Advanced Palladium-Catalyzed Synthesis of Substituted 2,3-Dihydroquinolone Intermediates for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Substituted 2,3-Dihydroquinolone Intermediates for Commercial Scale-Up
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern pharmaceutical process chemistry. Specifically, the 2,3-dihydroquinolone scaffold is a privileged structure found in numerous bioactive molecules, ranging from potent antitumor agents to analgesics. As illustrated in the structural diversity of known bioactive compounds, the ability to access these cores with high flexibility is paramount for drug discovery teams. 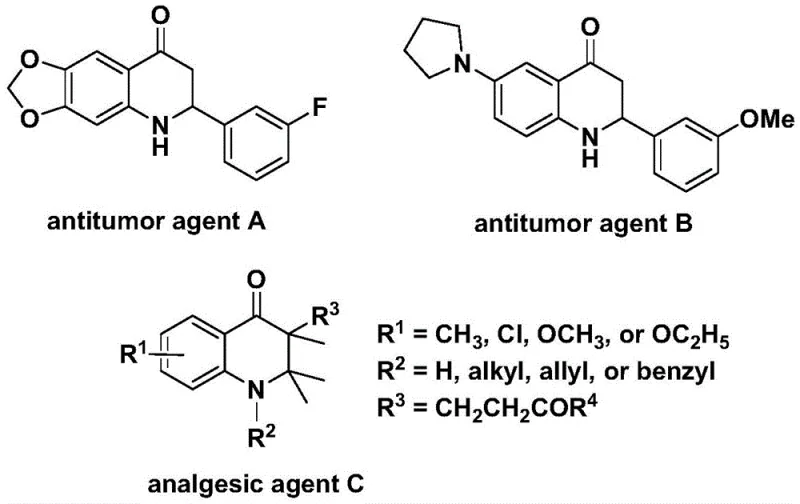 . A recent breakthrough detailed in patent CN112239456B introduces a robust preparation method for substituted 2,3-dihydroquinolone compounds that addresses many historical limitations in carbonylation chemistry. This technology leverages a transition metal palladium-catalyzed system using N-pyridine sulfonyl-o-iodoaniline and olefins as key starting materials. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this methodology represents a significant leap forward in terms of safety, scalability, and substrate tolerance, offering a viable pathway for the commercial production of complex heterocyclic APIs.
. A recent breakthrough detailed in patent CN112239456B introduces a robust preparation method for substituted 2,3-dihydroquinolone compounds that addresses many historical limitations in carbonylation chemistry. This technology leverages a transition metal palladium-catalyzed system using N-pyridine sulfonyl-o-iodoaniline and olefins as key starting materials. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this methodology represents a significant leap forward in terms of safety, scalability, and substrate tolerance, offering a viable pathway for the commercial production of complex heterocyclic APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of carbonyl-containing heterocycles like 2,3-dihydroquinolones has relied heavily on direct carbonylation reactions. While effective in theory, conventional carbonylation often necessitates the use of carbon monoxide gas, which poses severe safety hazards due to its high toxicity and flammability. Handling CO gas requires specialized high-pressure equipment and rigorous safety protocols, which drastically increases capital expenditure and operational complexity for manufacturing facilities. Furthermore, traditional methods frequently suffer from poor substrate compatibility, particularly when dealing with sensitive functional groups that might degrade under harsh thermal or pressure conditions. Many existing protocols also struggle with regioselectivity issues or require expensive and difficult-to-remove catalysts, leading to impure product streams that demand extensive downstream purification. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks in the supply chain for critical pharmaceutical intermediates.
The Novel Approach
The innovative method disclosed in the patent circumvents these challenges by employing a solid carbon monoxide substitute, specifically 1,3,5-trimesic acid phenol ester (TFBen), within a palladium-catalyzed cycle. This approach allows for the generation of CO in situ under mild conditions, effectively eliminating the need for handling gaseous CO. The reaction utilizes N-pyridine sulfonyl-o-iodoaniline and various olefins to construct the dihydroquinolone core through a cascade cyclization-carbonylation sequence. 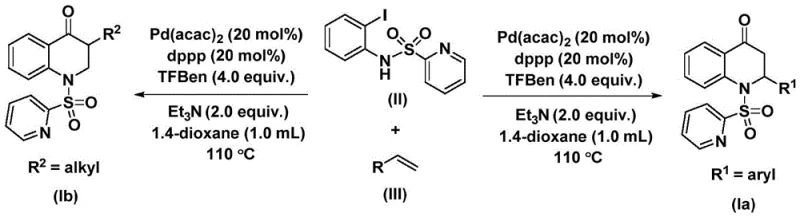 . This novel route is characterized by its operational simplicity, as it proceeds in common organic solvents like 1,4-dioxane at moderate temperatures around 110°C. The method demonstrates exceptional versatility, capable of synthesizing both 2-aryl and 3-alkyl substituted derivatives simply by varying the olefin substrate. This modularity provides medicinal chemists with a powerful tool for rapid structure-activity relationship (SAR) studies while offering process chemists a scalable solution for cost reduction in API manufacturing.
