Revolutionizing Quinolin-2-one Production: A Technical Deep Dive into Patent CN113045489B
The pharmaceutical industry continuously seeks robust synthetic pathways for heterocyclic scaffolds that serve as critical backbones in modern therapeutics. Patent CN113045489B introduces a groundbreaking preparation method for 3-arylquinoline-2(1H) ketone derivatives, addressing long-standing challenges in efficiency and raw material accessibility. These quinolin-2-one structures are not merely academic curiosities; they are privileged motifs found in potent bioactive molecules ranging from MAP Kinase inhibitors to HBV inhibitors, as illustrated by the diverse pharmacological profiles in the referenced data. This novel approach leverages a palladium-catalyzed aminocarbonylation strategy that fundamentally rethinks the construction of the quinolone core, moving away from harsh classical conditions toward a more atom-economical and operationally simple protocol. By integrating benzisoxazole as a dual-purpose reagent, the invention streamlines the synthetic sequence, offering a compelling value proposition for R&D teams focused on rapid analog generation and process optimization.
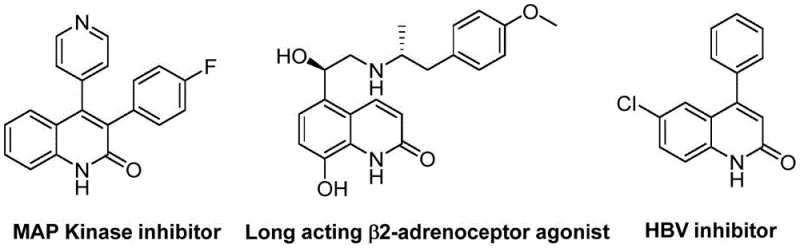
The limitations of conventional methods for synthesizing quinolin-2(1H)one derivatives have historically constrained both research velocity and manufacturing economics. Traditional approaches such as the Vilsmeier-Haack, Knorr, or Friedlander reactions often necessitate the use of corrosive reagents, extreme temperatures, or multi-step sequences that generate significant chemical waste. Furthermore, transition metal-catalyzed improvements in the past have frequently relied on high-pressure carbon monoxide gas, introducing substantial safety hazards and engineering complexities that hinder seamless technology transfer from the laboratory to the pilot plant. These legacy methods often suffer from narrow substrate scope, failing to tolerate sensitive functional groups that are increasingly common in modern drug design, thereby forcing chemists to employ tedious protecting group strategies that erode overall yield and increase production timelines.
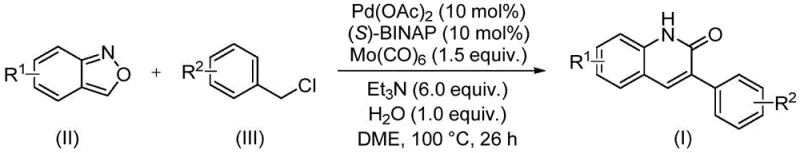
In stark contrast, the novel approach detailed in this patent utilizes a sophisticated palladium catalytic system that operates under remarkably mild and manageable conditions. The reaction employs benzisoxazole and benzyl chloride compounds as starting materials, which are not only inexpensive and widely available in the global chemical market but also exhibit excellent stability during storage and handling. The use of Mo(CO)6 as a solid carbon monoxide surrogate effectively bypasses the need for specialized high-pressure equipment, allowing the reaction to proceed safely at atmospheric pressure in standard glassware or reactors. This methodological shift drastically simplifies the operational workflow, reduces the environmental footprint associated with hazardous gas handling, and opens up new possibilities for the late-stage functionalization of complex molecules without compromising the integrity of sensitive moieties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolin-2-one scaffold has been plagued by inefficiencies inherent to classical cyclization strategies. Methods relying on strong acids or Lewis acids often require stoichiometric amounts of reagents, leading to difficult workup procedures and substantial salt waste that complicates downstream purification. Additionally, the reliance on gaseous carbon monoxide in earlier carbonylation attempts imposes severe restrictions on reactor design, requiring autoclaves and rigorous safety protocols that increase capital expenditure and operational overhead. The functional group tolerance in these older methodologies is frequently poor, meaning that substrates containing halides, nitriles, or ether linkages might decompose or fail to react, limiting the chemical space accessible to medicinal chemists. Consequently, the development of new drug candidates based on this scaffold has often been slowed by the inability to rapidly synthesize diverse libraries with high purity and consistency.
The Novel Approach
The methodology disclosed in CN113045489B represents a paradigm shift by integrating a dual-function nitrogen and formyl source directly into the catalytic cycle. By employing benzisoxazole, the reaction eliminates the need for separate formylation steps, effectively condensing the synthetic route and improving the overall atom economy. The catalytic system, comprising palladium acetate and the chiral ligand (S)-BINAP, ensures high selectivity and turnover, while the presence of triethylamine and water facilitates the necessary proton transfers and hydrolysis steps without requiring anhydrous conditions. This robustness allows for a wider range of substituents on both the benzisoxazole and benzyl chloride components, enabling the synthesis of derivatives with electron-donating or electron-withdrawing groups in high yields. The simplicity of the post-processing, involving basic filtration and chromatography, further underscores the practical utility of this method for both small-scale discovery and larger-scale production environments.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this technological advancement lies in the intricate interplay between the palladium catalyst and the unique reactivity of the benzisoxazole ring. The reaction initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species, generating a benzyl-palladium(II) intermediate that is primed for subsequent insertion steps. The presence of Mo(CO)6 serves as a controlled release source of carbon monoxide, which coordinates to the palladium center to form an acyl-palladium complex. This step is critical as it avoids the safety risks associated with handling toxic CO gas while maintaining the high concentration of CO required for efficient carbonylation. The benzisoxazole then participates in a nucleophilic attack or insertion sequence, where the N-O bond cleavage provides the necessary nitrogen atom for the ring closure and the oxygen atom is expelled, driving the thermodynamic equilibrium toward the formation of the stable quinolin-2-one product.
