Advanced Synthesis of Asymmetric Pyrene Derivatives for Next-Generation Optoelectronic Applications
Advanced Synthesis of Asymmetric Pyrene Derivatives for Next-Generation Optoelectronic Applications
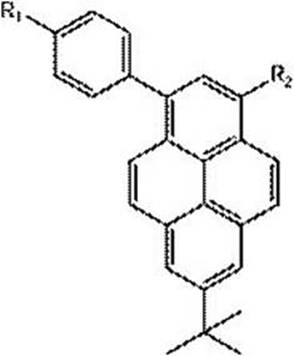
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials with superior color purity and charge transport properties. Patent CN111848413B introduces a groundbreaking strategy for synthesizing pyrene-based derivatives modified along the short axis, specifically targeting the 1- and 3-positions. This innovation addresses the critical limitation of low color purity found in conventional symmetric pyrene derivatives by establishing an asymmetric push-pull electronic structure. By leveraging intramolecular charge transfer states, this technology enables precise tuning of optoelectronic performance, making it a vital asset for manufacturers seeking high-efficiency luminescent materials. As a leading reliable OLED material supplier, understanding these structural nuances is key to developing next-generation display technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of pyrene cores has predominantly focused on symmetric substitution patterns, particularly at the 1,3,6,8-positions or the 2,7-positions. While these methods are well-documented, they often result in materials with compromised color purity due to the inherent symmetry of the molecular orbitals. The lack of asymmetry limits the ability to induce strong intramolecular charge transfer, which is essential for red-shifting emission and enhancing quantum efficiency in organic electroluminescence. Furthermore, achieving high regioselectivity at multiple active sites simultaneously often requires harsh reaction conditions and complex protection-deprotection sequences, leading to lower overall yields and increased waste generation. These inefficiencies pose significant challenges for cost reduction in electronic chemical manufacturing, as the purification of symmetric byproducts becomes increasingly difficult and expensive at scale.
The Novel Approach
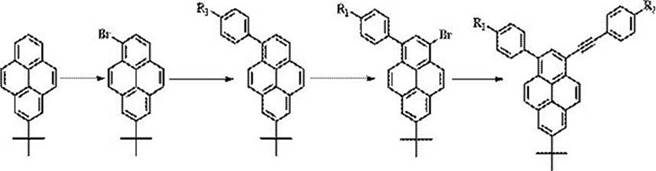
The methodology disclosed in the patent revolutionizes this landscape by proposing a stepwise functionalization strategy specifically along the short axis of the pyrene molecule. By selectively introducing substituents with different electronic properties at the 1- and 3-positions, the synthesis creates a distinct donor-acceptor architecture within a single molecule. This asymmetric design effectively breaks the molecular symmetry, facilitating a robust intramolecular charge transfer that significantly enhances the material's fluorescence quantum yield and thermal stability. The approach utilizes 7-tert-butylpyrene as a stable starting scaffold, allowing for controlled mono-bromination followed by sequential cross-coupling reactions. This logical progression not only simplifies the synthetic route but also ensures high regioselectivity, thereby minimizing the formation of unwanted isomers and streamlining the purification process for commercial scale-up of complex polymer additives and small molecule emitters.
Mechanistic Insights into Selective Bromination and Cross-Coupling
The core of this synthesis lies in the precise control of electrophilic aromatic substitution and transition metal-catalyzed coupling. The initial step involves the selective bromination of 7-tert-butylpyrene, where the steric bulk of the tert-butyl group directs the incoming bromine species primarily to the 1-position. Utilizing reagents such as N-bromosuccinimide (NBS) or benzyltrimethylammonium tribromide in dichloromethane at controlled temperatures between 0°C and 40°C ensures high selectivity. Following this, a Suzuki-Miyaura cross-coupling reaction installs the first aryl group, utilizing palladium catalysts like tetrakis(triphenylphosphine)palladium. The subsequent second bromination targets the 3-position, activated by the existing substituents, before a final Sonogashira coupling introduces the alkynyl moiety. This sequence creates a conjugated system that extends the pi-electron cloud effectively.
Impurity control is paramount in the production of high-purity OLED material, and this mechanism offers inherent advantages. The stepwise nature of the synthesis allows for intermediate purification via column chromatography after each critical transformation, preventing the carryover of impurities into the final product. The use of specific brominating agents like bromine water or benzyltrimethylammonium tribromide has been verified to offer superior selectivity compared to elemental bromine, reducing the formation of poly-brominated side products. Additionally, the inert gas protection during coupling reactions prevents oxidative degradation of the sensitive palladium catalysts and the pyrene core. This rigorous control over reaction parameters ensures that the final asymmetric pyrene derivatives meet stringent purity specifications required for high-performance optoelectronic devices, minimizing defects in the final display panels.
