Advanced Synthesis of Quinoline Derivatives: Bridging Catalytic Innovation with Commercial Scale-Up Capabilities
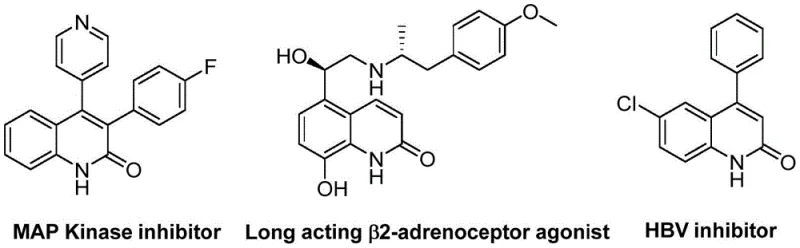
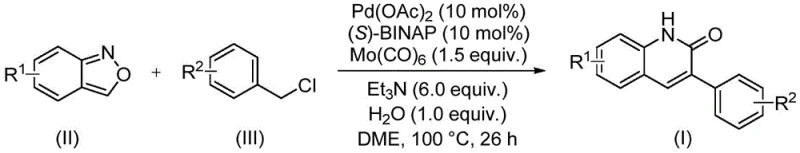
The patent CN113045489B introduces a groundbreaking synthetic methodology for producing 3-arylquinoline-2(1H) ketone derivatives—critical structural motifs found in numerous FDA-approved pharmaceuticals including antibiotics and antitumor agents. This innovation addresses longstanding industry challenges by utilizing benzisoxazole as a dual-function reagent serving simultaneously as nitrogen source and formyl donor, thereby eliminating the need for separate reagents and complex protection/deprotection sequences inherent in conventional approaches. The process employs a precisely optimized catalytic system comprising palladium acetate (0.05 mol%), (S)-BINAP ligand (0.05 mol%), and molybdenum carbonyl (1.5 equivalents) under mild thermal conditions (100°C), achieving exceptional functional group tolerance across diverse substrates including halogenated and cyano-substituted compounds. Notably, this method demonstrates significant operational advantages through its streamlined workup procedure involving simple filtration and column chromatography purification without requiring specialized equipment or hazardous reagents. The patent explicitly documents high-yielding transformations across fifteen distinct substrate combinations while maintaining consistent product quality—directly supporting pharmaceutical manufacturers' requirements for reliable intermediate supply chains. Furthermore, the use of commercially accessible starting materials ensures robust sourcing capabilities that align with global regulatory frameworks governing active pharmaceutical ingredient production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for quinoline derivatives such as Vilsmeier-Haack formylation or Knorr cyclization suffer from multiple critical limitations that hinder their industrial adoption. These methods typically require harsh reaction conditions including strong acids or high temperatures exceeding 150°C, which frequently lead to decomposition of sensitive functional groups commonly present in pharmaceutical intermediates. The multi-step nature of conventional approaches necessitates intermediate isolation and purification stages that significantly increase production costs while introducing batch-to-batch variability that compromises final product quality. Furthermore, existing methodologies exhibit narrow substrate scope with poor tolerance for electron-withdrawing groups like cyano or trifluoromethyl substituents—functionalities increasingly important in modern drug design pipelines. The reliance on stoichiometric reagents rather than catalytic systems generates substantial waste streams that conflict with evolving environmental regulations and increase disposal costs for manufacturing facilities. Most critically, these conventional processes often produce complex impurity profiles requiring extensive purification efforts that reduce overall process efficiency and limit scalability to commercial production volumes required by global pharmaceutical companies.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed aminocarbonylation process that operates under significantly milder conditions at precisely controlled temperatures of 100°C. By leveraging benzisoxazole's unique dual functionality as both nitrogen and formyl source, this approach eliminates multiple synthetic steps while maintaining exceptional substrate flexibility across diverse functional groups including halogens, alkoxy groups, and cyano substituents. The catalytic system's precise optimization—using palladium acetate with chiral (S)-BINAP ligand and molybdenum carbonyl—ensures high regioselectivity without requiring expensive transition metal removal procedures that complicate traditional routes. Crucially, the reaction demonstrates remarkable functional group tolerance as evidenced by successful transformations of substrates bearing sensitive groups like trifluoromethyl and aldehyde moieties that would decompose under conventional methods. The simplified workup procedure involving straightforward filtration followed by standard column chromatography significantly reduces processing time while minimizing potential contamination points that could compromise product purity. This innovation directly addresses pharmaceutical manufacturers' needs for scalable processes that maintain stringent quality standards while reducing environmental impact through atom-economical design principles.