Impurity control in this system is inherently managed by the high specificity of the palladium catalytic cycle and the stability of the intermediates formed. Unlike radical-based processes that can lead to polymerization or non-selective coupling, this organometallic pathway follows a well-defined coordination geometry that minimizes side reactions. The use of (S)-BINAP as a ligand not only enhances the electronic properties of the palladium center but also provides steric bulk that discourages the formation of undesired homocoupling byproducts from the benzyl chloride. Furthermore, the reaction conditions are optimized to ensure complete consumption of the starting materials, as evidenced by the high conversion rates observed across various substrates. The resulting crude mixtures are typically clean, requiring only standard silica gel chromatography to achieve pharmaceutical-grade purity, which significantly reduces the burden on quality control laboratories and accelerates the release of materials for biological testing.
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
Implementing this synthesis requires precise adherence to the molar ratios and thermal conditions specified in the patent to maximize yield and reproducibility. The protocol involves charging a reaction vessel with palladium acetate, (S)-BINAP, Mo(CO)6, triethylamine, water, benzisoxazole, and benzyl chloride in a solvent system such as ethylene glycol dimethyl ether (DME). The mixture is then heated to 100°C and maintained for approximately 26 hours, a duration that balances reaction completeness with operational efficiency. Detailed standardized synthesis steps see the guide below.
- Combine palladium acetate, (S)-BINAP, Mo(CO)6, triethylamine, water, benzisoxazole, and benzyl chloride in DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Perform post-processing via filtration and silica gel mixing, followed by column chromatography purification to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial strategic benefits by leveraging commodity chemicals that are readily available from multiple global suppliers. The primary starting materials, benzisoxazoles and benzyl chlorides, are produced on a massive scale for various industrial applications, ensuring a stable supply chain that is resistant to market fluctuations or single-source bottlenecks. The elimination of exotic or custom-synthesized reagents reduces the complexity of vendor management and lowers the risk of production delays caused by raw material shortages. Furthermore, the simplified reaction setup reduces the demand for specialized infrastructure, allowing manufacturing partners to utilize existing general-purpose reactors rather than investing in dedicated high-pressure carbonylation suites. This flexibility translates directly into lower capital barriers for entry and faster time-to-market for new products utilizing this scaffold.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the consolidation of synthetic steps and the use of cost-effective reagents. By utilizing benzisoxazole as a dual nitrogen and formyl source, the need for separate formylation reagents and additional reaction vessels is completely eliminated, which significantly reduces solvent consumption and energy usage. The replacement of gaseous carbon monoxide with solid Mo(CO)6 removes the need for expensive gas containment systems and safety monitoring equipment, leading to drastic simplification in facility requirements. Additionally, the high efficiency of the catalyst system means that lower loading levels can potentially be achieved upon further optimization, reducing the consumption of precious palladium metals. These factors combine to create a manufacturing profile that is inherently leaner and more cost-competitive than traditional multi-step alternatives.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the widespread availability and chemical stability of the key reactants involved in this protocol. Benzyl chlorides and benzisoxazole derivatives are staple intermediates in the fine chemical industry, meaning they can be sourced from a diverse network of qualified vendors across different geographic regions. This diversity mitigates the risk of supply disruptions caused by regional logistics issues or regulatory changes affecting specific manufacturers. Moreover, the robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling production planners to respond quickly to changes in demand without the long lead times associated with complex custom synthesis. The ability to store intermediates and reagents under standard conditions further simplifies inventory management and reduces warehousing costs.
- Scalability and Environmental Compliance: Scaling this process to commercial volumes is facilitated by the absence of high-pressure gases and the use of moderate temperatures that are easily managed in large-scale reactors. The reaction generates minimal hazardous waste compared to classical methods that rely on stoichiometric amounts of corrosive acids or oxidants, aligning well with modern green chemistry principles and environmental regulations. The workup procedure, which involves simple filtration and chromatography, is easily adaptable to continuous processing or large-batch isolation techniques, ensuring that purity standards can be maintained even at the multi-ton scale. This environmental and operational compatibility makes the technology highly attractive for contract manufacturing organizations seeking to expand their portfolio of heterocyclic intermediates without incurring significant compliance burdens.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on feasibility and advantages. Understanding these details is crucial for technical teams evaluating the integration of this route into their existing production pipelines.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl source, significantly simplifying the reactant list and reducing raw material costs compared to traditional multi-component couplings.
Q: How does this method improve safety over conventional carbonylation?
A: By utilizing Mo(CO)6 as a solid carbon monoxide surrogate instead of pressurized CO gas, the process eliminates high-pressure safety risks and simplifies reactor requirements for commercial scale-up.
Q: Is this protocol suitable for large-scale pharmaceutical manufacturing?
A: Yes, the use of commercially available starting materials like benzyl chlorides and the robustness of the palladium catalyst system under moderate temperatures (100°C) supports efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinoline-2(1H) ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for high-value pharmaceutical intermediates like 3-arylquinoline-2(1H) ketone derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of validating the complex impurity profiles associated with palladium-catalyzed reactions. We are committed to delivering materials that meet the highest international standards, supporting your drug development timelines with consistent quality and unwavering reliability.
We invite you to collaborate with our technical team to explore how this innovative synthesis can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce overall project costs.
