Scalable Synthesis of Isoindolinone Dipeptides via Heterogeneous MOF-Cu Catalysis for Industrial Applications
Introduction to Advanced Peptide Mimetic Synthesis
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more sustainable and efficient synthetic routes. A significant breakthrough in this domain is documented in patent CN110372774B, which details a novel method for synthesizing isoindolinone-substituted α-acyloxyamide dipeptide derivatives. These compounds serve as critical scaffolds in the development of bioactive molecules, particularly exhibiting potent inhibitory activity against agricultural pathogens such as Penicillium digitatum and Rhizoctonia solani. The core innovation lies in the utilization of a Metal-Organic Framework supported copper catalyst (MOF-Cu) to facilitate a three-component Ugi reaction. This approach represents a paradigm shift from traditional multi-step syntheses, offering a streamlined, one-pot methodology that operates under remarkably mild conditions. By leveraging the unique porous structure and Lewis acidity of the MOF-Cu material, the process achieves high atom economy and selectivity, making it an attractive candidate for the commercial scale-up of complex agrochemical intermediates.
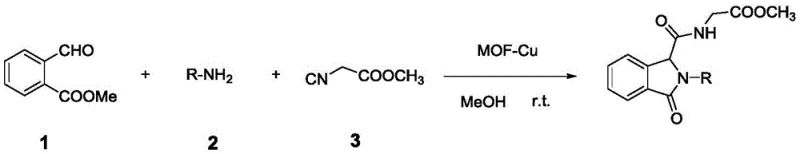
The strategic implementation of this technology addresses several pain points inherent in conventional peptide chemistry. Traditional methods often suffer from low yields, harsh reaction conditions requiring extreme temperatures or pressures, and the generation of substantial chemical waste. In contrast, the MOF-Cu catalyzed route described in the patent utilizes readily available starting materials—aldehydes, amines, and isonitriles—to construct the dipeptide backbone efficiently. The reaction proceeds at room temperature in environmentally benign solvents like methanol or ethanol, significantly reducing the energy footprint of the manufacturing process. Furthermore, the heterogeneous nature of the catalyst allows for straightforward separation and recycling, a feature that is crucial for maintaining cost-effectiveness and supply chain continuity in large-scale operations. This technological advancement positions manufacturers as a reliable agrochemical intermediate supplier capable of delivering high-purity products with reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted dipeptide derivatives has been fraught with challenges that hinder industrial viability. Conventional pathways frequently rely on homogeneous transition metal catalysts or stoichiometric amounts of activating agents, which complicate downstream processing. The removal of residual metal contaminants from the final product often necessitates expensive and time-consuming purification steps, such as specialized scavenging resins or extensive chromatography, which drastically inflate production costs. Moreover, many traditional protocols require anhydrous conditions and inert atmospheres to prevent catalyst deactivation or side reactions, imposing strict operational constraints that increase capital expenditure and energy consumption. The use of volatile organic compounds (VOCs) as solvents in these legacy processes also raises significant environmental and safety concerns, conflicting with modern green chemistry principles. Additionally, the lack of catalyst recyclability in homogeneous systems means that valuable metals are lost after a single run, contributing to resource depletion and higher raw material costs for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology outlined in patent CN110372774B offers a robust solution to these longstanding inefficiencies by employing a heterogeneous MOF-Cu catalyst. This innovative approach enables the direct assembly of isoindolinone-substituted α-acyloxyamide dipeptide derivatives through a tandem cyclization-Ugi sequence in a single reaction vessel. The MOF-Cu catalyst, composed of divalent copper ions and terphenyltetracarboxylic acid, provides a stable and reusable platform that maintains its structural integrity throughout the reaction cycle. One of the most compelling advantages of this novel route is its operational simplicity; the reaction proceeds smoothly at room temperature without the need for rigorous exclusion of moisture or oxygen. The use of green solvents such as water or alcohols further enhances the sustainability profile of the process. Post-reaction, the solid catalyst can be recovered simply by filtration, washed, and dried for subsequent reuse, as evidenced by experimental data showing consistent catalytic performance over multiple cycles. This eliminates the need for complex metal removal procedures, thereby streamlining the workflow and enhancing the overall economic feasibility of producing high-purity OLED material precursors or agrochemical actives.
