Revolutionizing Chiral Alcohol Synthesis with Advanced Monochiral Center Ruthenium Catalysts for Commercial Scale
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift in the synthesis of chiral compounds, driven by the urgent demand for more efficient and cost-effective catalytic systems. Patent CN112675920B introduces a groundbreaking class of monochiral center catalysts that address the longstanding challenges associated with producing high-purity chiral secondary alcohols, which serve as critical intermediates in the development of modern therapeutics. With over two-thirds of new drug candidates being chiral in nature, the ability to synthesize these molecules with high stereocontrol is paramount for ensuring drug efficacy and safety. This innovative technology leverages a unique Ruthenium-based coordination complex that simplifies the catalyst architecture while enhancing performance, offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates. 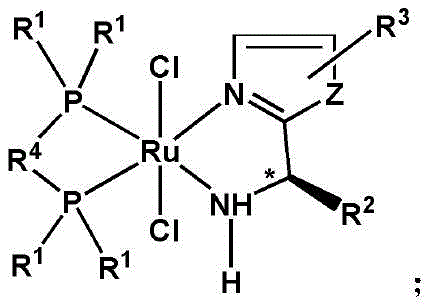
Traditionally, the asymmetric hydrogenation of ketones to chiral alcohols has relied heavily on sophisticated catalytic systems such as those developed by Noyori, which necessitate the precise matching of multiple chiral elements within the ligand framework. These conventional methods often require the synthesis of expensive chiral bisphosphine ligands possessing axial chirality, alongside chiral diamine ligands, creating a significant barrier to entry due to high material costs and complex preparation protocols. Furthermore, the sensitivity of these multi-chiral systems to mismatched configurations can lead to drastic reductions in enantiomeric excess, complicating process optimization and quality control in large-scale manufacturing environments. The reliance on such intricate ligand designs not only inflates the cost of goods but also introduces supply chain vulnerabilities related to the availability of specialized chiral starting materials.
In stark contrast, the novel approach detailed in this patent utilizes a streamlined catalyst design that relies on a single chiral center to govern the stereoselectivity of the reaction, paired with achiral bidentate phosphine ligands that enhance stability and solubility. This strategic simplification eliminates the need for costly axially chiral phosphines, thereby drastically reducing the economic burden of catalyst production without compromising catalytic activity or selectivity. The new system operates through an outer-sphere mechanism where the metal center does not directly coordinate with the carbonyl oxygen, instead facilitating hydrogen transfer via a six-membered transition state that is tightly controlled by the single chiral amine moiety. 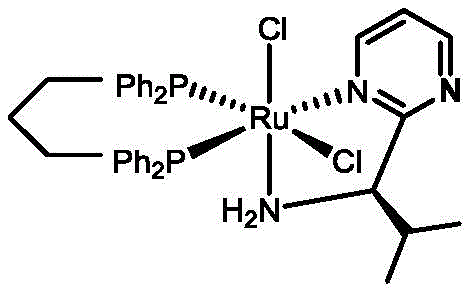
The mechanistic insights into this Ruthenium-catalyzed asymmetric transfer hydrogenation reveal a highly efficient pathway for converting prochiral ketones into optically active alcohols with exceptional fidelity. The catalyst functions by activating the hydrogen source, typically isopropanol, through a metal-ligand bifunctional mechanism where the amine proton and the hydride on the ruthenium center are transferred simultaneously to the carbonyl group. The presence of the single chiral center adjacent to the coordinating nitrogen atom creates a well-defined chiral pocket that effectively differentiates between the enantiotopic faces of the ketone substrate, even when the substituents possess similar steric and electronic properties. 
This precise control allows for the successful transformation of challenging substrates such as aryl-aryl and heterocyclic-heterocyclic ketones, which have historically been difficult to reduce with high enantioselectivity using standard catalytic methods. The robustness of the catalyst is further evidenced by its ability to maintain high turnover numbers and enantiomeric excess values exceeding 90% across a broad range of substrates, ensuring consistent product quality essential for regulatory compliance in pharmaceutical manufacturing. Additionally, the system demonstrates remarkable versatility by enabling the kinetic resolution of racemic alpha-allyl alcohols, expanding its utility beyond simple hydrogenation to include dynamic kinetic processes that maximize atom economy.
How to Synthesize Chiral Alcohols Efficiently
The implementation of this advanced catalytic technology into existing production workflows requires a clear understanding of the preparation and application protocols to ensure optimal results. The synthesis of the catalyst itself is designed to be straightforward, involving the sequential coordination of ligands to a ruthenium precursor under mild conditions, which facilitates easy handling and storage for industrial users. Once prepared, the catalyst is employed in a transfer hydrogenation setup where it activates the hydrogen donor to reduce the target ketone with high precision and speed. For detailed standardized synthesis steps and specific reaction parameters, please refer to the guide below.
