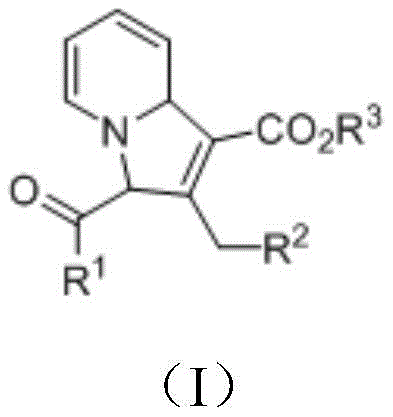
Scalable Synthesis of High-Purity Indolizine Derivatives via Metal-Free [3+2] Cycloaddition
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective routes to synthesize complex heterocyclic scaffolds. Patent CN111909148A introduces a groundbreaking methodology for the preparation of indolizine derivatives, a class of nitrogen-containing heterocycles renowned for their significant biological and pharmaceutical activities. This innovation addresses critical bottlenecks in traditional synthesis by employing a metal-free [3+2] cycloaddition reaction between a brominated pyridinium salt and a dienoic acid ester. Unlike conventional methods that often rely on harsh conditions or expensive transition metals, this process operates under mild temperatures ranging from 15-40°C and utilizes readily available inorganic bases and Lewis base promoters. For R&D directors and procurement specialists, this represents a paradigm shift towards greener chemistry that does not compromise on yield or purity, offering a robust pathway for generating high-value intermediates used in drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized indolizine derivatives has been plagued by significant operational complexities and economic inefficiencies. Traditional routes frequently depend on transition metal catalysts, which introduce severe challenges regarding product purity due to metal residue contamination. Removing these trace metals often requires additional, costly purification steps such as chromatography or specialized scavenging resins, which drastically increases the overall production cost and extends lead times. Furthermore, many existing methods demand rigorous reaction conditions, including high temperatures or inert atmospheres that are difficult to maintain on a large scale. The reliance on expensive, specialized reagents and the generation of substantial chemical waste further diminish the atom economy, making these processes less attractive for commercial-scale manufacturing where margin compression is a constant concern for supply chain managers.
The Novel Approach
The methodology disclosed in patent CN111909148A offers a transformative solution by leveraging a [3+2] cycloaddition strategy that is both atom-economical and operationally simple. By utilizing a brominated pyridinium salt and a dienoic acid ester as primary substrates, the reaction avoids the use of transition metals entirely, thereby eliminating the risk of heavy metal contamination in the final API intermediate. The reaction is promoted by a Lewis base and catalyzed by an inexpensive inorganic base, proceeding efficiently at room temperature or slightly above (15-40°C). This mildness not only enhances safety profiles but also reduces energy consumption significantly. The simplicity of the substrate synthesis and the high efficiency of the cycloaddition step result in superior yields, with some embodiments reporting yields up to 80% or higher, providing a reliable and scalable alternative for the production of high-purity indolizine derivatives.
![Reaction scheme of [3+2] cycloaddition between compound II and III](/insights/img/indolizine-derivative-synthesis-pharma-supplier-20260305085310-07.webp)
Mechanistic Insights into Lewis Base-Promoted [3+2] Cycloaddition
The core of this technological advancement lies in the synergistic interaction between the inorganic base catalyst and the Lewis base promoter. The reaction initiates with the activation of the dienoic acid ester (Compound III) by the Lewis base, which facilitates the formation of a reactive zwitterionic intermediate or enhances the nucleophilicity of the allenoate species. Simultaneously, the inorganic base, such as potassium carbonate or sodium bicarbonate, helps to neutralize acidic byproducts and maintain the optimal pH environment for the cycloaddition to proceed. This dual-catalyst system ensures that the [3+2] cyclization occurs with high regioselectivity and stereoselectivity, minimizing the formation of unwanted isomers or side products. The mechanism effectively bypasses the high energy barriers associated with thermal cycloadditions, allowing the reaction to reach completion within a short timeframe of 2-5 hours. This mechanistic elegance translates directly to process robustness, as the reaction tolerates a wide variety of substituents on the phenyl and heteroaryl rings without significant loss in efficiency.
From an impurity control perspective, the absence of transition metals is a decisive advantage for pharmaceutical applications. Metal residues are a major regulatory concern, often requiring stringent limits (e.g., ppm levels) in final drug substances. By designing a synthetic route that inherently excludes these contaminants, the purification burden is shifted from complex metal scavenging to standard organic workups like extraction and crystallization. Furthermore, the high atom economy of the [3+2] cycloaddition means that a larger proportion of the starting material mass is incorporated into the final product, reducing the volume of waste solvent and byproducts generated per kilogram of product. This cleaner reaction profile simplifies the isolation of the indolizine derivative, often allowing for high-purity isolation via simple column chromatography or recrystallization, ensuring that the final material meets the rigorous quality standards required for clinical and commercial use.
