Advanced Synthetic Strategy for N-Boc-piperazine: Enhancing Purity and Commercial Scalability
Advanced Synthetic Strategy for N-Boc-piperazine: Enhancing Purity and Commercial Scalability
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for synthesizing critical heterocyclic intermediates, particularly those serving as building blocks for antitussives, antiallergics, and antipsychotic drugs. A significant technological breakthrough in this domain is documented in Chinese Patent CN113185478A, which discloses a novel preparation method for N-tert-butyloxycarbonyl piperazine (N-Boc-piperazine). Unlike traditional methods that rely on the direct acylation of pre-formed piperazine rings, this innovative approach utilizes ethylenediamine as the primary starting material. Through a sophisticated sequence of ring-closing, acylation, and reduction reactions, the process achieves exceptional control over the molecular architecture. This shift in synthetic strategy addresses long-standing challenges regarding selectivity and atom economy, positioning this technology as a vital asset for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N-Boc-piperazine has relied heavily on the direct reaction between piperazine and di-tert-butyl dicarbonate (Boc2O). As illustrated in the conventional reaction pathway below, this method typically requires a substantial molar excess of anhydrous piperazine, often ranging from 3 to 5 equivalents, to suppress the formation of the undesired bis-Boc-piperazine byproduct.
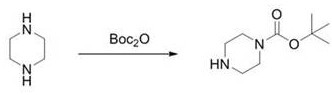
While this one-step synthesis appears operationally simple, it suffers from severe economic and technical drawbacks. The necessity of using such a large excess of piperazine drastically reduces atom utilization rates and inflates raw material costs, especially given the high market price of anhydrous piperazine. Furthermore, the recovery of unreacted piperazine from the reaction mixture is technically immature and energy-intensive, often leading to significant waste generation. Additionally, despite the excess reagent, the reaction selectivity remains suboptimal, inevitably generating approximately 20 percent of bis-Boc-piperazine impurities, which complicates downstream purification and lowers the overall yield to a range of merely 61 to 70 percent.
The Novel Approach
In stark contrast to the limitations of direct acylation, the novel methodology disclosed in the patent data reconstructs the piperazine scaffold from simpler, more abundant precursors. By initiating the synthesis with ethylenediamine and chloroacetaldehyde, the process builds the six-membered ring in situ before introducing the protecting group. This fundamental change in retrosynthetic logic eliminates the competitive reaction at the second nitrogen atom that plagues conventional methods. Consequently, the formation of bis-substituted impurities is effectively avoided at the source, rather than requiring complex separation post-reaction. This approach not only simplifies the purification workflow but also ensures a much higher consistency in product quality, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations where impurity profiles are strictly regulated.
Mechanistic Insights into Ethylenediamine-Based Cyclization and Reduction
The core of this technological advancement lies in its three-step mechanistic sequence, which offers precise control over reaction kinetics and thermodynamics. The first stage involves the condensation of ethylenediamine with chloroacetaldehyde under alkaline conditions to form a cyclic intermediate (Compound I). This ring-closing step is critical as it establishes the piperazine backbone with high fidelity. Following this, the secondary amine within the newly formed ring undergoes selective acylation with Boc2O to yield an unsaturated intermediate (Compound II). The final step employs a mild reducing agent, such as sodium borohydride or potassium borohydride, to saturate the double bond, yielding the target N-tert-butyloxycarbonyl piperazine.
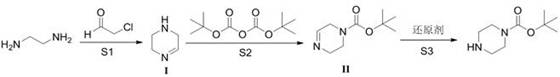
From an impurity control perspective, this mechanism provides inherent selectivity advantages. In the conventional route, both nitrogen atoms in piperazine are chemically equivalent and highly nucleophilic, leading to statistical over-acylation. However, in the new pathway, the nitrogen atoms are differentiated by the stage of the synthesis. The ring closure creates a specific electronic environment where the subsequent acylation can be tightly controlled. Moreover, the reduction step is highly chemoselective, targeting only the olefinic bond without affecting the carbamate protecting group. This results in a final product with gas chromatography purity reaching up to 99.5 percent, significantly reducing the burden on quality control laboratories and ensuring that the material meets the stringent specifications required for API synthesis.
