Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale Manufacturing
Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access bioactive heterocyclic scaffolds, particularly those containing fluorine motifs which are critical for modern drug design. Patent CN111675662B, published in late 2021, introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds that addresses many of the historical bottlenecks in this chemical space. Quinazolinones are privileged structures found in numerous natural products and therapeutic agents, exhibiting a broad spectrum of biological activities including anti-cancer, anticonvulsant, and anti-inflammatory properties. The strategic introduction of a trifluoromethyl group into these heterocycles significantly enhances their electronegativity, lipophilicity, and metabolic stability, making them highly desirable candidates for lead optimization in medicinal chemistry programs. This patent discloses a novel synthetic route that leverages inexpensive iron catalysis to construct these complex molecules from readily available precursors, marking a significant advancement over traditional methodologies.
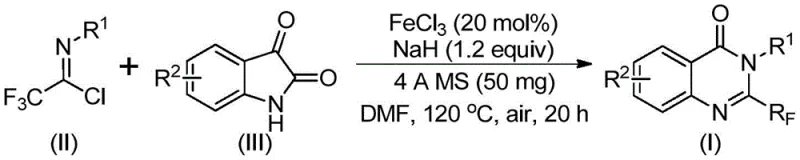
The limitations of conventional methods for synthesizing trifluoromethyl-substituted quinazolinones have long hindered their widespread adoption in cost-sensitive manufacturing environments. Historically, the literature reports that the synthesis of these valuable intermediates relies heavily on the cyclization of synthons bearing trifluoromethyl groups, such as trifluoroacetic anhydride or ethyl trifluoroacetate, with substrates like anthranilamide, anthranilic acid, or isatoic anhydride. While chemically feasible, these established protocols suffer from severe drawbacks that impact both economic viability and operational safety. The reaction conditions are often excessively harsh, requiring stringent anhydrous environments or extreme temperatures that complicate process control. Furthermore, the starting materials, particularly specialized trifluoromethylating agents, can be prohibitively expensive and difficult to source in bulk quantities. Perhaps most critically, these legacy methods frequently exhibit narrow substrate scope and low yields, generating significant chemical waste and necessitating complex purification steps that erode profit margins.
In stark contrast, the novel approach detailed in the patent utilizes a remarkably simple yet effective strategy involving the reaction of trifluoroethylimidoyl chloride with isatin derivatives. This new methodology employs ferric chloride (FeCl3) as a cheap and abundant Lewis acid catalyst, paired with sodium hydride as a base promoter, to drive the transformation. The reaction proceeds through a tandem sequence where a carbon-nitrogen bond is initially formed between the imidoyl chloride and the isatin, followed by an iron-catalyzed decarbonylation and cyclization event. This cascade reaction not only streamlines the synthetic steps but also operates under relatively mild conditions, typically initiating at 40°C and concluding at 120°C in common solvents like DMF. The use of 4A molecular sieves further enhances efficiency by scavenging moisture, ensuring high conversion rates. This robust protocol allows for the synthesis of a wide array of quinazolinone derivatives with excellent functional group tolerance, accommodating various substituents on the aromatic rings without compromising yield or purity.
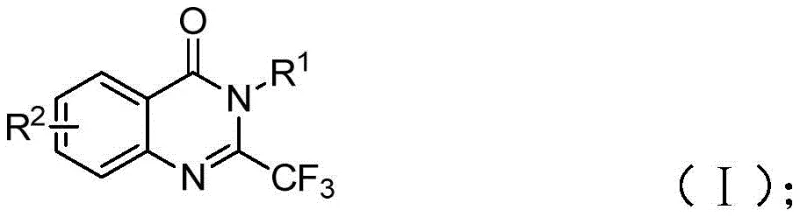
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Decarbonylation
The mechanistic pathway proposed for this transformation offers fascinating insights into how base metals can mimic the reactivity of precious metals in C-H activation and cyclization processes. The reaction initiates with the deprotonation of the isatin nitrogen by sodium hydride, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a key trifluoroacetamidine intermediate, establishing the crucial carbon-nitrogen linkage required for the fused ring system. Subsequently, the ferric chloride catalyst plays a pivotal role in facilitating the decarbonylation of the isatin moiety. Unlike traditional thermal decarbonylation which requires extreme energy input, the iron center likely coordinates with the carbonyl oxygen, lowering the activation energy barrier for CO extrusion. This generates a reactive intermediate that undergoes intramolecular cyclization to close the quinazolinone ring. The presence of the trifluoromethyl group stabilizes the final product through strong inductive effects, driving the equilibrium towards the desired heterocycle. This elegant mechanism avoids the use of toxic carbon monoxide gas or unstable diazo compounds often seen in alternative routes, enhancing the overall safety profile of the process.
