reductive elimination pathway over competing side reactions. This mechanistic precision ensures high selectivity and minimizes the formation of regioisomers, which is crucial for maintaining product quality.
From an impurity control perspective, the mechanism offers distinct advantages. The use of mild inorganic bases prevents the degradation of sensitive functional groups that might occur under strongly basic or acidic conditions found in traditional methods. Moreover, the catalytic nature of the palladium system means that metal loading can be kept low (0.05 to 0.15 mmol relative to 1 mmol of substrate), reducing the burden of heavy metal removal in downstream processing. The reaction's tolerance for various electronic environments on the aromatic rings suggests that the transition state is well-balanced, preventing the accumulation of unreacted starting materials or polymeric byproducts. This results in a cleaner crude reaction profile, which simplifies the final purification via column chromatography and ensures the delivery of high-purity polysubstituted oxazoles suitable for stringent pharmaceutical applications.
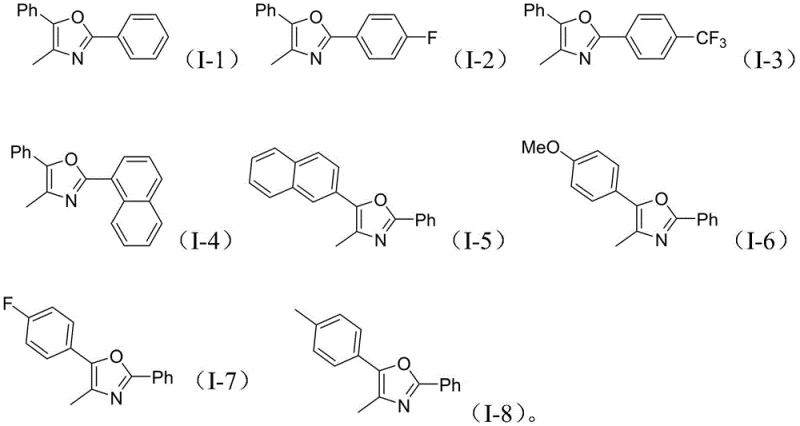
How to Synthesize Polysubstituted Oxazoles Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and reaction parameters to maximize yield and reproducibility. The protocol dictates a molar ratio where the benzamide is used in excess (2 to 4 equivalents) relative to the propargyl carbonate to drive the equilibrium towards product formation. The reaction is typically conducted in a Schlenk tube or similar vessel under an inert atmosphere to protect the sensitive palladium catalyst from oxidation. Heating the mixture to the optimal temperature window of 100-140°C for a duration of 10 to 20 hours allows for complete consumption of the limiting reagent. Following the reaction, a standard workup involving aqueous quenching, organic extraction, and drying provides the crude material, which is then purified to isolate the target compound. For detailed operational specifics and safety guidelines, please refer to the standardized synthesis steps provided below.
- Charge a reaction vessel with zero-valent palladium catalyst, phosphine ligand, benzamide compound, propargyl carbonate, and base in an organic solvent such as NMP or DMF.
- Heat the reaction mixture to a temperature range of 100-140°C and maintain stirring for 10 to 20 hours to ensure complete conversion.
- Upon completion, perform filtration and extraction, followed by silica gel mixing and column chromatography purification to isolate the target oxazole product.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented methodology offers tangible benefits for procurement managers and supply chain directors focused on optimizing the sourcing of complex chemical building blocks. The primary driver for cost efficiency is the utilization of benzamide derivatives, which are commodity chemicals available from numerous global suppliers at competitive price points. Unlike specialized heterocyclic precursors required by other methods, these starting materials do not suffer from supply bottlenecks, ensuring a stable and continuous flow of raw materials for production. The simplicity of the reaction setup, which does not require exotic equipment or extreme pressure conditions, further lowers the barrier to entry for contract manufacturing organizations (CMOs) looking to scale this process. This accessibility translates directly into a more resilient supply chain capable of meeting fluctuating market demands without significant lead time extensions.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the elimination of multi-step precursor synthesis. By converging two simple building blocks in a single catalytic step, the overall number of unit operations is drastically reduced, leading to lower labor and utility costs. Additionally, the use of inexpensive inorganic bases like potassium carbonate instead of costly organic bases or stoichiometric metal reagents contributes to substantial raw material savings. The high atom economy of the annulation reaction minimizes waste disposal costs, while the straightforward purification protocol reduces solvent consumption and processing time. These factors collectively drive down the manufacturing cost per kilogram, making the final oxazole intermediates more price-competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the broad availability of the requisite substrates. Benzamides and propargyl carbonates are produced on a large scale for various industrial applications, meaning that sourcing risks associated with niche custom synthesis are mitigated. The robustness of the reaction conditions also implies that the process is less susceptible to minor variations in raw material quality, reducing the rate of batch failures. For a reliable pharmaceutical intermediate supplier, this consistency is key to maintaining long-term contracts and ensuring that downstream API production schedules are met without interruption. The ability to source materials locally or from multiple regions further diversifies the supply base and reduces geopolitical risk.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram and eventually to tonnage levels is facilitated by the use of standard polar solvents and manageable thermal profiles. The absence of hazardous oxidants or pyrophoric reagents simplifies the safety assessment and regulatory approval process for new manufacturing sites. From an environmental standpoint, the catalytic nature of the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The simplified workup procedure generates less aqueous and organic waste compared to traditional oxidative methods, easing the burden on wastewater treatment facilities. This environmental compatibility is increasingly important for meeting the strict sustainability criteria imposed by major pharmaceutical clients and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing production portfolios. For further technical discussions or custom synthesis requests, our team is available to provide deeper insights tailored to your specific project requirements.
Q: What are the primary advantages of this Pd-catalyzed method over traditional oxazole synthesis?
A: Unlike conventional methods that often require harsh oxidants or difficult-to-prepare oxazoline precursors, this patented approach utilizes readily available benzamides and propargyl carbonates. It operates under relatively mild thermal conditions (100-140°C) and offers a broader substrate scope, allowing for the efficient introduction of diverse functional groups like halogens and trifluoromethyl groups.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the process is designed for scalability. The use of common polar solvents like NMP or DMF, combined with inexpensive inorganic bases such as potassium carbonate, simplifies the supply chain. Furthermore, the workup procedure involving standard filtration and column chromatography is robust enough to be adapted for industrial production environments.
Q: What types of substituents are tolerated in this reaction system?
A: The reaction demonstrates excellent tolerance for various substituents on both the benzamide and the alkyne components. Electron-withdrawing groups like fluorine and trifluoromethyl, as well as electron-donating groups like methoxy and methyl, are successfully incorporated. Additionally, bulky groups such as naphthyl rings are compatible, yielding the corresponding polysubstituted oxazoles in good yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Oxazole Supplier
The technological potential of this palladium-catalyzed annulation represents a significant opportunity for advancing the synthesis of bioactive oxazole derivatives. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of polysubstituted oxazole meets the highest industry standards. Our commitment to quality assurance ensures that the intermediates we supply are fully characterized and ready for immediate use in your downstream API synthesis.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this methodology. We encourage you to reach out to request specific COA data for our catalog compounds or to discuss route feasibility assessments for custom targets. Let us help you optimize your supply chain and accelerate your drug development timeline with our superior manufacturing capabilities and dedicated customer support.