Mechanistic Insights into MOF-Cu Catalyzed Ugi Reaction
Understanding the mechanistic underpinnings of this transformation is essential for R&D directors evaluating process robustness. The MOF-Cu catalyst functions primarily through Lewis acid activation, where the coordinatively unsaturated copper sites within the metal-organic framework interact with the carbonyl oxygen of the aldehyde component. This interaction increases the electrophilicity of the carbonyl carbon, facilitating the nucleophilic attack by the amine to form the imine intermediate, a critical step in the Ugi four-component reaction (though here adapted to a three-component variant involving an internal nucleophile). The porous architecture of the MOF not only stabilizes these reactive intermediates but also imposes a degree of steric control that can enhance regioselectivity. Unlike homogeneous copper salts which may aggregate or precipitate, the copper ions in the MOF are spatially isolated within the rigid organic linker matrix, preventing deactivation pathways and ensuring uniform catalytic activity throughout the reaction mixture. This structural stability is key to the catalyst's longevity and reusability, attributes that are often lacking in traditional homogeneous systems.
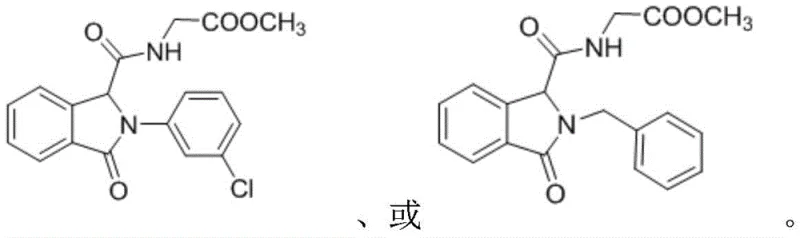
Furthermore, the mechanism involves an intramolecular cyclization that constructs the isoindolinone core concurrently with the formation of the peptide bond. The proximity effect within the MOF pores likely aids in bringing the reacting species into close contact, accelerating the reaction kinetics even at ambient temperatures. The patent data highlights the versatility of this system, accommodating a wide range of substituents on the amine component, including benzyl, alkyl, and various aryl groups, as illustrated by the successful synthesis of compounds 5a and 5b. The ability to tolerate diverse functional groups without compromising yield or purity demonstrates the broad substrate scope of this catalytic system. For quality control purposes, the impurity profile is significantly cleaner compared to acid-catalyzed variants, as the mild conditions minimize decomposition or polymerization of sensitive intermediates. This results in a final product that meets stringent purity specifications with minimal downstream purification, a critical factor for reducing lead time for high-purity agrochemical intermediates.
How to Synthesize Isoindolinone Dipeptide Derivatives Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory scale optimization and pilot plant production. The process begins with the preparation of the aldehyde precursor, methyl 2-formylbenzoate, which is readily synthesized from 2-formylbenzoic acid. In the main reaction step, the aldehyde and the chosen amine are dissolved in a protic solvent such as methanol. The MOF-Cu catalyst is then introduced to the mixture, followed by the addition of the isonitrile component. The reaction is allowed to stir at room temperature for a period ranging from 12 to 24 hours, monitored by TLC to ensure complete conversion. Upon completion, the heterogeneous catalyst is removed via simple filtration, and the solvent is evaporated under reduced pressure. The crude residue is then subjected to standard column chromatography to isolate the pure target compound. Detailed standardized synthesis steps are provided in the guide below.
- Preparation of Methyl 2-formylbenzoate by reacting 2-formylbenzoic acid with ethanol and concentrated sulfuric acid at 10-90°C.
- Dissolving methyl 2-formylbenzoate and amine in methanol, ethanol, or water at room temperature, followed by the addition of MOF-Cu catalyst.