How to Synthesize N-Boc-piperazine Efficiently
Implementing this advanced synthesis route requires careful attention to reaction conditions, particularly temperature control and stoichiometric ratios during the ring-closing phase. The patent outlines a robust protocol where ethylenediamine reacts with chloroacetaldehyde in solvents like methanol or water at moderate temperatures between 25°C and 35°C. Following the isolation of the cyclic intermediate, the acylation and reduction steps can potentially be telescoped into a one-pot reaction, further streamlining the manufacturing process. For detailed operational parameters, safety guidelines, and workup procedures, please refer to the standardized synthesis guide provided below.
- React ethylenediamine with chloroacetaldehyde in the presence of alkali to form the cyclic intermediate (Compound I).
- Perform acylation on Compound I using di-tert-butyl dicarbonate (Boc2O) in methanol to generate the unsaturated intermediate (Compound II).
- Reduce Compound II using a borohydride reducing agent to obtain the final N-tert-butyloxycarbonyl piperazine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this ethylenediamine-based route represents a strategic opportunity to enhance cost efficiency and supply reliability. The primary economic driver is the drastic reduction in raw material consumption. By eliminating the need for a multi-fold excess of expensive anhydrous piperazine, the process significantly lowers the direct material cost per kilogram of the final product. Furthermore, the avoidance of bis-Boc impurities means that less material is lost during recrystallization or chromatographic purification, thereby improving the overall mass balance and reducing waste disposal costs associated with solvent usage and byproduct treatment.
- Cost Reduction in Manufacturing: The novel process fundamentally alters the cost structure by replacing high-cost stoichiometric inefficiencies with a high-yield linear synthesis. Since the reaction does not require recovering large quantities of unreacted starting material, the energy consumption associated with distillation and recycling is substantially diminished. Additionally, the use of common solvents like methanol and water, rather than specialized anhydrous conditions required for some alternative methods, further contributes to operational expenditure savings. These factors combine to create a more economically resilient manufacturing model that can better withstand fluctuations in raw material pricing markets.
- Enhanced Supply Chain Reliability: Relying on ethylenediamine and chloroacetaldehyde as feedstocks diversifies the supply base away from the bottleneck of anhydrous piperazine availability. These starting materials are commodity chemicals with robust global supply chains, reducing the risk of production stoppages due to raw material shortages. The simplified process flow, which potentially allows for one-pot operations in later stages, also reduces the equipment footprint and cycle time. This increased operational flexibility enables manufacturers to respond more rapidly to demand spikes, ensuring consistent delivery schedules for downstream pharmaceutical clients who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, the high atom utilization rate of this method aligns perfectly with green chemistry principles. The reduction in waste generation, specifically the absence of large volumes of piperazine salts and bis-Boc byproducts, simplifies effluent treatment processes. This makes the technology highly scalable from pilot plants to multi-ton commercial reactors without encountering disproportionate increases in environmental compliance costs. The ability to produce high-purity material with minimal purification steps also reduces the solvent intensity of the process, supporting corporate sustainability goals and regulatory adherence in increasingly strict environmental jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: How does the ethylenediamine route improve impurity profiles compared to direct acylation?
A: The novel route constructs the piperazine ring before acylation, inherently preventing the formation of bis-Boc-piperazine impurities that commonly occur when reacting excess piperazine with Boc2O.
Q: What are the raw material advantages of this synthesis method?
A: By utilizing ethylenediamine and chloroacetaldehyde, the process avoids the need for a 3-5 fold molar excess of expensive anhydrous piperazine, significantly improving atom utilization and reducing raw material costs.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent demonstrates a total yield of over 81.5% with product purity reaching 99.5%, utilizing common solvents like methanol and water, which indicates strong feasibility for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Boc-piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical intermediate market. Our technical team has thoroughly analyzed the potential of the ethylenediamine-based pathway described in CN113185478A and is fully prepared to implement this technology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and purity are realized in actual manufacturing. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-Boc-piperazine meets the exacting standards required for drug substance synthesis.
We invite procurement leaders and R&D directors to collaborate with us to leverage this technological advantage for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this optimized route for your projects. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your volume requirements. Let us help you secure a stable, cost-effective, and high-quality supply of this essential heterocyclic building block.