How to Synthesize 1-phenyl-3-(4-N,N-dimethylphenylethynyl)-7-tert-butylpyrene Efficiently
The synthesis of this specific high-performance derivative exemplifies the robustness of the patented method. Starting from 7-tert-butylpyrene, the process involves a sequence of bromination, Suzuki coupling, a second bromination, and finally a Sonogashira coupling. Each step is optimized for yield and purity, utilizing common laboratory solvents and commercially available catalysts. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios, temperature controls, and workup procedures necessary to achieve the reported yields of up to 88% for intermediate bromination steps. This protocol serves as a blueprint for scaling the production of advanced luminescent materials.
- Perform selective bromination of 7-tert-butylpyrene using brominating agents like NBS or bromine water in dichloromethane at 0-40°C to obtain 1-bromo-7-tert-butylpyrene.
- Conduct a Suzuki cross-coupling reaction with arylboronic acids using a palladium catalyst to introduce the first substituent at the 1-position.
- Execute a second selective bromination at the 3-position followed by a Sonogashira coupling to install the final alkynyl group, completing the asymmetric structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits regarding cost efficiency and operational reliability. The reliance on 7-tert-butylpyrene as a starting material leverages an existing supply chain for polycyclic aromatic hydrocarbons, ensuring raw material availability without the need for exotic precursors. The reaction conditions are relatively mild, particularly the bromination steps which proceed at near-ambient temperatures, reducing energy consumption compared to high-temperature pyrolysis methods often used in PAH functionalization. Furthermore, the use of standard catalysts and solvents means that existing manufacturing infrastructure can be adapted with minimal capital expenditure, facilitating a smoother transition from pilot scale to full commercial production.
- Cost Reduction in Manufacturing: The elimination of complex protection groups and the high selectivity of the bromination steps significantly reduce the volume of waste solvents and silica gel required for purification. By avoiding the formation of difficult-to-separate symmetric isomers, the overall process mass intensity is lowered, leading to substantial cost savings in downstream processing. The high yields reported in the patent examples, such as 83% for the initial bromination and 76% for the Suzuki coupling, indicate a material-efficient process that maximizes the output from expensive pyrene feedstocks.
- Enhanced Supply Chain Reliability: The reagents specified, including palladium catalysts, phosphine ligands, and common organic solvents like toluene and dichloromethane, are commodity chemicals with stable global supply chains. This reduces the risk of production delays caused by the shortage of specialized reagents. Additionally, the stability of the intermediates, such as 1-bromo-7-tert-butylpyrene, allows for potential stockpiling or semi-finished goods inventory, providing flexibility in meeting fluctuating market demand for display materials without compromising on lead times.
- Scalability and Environmental Compliance: The synthetic route avoids the use of highly toxic or regulated reagents that would complicate environmental permitting. The waste streams generated are typical of standard organic synthesis and can be managed through established treatment protocols. The simplicity of the four-step sequence makes it highly amenable to continuous flow chemistry or large-batch reactor processing, ensuring that the technology can be scaled to meet the growing demand for high-resolution displays and lighting applications while maintaining strict environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrene derivative synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of this approach for industrial partners.
Q: What is the primary advantage of short-axis modification in pyrene derivatives?
A: Short-axis modification at the 1,3-positions allows for the construction of asymmetric push-pull electronic structures, which induce intramolecular charge transfer. This significantly improves color purity and charge transport capabilities compared to traditional symmetric substitutions.
Q: How does this synthesis method improve supply chain stability?
A: The process utilizes commercially available reagents like 7-tert-butylpyrene and standard brominating agents. The mild reaction conditions and high selectivity reduce the need for complex purification, ensuring consistent batch-to-batch quality and reliable delivery.
Q: Can this method be scaled for industrial production of OLED materials?
A: Yes, the synthesis relies on robust reactions such as Suzuki and Sonogashira couplings which are well-established in industrial settings. The use of common solvents like dichloromethane and toluene facilitates straightforward scale-up from laboratory to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrene Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical expertise to optimize this specific pyrene functionalization route for your unique application needs. Our rigorous QC labs and stringent purity specifications ensure that every batch of asymmetric pyrene derivatives meets the exacting standards required for high-end optoelectronic applications. We understand that consistency is key in the display industry, and our robust process control systems guarantee batch-to-batch reproducibility.
We invite you to collaborate with us to unlock the full potential of these high-efficiency luminescent materials. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and manufacturing goals, ensuring a seamless integration of these advanced materials into your supply chain.