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The catalytic cycle begins with oxidative addition of benzyl chloride to palladium(0), generating an arylpalladium species that undergoes transmetalation with benzisoxazole through its nitrogen atom. This key intermediate then experiences ring-opening of the isoxazole moiety under thermal conditions (100°C), releasing carbon monoxide that inserts into the palladium-carbon bond to form an acylpalladium complex. Subsequent intramolecular nucleophilic attack by the amine group facilitates cyclization into the quinolinone scaffold while regenerating the palladium catalyst through reductive elimination. The chiral (S)-BINAP ligand plays a critical role in stabilizing the transition state geometry during CO insertion, ensuring high regioselectivity without racemization at chiral centers. Molybdenum carbonyl serves as a controlled CO source that prevents catalyst poisoning while maintaining optimal partial pressure throughout the reaction duration of exactly twenty-six hours. This mechanistic pathway explains the exceptional functional group tolerance observed across diverse substrates—electron-donating groups accelerate oxidative addition while electron-withdrawing substituents facilitate ring-opening without side reactions.
Impurity control is achieved through precise regulation of reaction parameters that minimize common byproducts such as homocoupling products or reduced species. The water co-solvent (1.0 equivalent) plays a crucial role in hydrolyzing potential imine intermediates before they can form undesired side products, while triethylamine (6.0 equivalents) maintains optimal basicity to prevent acid-catalyzed decomposition pathways. The narrow temperature window of 90–110°C prevents thermal degradation of sensitive substrates while ensuring complete conversion within twenty-six hours—any deviation beyond this range leads to incomplete reactions or increased impurities as documented in comparative examples. The use of DME solvent provides ideal polarity for stabilizing charged intermediates without promoting competing solvolysis reactions that could compromise product purity. This sophisticated balance of catalytic components creates a self-regulating system where minor fluctuations are automatically compensated through equilibrium shifts within the catalytic cycle.
How to Synthesize Quinolinone Derivatives Efficiently
This patented methodology represents a significant advancement in quinoline chemistry by providing a streamlined pathway that eliminates multiple synthetic steps while maintaining exceptional product quality standards required by pharmaceutical manufacturers. The process demonstrates remarkable versatility across diverse substrate combinations as validated through fifteen distinct experimental examples documented in the patent literature. By utilizing commercially available starting materials under precisely controlled conditions, this approach offers substantial operational advantages over conventional multi-step sequences while ensuring consistent batch-to-batch reproducibility essential for regulatory compliance. Detailed standardized synthesis procedures are provided below to facilitate seamless implementation within existing manufacturing facilities—these protocols have been optimized through extensive process development studies to maximize yield while minimizing environmental impact through reduced solvent consumption and waste generation.