How to Synthesize Indolizine Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear, three-stage workflow that is amenable to both laboratory optimization and industrial scale-up. The process begins with the preparation of the key electrophilic partner, the brominated pyridinium salt, followed by the synthesis of the nucleophilic allenoic acid ester. These two precursors are then combined in the presence of the catalytic system to effect the ring closure. The detailed standardized synthesis steps below provide a comprehensive guide for replicating this high-yielding transformation, ensuring consistency and reproducibility across different batches and production scales.
- Prepare brominated 1-(2-oxo-2-arylethyl) pyridinium salts by reacting pyridine with alpha-bromoacetophenone in ether.
- Synthesize allenoic acid esters using alkoxyacyl methyl triphenylphosphine bromide, triethylamine, and acyl chloride in dichloromethane.
- Perform the [3+2] cycloaddition by mixing the pyridinium salt and allenoic ester with an inorganic base and Lewis base promoter in solvent at 15-40°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of precious metal catalysts removes a volatile cost component from the bill of materials, as prices for metals like palladium or rhodium can fluctuate wildly based on geopolitical factors. Additionally, the use of commodity chemicals such as pyridine, alpha-bromo ketones, and simple inorganic bases ensures a stable and diverse supply chain, reducing the risk of single-source dependency. The mild reaction conditions also imply lower energy costs and reduced wear on reactor equipment, contributing to a lower total cost of ownership for the manufacturing process. These factors collectively enhance the commercial viability of producing indolizine-based intermediates, making it a highly attractive option for long-term supply contracts.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of expensive transition metal catalysts and ligands from the process. In traditional pharmaceutical manufacturing, the cost of catalysts and the subsequent purification steps to remove metal traces can account for a substantial portion of the COGS (Cost of Goods Sold). By switching to an inorganic base and Lewis base system, manufacturers can achieve drastic cost savings on raw materials. Furthermore, the high atom economy and reduced waste generation lower the costs associated with waste disposal and solvent recovery. The simplified purification process, often requiring fewer chromatographic steps, also reduces labor and consumable costs, leading to a leaner and more profitable production model for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade starting materials significantly de-risks the supply chain. Pyridine, acyl chlorides, and inorganic bases are produced globally in massive quantities, ensuring that supply disruptions are unlikely to impact production schedules. This contrasts sharply with specialized organometallic reagents that may have limited suppliers and long lead times. The robustness of the reaction conditions (15-40°C) also means that the process can be run in a wider variety of manufacturing facilities without requiring specialized high-temperature or high-pressure equipment. This flexibility allows for greater agility in sourcing and production planning, ensuring consistent delivery of critical intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this methodology is inherently designed for scalability. The exothermic nature of the reaction is manageable under mild conditions, reducing the risk of thermal runaway incidents common in high-temperature syntheses. From an environmental standpoint, the absence of heavy metals simplifies wastewater treatment and compliance with increasingly strict environmental regulations regarding metal discharge. The high yield and selectivity minimize the formation of byproducts, aligning with green chemistry principles and reducing the facility's overall environmental footprint. This makes the process not only easier to scale from kilograms to tons but also more sustainable, which is a growing priority for corporate social responsibility goals in the chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this indolizine synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in patent CN111909148A, offering clarity on the practical aspects of adopting this novel route. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows.
Q: Does this synthesis method involve transition metal catalysts?
A: No, this novel method utilizes an inorganic base catalyst and a Lewis base promoter, completely eliminating the need for expensive and difficult-to-remove transition metal catalysts.
Q: What are the typical reaction conditions for this cycloaddition?
A: The reaction proceeds under mild conditions, typically at temperatures between 15-40°C for 2-5 hours, ensuring high energy efficiency and safety.
Q: What is the purity profile of the resulting indolizine derivatives?
A: The process yields high-purity products with no catalyst residue, significantly simplifying downstream purification and making it ideal for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free [3+2] cycloaddition technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to pilot plant is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including precise temperature control for the mild cycloaddition steps and rigorous QC labs capable of verifying stringent purity specifications. We are committed to delivering indolizine derivatives that meet the highest quality standards, free from metal residues and ready for immediate use in drug development pipelines.
We invite potential partners to engage with our technical team to explore how this innovative synthesis route can optimize your supply chain and reduce manufacturing costs. By leveraging our expertise in process chemistry and scale-up, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and discover how NINGBO INNO PHARMCHEM can serve as your trusted partner for the reliable supply of complex heterocyclic intermediates.