. This novel route is characterized by its operational simplicity, as it proceeds in common organic solvents like 1,4-dioxane at moderate temperatures around 110°C. The method demonstrates exceptional versatility, capable of synthesizing both 2-aryl and 3-alkyl substituted derivatives simply by varying the olefin substrate. This modularity provides medicinal chemists with a powerful tool for rapid structure-activity relationship (SAR) studies while offering process chemists a scalable solution for cost reduction in API manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The catalytic cycle begins with the oxidative addition of the palladium catalyst, specifically bis(acetylacetone)palladium coordinated with the dppp ligand, into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a reactive aryl-palladium intermediate, which is the entry point for the carbonylation sequence. Subsequently, carbon monoxide, released thermally from the TFBen additive, inserts into the palladium-carbon bond to form an acyl-palladium species. This insertion is the critical carbonyl-forming step that distinguishes this process from simple coupling reactions. Following CO insertion, the olefin substrate coordinates to the palladium center and undergoes migratory insertion into the acyl-palladium bond. This step establishes the new carbon-carbon bond at the 3-position of the quinolone ring, determining whether the final product is a 2-aryl or 3-alkyl derivative depending on the olefin used. The cycle concludes with an intramolecular nucleophilic attack or reductive elimination that closes the ring and regenerates the active palladium catalyst, releasing the substituted 2,3-dihydroquinolone product.
From an impurity control perspective, the use of the pyridine sulfonyl group serves a dual purpose: it acts as a protecting group for the nitrogen atom and facilitates the cyclization process through electronic activation. The choice of ligands, such as 1,3-bis(diphenylphosphino)propane (dppp), is crucial for stabilizing the palladium intermediates and preventing the formation of palladium black, which can lead to catalyst deactivation and metal contamination in the final product. The reaction conditions, including the use of triethylamine as a base, help to neutralize acidic byproducts and drive the equilibrium towards product formation. By optimizing the molar ratios of the catalyst, ligand, and CO source, the process achieves high conversion rates with minimal side reactions. This mechanistic understanding allows for precise tuning of reaction parameters to maximize yield and purity, ensuring that the resulting intermediates meet the stringent quality standards required for pharmaceutical applications.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
To implement this synthesis effectively, one must adhere to the optimized conditions described in the patent examples, which balance reaction rate with product integrity. The process involves charging a reaction vessel with the palladium catalyst, ligand, base, CO source, and substrates in an aprotic solvent like dioxane. The mixture is then heated to promote the catalytic cycle. Detailed standard operating procedures for this synthesis are provided below to ensure reproducibility and safety.
- Combine palladium bis(acetylacetonate), dppp ligand, triethylamine, TFBen (CO source), N-pyridine sulfonyl-o-iodoaniline, and the desired olefin substrate in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature between 100°C and 120°C, typically 110°C, and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the reaction mixture, adsorb the crude product onto silica gel, and purify using column chromatography to isolate the high-purity substituted 2,3-dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits beyond mere chemical novelty. The shift from gaseous CO to a solid surrogate fundamentally alters the risk profile of the manufacturing process, reducing insurance costs and regulatory burdens associated with toxic gas handling. This safety improvement translates directly into enhanced supply chain reliability, as production is less susceptible to shutdowns caused by safety incidents or strict environmental inspections. Furthermore, the broad substrate scope means that a single platform technology can be used to produce a wide variety of intermediates, simplifying inventory management and reducing the need for multiple specialized production lines.
- Cost Reduction in Manufacturing: The elimination of high-pressure CO gas infrastructure significantly lowers capital expenditure requirements for new production facilities. Additionally, the use of commercially available and relatively inexpensive starting materials, such as simple olefins and iodoanilines, drives down raw material costs. The high efficiency of the palladium catalyst system, combined with the ability to recycle or minimize catalyst loading through optimization, further reduces the cost per kilogram of the final intermediate. By avoiding complex multi-step sequences often required in traditional syntheses, this one-pot carbonylative cyclization streamlines the manufacturing workflow, leading to substantial overall cost savings in the production of high-value heterocyclic compounds.
- Enhanced Supply Chain Reliability: The robustness of this reaction against various functional groups ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream API manufacturers. The use of stable, shelf-stable reagents like TFBen eliminates the logistical challenges associated with sourcing and storing hazardous gases. This stability allows for better planning and forecasting, reducing the risk of stockouts. Moreover, the scalability of the process from gram to kilogram levels, as demonstrated in the patent data, assures partners that the technology can seamlessly transition from pilot plant to full commercial production without significant re-engineering, thereby securing long-term supply continuity.
- Scalability and Environmental Compliance: The reaction operates in common organic solvents and generates manageable waste streams compared to traditional heavy-metal mediated oxidations. The simplified workup procedure, involving filtration and standard column chromatography or crystallization, minimizes solvent consumption and energy usage. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations. The ability to scale this process efficiently means that large-volume demands can be met without proportionally increasing the environmental footprint, making it a sustainable choice for the long-term manufacturing of pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the specific experimental data and beneficial effects outlined in the patent documentation. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing supply chain.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-trimethoxybenzene phenol ester) acts as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous CO gas, thereby significantly enhancing operational safety and simplifying reactor requirements for industrial scale-up.
Q: What types of substrates are compatible with this palladium-catalyzed method?
A: The method exhibits excellent substrate compatibility, successfully accommodating both aryl-substituted olefins (yielding 2-aryl derivatives) and alkyl-substituted olefins (yielding 3-alkyl derivatives), including those with sensitive functional groups like halogens and silyl groups.
Q: What are the typical reaction conditions required for high yields?
A: Optimal results are achieved using Pd(acac)2 (20 mol%) and dppp (20 mol%) in 1,4-dioxane at 110°C for 48 hours, utilizing triethylamine as a base and TFBen as the carbonyl source to achieve yields up to 88%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the palladium-catalyzed carbonylation described in CN112239456B. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into robust industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic synthesis positions us as a preferred partner for global pharmaceutical companies seeking to optimize their supply chains.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential clients to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a reliable supply of high-quality 2,3-dihydroquinolone derivatives while achieving your cost and efficiency targets.