- Adding isonitrile last, stirring for 12-24 hours, filtering the reusable catalyst, and purifying the residue via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this MOF-Cu catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly impacts the bottom line. By eliminating the need for expensive homogeneous catalysts and the associated metal scavenging steps, the process significantly reduces raw material costs and waste disposal fees. The ability to recycle the MOF-Cu catalyst multiple times without loss of activity means that the effective cost of the catalyst per kilogram of product is negligible, driving down the overall cost of goods sold (COGS). Furthermore, the use of common, low-cost solvents like methanol and ethanol, along with the avoidance of cryogenic conditions or high-pressure equipment, lowers the barrier to entry for production and reduces energy consumption. These factors collectively contribute to substantial cost savings and improved margin potential for high-volume manufacturing.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the reusability of the MOF-Cu catalyst. In traditional homogeneous catalysis, the metal catalyst is often lost in the aqueous workup or requires complex extraction, representing a sunk cost for every batch. Here, the solid catalyst is filtered, washed, and dried for immediate reuse, as validated by patent data showing consistent yields over six consecutive runs. This circular usage model decouples production volume from catalyst consumption, leading to a flatter cost curve as scale increases. Additionally, the mild reaction conditions eliminate the need for specialized high-energy reactors or extensive cooling systems, further reducing utility costs. The simplified purification process, necessitated by the cleaner reaction profile, also reduces the consumption of silica gel and eluents, adding another layer of cost efficiency to the operation.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the synthetic route. The reliance on commodity chemicals—simple aldehydes, amines, and isonitriles—ensures that raw material sourcing is not a bottleneck, as these are widely available from multiple global vendors. The tolerance of the reaction to ambient conditions means that production is less susceptible to disruptions caused by utility failures or equipment malfunctions associated with extreme process parameters. Moreover, the scalability of the reaction has been proven, with patent examples demonstrating successful expansion of the reaction scale by tenfold while maintaining high yields. This predictability allows for more accurate demand forecasting and inventory planning, ensuring a steady flow of reliable agrochemical intermediate supplier products to meet market needs without unexpected delays.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology aligns perfectly with modern regulatory standards. The replacement of hazardous chlorinated solvents with alcohols or water reduces the facility's VOC emissions and fire risk. The heterogeneous nature of the catalyst minimizes heavy metal contamination in the wastewater stream, simplifying effluent treatment and ensuring compliance with strict environmental discharge limits. The high atom economy of the Ugi reaction means that a greater proportion of the starting materials ends up in the final product, reducing the generation of chemical waste that requires costly disposal. These green chemistry attributes not only mitigate regulatory risk but also enhance the corporate sustainability profile, which is increasingly important for securing contracts with major multinational corporations that prioritize responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this MOF-Cu catalyzed synthesis. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering a transparent view of the technology's capabilities and limitations. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the advantages of using MOF-Cu catalyst over traditional homogeneous catalysts?
A: The MOF-Cu catalyst is heterogeneous, allowing for easy separation via simple filtration. Unlike homogeneous catalysts, it can be recycled and reused multiple times without significant loss of catalytic activity, as demonstrated by stable yields over six cycles in the patent data.
Q: What are the typical reaction conditions for this Ugi transformation?
A: The reaction proceeds under mild conditions at room temperature in green solvents such as methanol, ethanol, or water. This eliminates the need for high energy input or hazardous reaction environments typically associated with traditional peptide synthesis.
Q: Can this synthesis method be scaled up for industrial production?
A: Yes, the patent data indicates successful scale-up experiments where the reaction was expanded 10-fold with maintained yields (83%). The simple workup procedure involving filtration and solvent removal supports robust commercial scale-up of complex agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoindolinone Dipeptide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the MOF-Cu catalyzed Ugi reaction in the production of advanced peptide mimetics and agrochemical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into a robust, commercial-scale manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench to plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch conforms to the highest industry standards. Our capability to handle complex heterocyclic synthesis, combined with our dedication to green chemistry principles, makes us the ideal partner for bringing these innovative dipeptide derivatives to market.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific application needs. Whether you require custom synthesis for drug discovery or bulk manufacturing for agrochemical formulations, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your project. We encourage you to reach out to our experts to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability.