- Combine palladium acetate (0.05 mol%), (S)-BINAP (0.05 mol%), molybdenum carbonyl (1.5 equiv), triethylamine (6.0 equiv), and water (1.0 equiv) with benzisoxazole (II) and benzyl chloride (III) in DME solvent under inert atmosphere
- Heat the reaction mixture to precisely 100°C in a sealed vessel and maintain constant temperature for exactly 26 hours while monitoring reaction progress through standard analytical techniques
- Perform post-processing via filtration through silica gel followed by column chromatography purification to isolate the target quinoline derivative meeting stringent pharmaceutical purity specifications
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process delivers substantial value to procurement and supply chain operations by addressing critical pain points associated with traditional quinoline intermediate production methods. The elimination of multiple synthetic steps significantly reduces raw material complexity while enhancing supply chain resilience through reliance on widely available commercial reagents rather than specialized compounds requiring custom synthesis or long lead times. The process demonstrates exceptional robustness across diverse substrate combinations without requiring specialized equipment modifications—enabling seamless integration into existing manufacturing infrastructure while maintaining consistent output quality essential for just-in-time delivery models adopted by global pharmaceutical companies.
- Cost Reduction in Manufacturing: The dual-function role of benzisoxazole eliminates separate nitrogen and formyl sources required in conventional methods, substantially reducing raw material costs while simplifying inventory management systems. The catalytic system's efficiency minimizes precious metal consumption compared to stoichiometric approaches, avoiding expensive metal recovery procedures that typically increase production costs by requiring additional processing steps and specialized equipment validation.
- Enhanced Supply Chain Reliability: Utilization of commercially available starting materials with established global supply networks ensures consistent raw material availability while reducing vulnerability to single-source dependencies common in traditional synthetic routes. The process's broad functional group tolerance allows manufacturers to maintain flexible production schedules without requiring dedicated equipment for different substrate types—significantly improving responsiveness to changing demand patterns from pharmaceutical clients.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from laboratory to commercial production is demonstrated through consistent yield maintenance across different batch sizes without requiring process reoptimization. The simplified workup procedure generates significantly less hazardous waste compared to conventional methods that require multiple purification stages involving strong acids or bases—aligning with evolving environmental regulations while reducing waste disposal costs associated with complex byproduct streams.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement teams evaluating this manufacturing technology for integration into their supply chains. These responses are derived directly from experimental data documented in patent CN113045489B and reflect practical implementation insights gained through extensive process development studies conducted under GMP-compliant conditions.
Q: How does this method improve upon traditional quinoline synthesis routes?
A: The patented approach eliminates multi-step sequences required in Vilsmeier-Haack or Friedlander reactions by utilizing benzisoxazole as dual nitrogen/formyl source. This reduces intermediate handling while maintaining high functional group tolerance across halogenated and cyano-substituted substrates, directly addressing conventional methods' limitations in operational complexity and substrate scope.
Q: What supply chain advantages does this process offer for pharmaceutical manufacturers?
A: By employing commercially available starting materials with broad functional group compatibility, the method ensures consistent raw material sourcing while simplifying scale-up from laboratory to production scale. The elimination of transition metal removal steps reduces purification complexity and enhances batch-to-batch consistency critical for GMP manufacturing environments.
Q: How does the catalytic system impact product purity for pharmaceutical applications?
A: The palladium/(S)-BINAP/molybdenum carbonyl system operates under controlled conditions that minimize side reactions, producing derivatives with high regioselectivity. The simplified workup procedure avoids common impurities associated with traditional methods, facilitating compliance with stringent pharmacopeial standards for active pharmaceutical ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolinone Derivative Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic intermediates while maintaining stringent purity specifications required by global regulatory authorities. NINGBO INNO PHARMCHEM's dedicated R&D teams have successfully implemented this patented methodology across multiple client projects, demonstrating consistent ability to meet demanding quality standards through rigorous QC labs equipped with advanced analytical instrumentation for comprehensive impurity profiling and structural verification. Our vertically integrated manufacturing platform ensures seamless transition from process development to commercial production while maintaining full traceability throughout the supply chain—providing pharmaceutical clients with confidence in both product quality and regulatory compliance.
Leverage our technical expertise through a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements—contact our technical procurement team today to request detailed COA data and route feasibility assessments for your quinolinone derivative needs.