- Prepare the monochiral center catalyst by reacting tris(triphenylphosphine)ruthenium dichloride with a chiral pyrimidine or pyridine ligand in dichloromethane, followed by coordination with an achiral bidentate phosphine ligand in toluene.
- Dissolve the prepared catalyst and potassium tert-butoxide in a mixed solvent system of isopropanol and dichloromethane under an inert atmosphere to activate the catalytic species.
- Add the target ketone substrate to the reaction mixture at temperatures between 20°C and 40°C, allowing the asymmetric transfer hydrogenation to proceed for 2 to 15 minutes to yield the chiral alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this monochiral center catalyst technology offers transformative benefits that directly impact the bottom line and operational resilience of chemical manufacturing operations. The most significant advantage lies in the substantial cost reduction achieved by eliminating the requirement for expensive, multi-step synthesized chiral bisphosphine ligands that are characteristic of legacy catalytic systems. By utilizing readily available achiral bidentate phosphine ligands in conjunction with a simpler chiral amine component, manufacturers can secure raw materials more easily and at a fraction of the cost, leading to significant savings in the overall cost of goods sold for chiral intermediates.
- Cost Reduction in Manufacturing: The simplified ligand architecture translates directly into lower procurement costs and reduced waste generation during catalyst synthesis, as fewer synthetic steps are required to prepare the active catalytic species. This efficiency gain allows for a more competitive pricing structure for the final chiral alcohol products, enabling companies to improve their profit margins or offer more attractive pricing to downstream pharmaceutical clients without sacrificing quality. Furthermore, the high catalytic activity means that lower catalyst loadings are required to achieve complete conversion, further minimizing the consumption of precious metal resources and reducing the environmental footprint associated with metal waste disposal.
- Enhanced Supply Chain Reliability: The reliance on commercially available and structurally simple ligand components mitigates the risk of supply chain disruptions that often plague the sourcing of specialized chiral reagents. This stability ensures consistent production schedules and reliable delivery timelines for critical pharmaceutical intermediates, which is essential for maintaining the continuity of drug development pipelines. The robustness of the catalyst also allows for flexibility in sourcing, as the achiral phosphine ligands can often be obtained from multiple suppliers, reducing dependency on single-source vendors and enhancing overall supply chain security.
- Scalability and Environmental Compliance: The reaction conditions employed by this catalytic system are notably mild, typically operating at room temperature or slightly elevated temperatures with short reaction times, which significantly reduces energy consumption during the manufacturing process. This energy efficiency, combined with the use of common solvents like isopropanol and dichloromethane, simplifies the scale-up process from laboratory to commercial production volumes while adhering to strict environmental regulations regarding solvent emissions and energy usage. The high selectivity of the reaction also minimizes the formation of by-products, reducing the burden on downstream purification processes and waste treatment facilities.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their specific applications, we have compiled answers to common inquiries regarding the performance and scope of the monochiral center catalyst. These responses are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for process development decisions. Understanding these key aspects will help R&D and engineering teams assess the potential impact on their current synthetic routes.
Q: What is the primary advantage of this monochiral catalyst over traditional Noyori systems?
A: Unlike traditional Noyori systems that require complex matching of three chiral elements including expensive chiral bisphosphine ligands, this novel catalyst utilizes a single chiral center paired with readily available achiral bidentate phosphine ligands, significantly simplifying synthesis and reducing raw material costs while maintaining high enantioselectivity.
Q: What types of substrates are compatible with this catalytic system?
A: The catalyst demonstrates exceptional versatility, effectively catalyzing the asymmetric reduction of aryl-aryl substituted, heterocyclic aromatic-aryl substituted, and heterocyclic-heterocyclic substituted ketones, which are traditionally challenging substrates due to similar steric and electronic properties of the substituents.
Q: Can this catalyst be used for kinetic resolution of allylic alcohols?
A: Yes, beyond asymmetric hydrogenation, the catalyst is highly effective for the kinetic resolution of racemic alpha-allyl alcohols, achieving high selectivity factors (S > 10) and excellent enantiomeric excess values, providing a non-enzymatic alternative for synthesizing chiral allylic building blocks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monochiral Center Catalyst Supplier
As the global demand for high-purity chiral intermediates continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides the strategic advantage needed to navigate complex synthetic challenges efficiently. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and compliant with international quality standards. We leverage our rigorous QC labs and stringent purity specifications to guarantee that every batch of chiral alcohol or catalyst meets the exacting requirements of the pharmaceutical industry, minimizing the risk of delays in drug registration and market entry.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic technology can be tailored to your specific project needs and to request a Customized Cost-Saving Analysis. By collaborating with us, you can access specific COA data and comprehensive route feasibility assessments that will empower your organization to make informed decisions about adopting this next-generation synthesis method. Let us help you optimize your supply chain and accelerate your time to market with our proven expertise in chiral chemistry and process development.