From an impurity control perspective, this catalytic system demonstrates remarkable selectivity, which is paramount for pharmaceutical intermediate manufacturing. The specific combination of FeCl3 and NaH in DMF appears to suppress common side reactions such as hydrolysis of the imidoyl chloride or polymerization of the isatin. The patent data indicates that even with diverse substituents like halogens (fluorine, chlorine, bromine) and electron-withdrawing nitro groups, the reaction maintains high fidelity, producing the target quinazolinones with minimal byproduct formation. The use of 4A molecular sieves is a critical engineering control that prevents water-mediated degradation of the sensitive imidoyl chloride reactant, thereby maximizing atom economy. Post-reaction processing is equally straightforward; the crude mixture can be filtered to remove the molecular sieves and inorganic salts, followed by standard silica gel column chromatography. This simplicity in purification suggests that the impurity profile is clean enough for scalable production, reducing the burden on downstream processing teams who often struggle with removing trace metal contaminants from precious metal catalyzed reactions.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The synthesis of these high-value intermediates has been optimized to balance reaction kinetics with operational simplicity, making it accessible for both laboratory R&D and pilot plant operations. The process begins by charging a reactor with the specified molar ratios of ferric chloride, sodium hydride, and 4A molecular sieves in an anhydrous organic solvent, preferably DMF. To this suspension, the trifluoroethylimidoyl chloride and the chosen isatin derivative are added. The reaction is carefully managed in two temperature stages: an initial period at 40°C for roughly 10 hours allows for the controlled formation of the amidine intermediate without excessive exotherm, followed by heating to 120°C for approximately 20 hours to drive the cyclization and decarbonylation to completion. Detailed standardized synthetic steps for specific derivatives are provided in the technical guide below.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in anhydrous DMF solvent.
- Stir the reaction mixture at 40°C for approximately 10 hours to initiate the base-promoted coupling, then heat to 120°C for 20 hours under air to complete the cyclization.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from precious metal catalysis to this iron-based system represents a substantial opportunity for cost optimization and risk mitigation. The reliance on ferric chloride, a commodity chemical available in massive quantities globally, eliminates the volatility associated with the pricing and supply of palladium, platinum, or rhodium catalysts. This transition not only lowers the direct cost of goods sold (COGS) but also simplifies the supply chain by reducing dependency on specialized catalyst vendors who often have long lead times. Furthermore, the starting materials—isatins and aromatic amines used to make the imidoyl chlorides—are widely produced bulk chemicals with stable market availability. This ensures a continuous supply of raw materials, shielding manufacturing schedules from the disruptions that frequently plague the sourcing of exotic fluorinated building blocks.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive reagents with commodity chemicals. By utilizing ferric chloride instead of precious metals, manufacturers avoid the high capital expenditure associated with catalyst recovery systems and the ongoing cost of catalyst replenishment. Additionally, the reaction operates under aerobic conditions (in air), removing the need for costly inert gas purging infrastructure and the associated utility costs of maintaining nitrogen or argon atmospheres. The high yields reported, often exceeding 90% for optimized substrates, mean that less raw material is wasted, directly improving the overall mass balance and reducing the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route translates directly into supply chain resilience. Because the reaction tolerates a wide range of functional groups and uses stable, commercially available solvents like DMF, there is flexibility in sourcing. If a specific isatin derivative is temporarily unavailable, the chemistry is flexible enough to accommodate alternative substitution patterns without requiring a complete process redevelopment. The ability to run the reaction in air rather than under strict inert conditions also reduces the technical complexity required at contract manufacturing organizations (CMOs), allowing a broader base of suppliers to bid on production contracts, thereby increasing competition and driving down prices.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage is facilitated by the absence of hazardous reagents like diazomethane or high-pressure carbon monoxide. The use of iron, a non-toxic metal, significantly eases the regulatory burden regarding heavy metal residuals in the final product, a critical specification for pharmaceutical regulators. Waste streams are easier to treat since they do not contain persistent organic pollutants associated with complex ligand systems found in noble metal catalysis. This environmental compatibility aligns with modern green chemistry principles, potentially qualifying the manufacturing process for sustainability incentives and reducing the costs associated with hazardous waste disposal and environmental permitting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is crucial for R&D teams evaluating the technology for integration into existing pipelines and for procurement teams negotiating supply agreements.
Q: What are the primary advantages of using FeCl3 over precious metal catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) eliminates the need for expensive palladium or rhodium catalysts, drastically reducing raw material costs. Furthermore, iron is non-toxic and environmentally benign, simplifying waste disposal and regulatory compliance compared to heavy metal residues.
Q: Can this synthetic method tolerate diverse functional groups on the aromatic ring?
A: Yes, the patent data demonstrates excellent functional group tolerance. The method successfully accommodates electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens (F, Cl, Br) and nitro groups, yielding products with high purity and consistent yields ranging from 74% to 93%.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The protocol utilizes commercially available starting materials like isatins and imidoyl chlorides, operates under aerobic conditions (air), and uses standard solvents like DMF. These factors, combined with the absence of sensitive organometallic reagents, make the process highly robust and easily scalable from gram-level laboratory synthesis to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
As the global demand for fluorinated heterocycles continues to surge, partnering with a manufacturer that possesses deep technical expertise in these specific transformations is essential. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging advanced catalytic methodologies like the one described in CN111675662B to deliver superior quality intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our 2-trifluoromethyl quinazolinones meet the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to explore how this innovative synthesis can optimize your specific project requirements. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this iron-catalyzed route can improve your bottom line. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments for your proprietary targets. Let us be your partner in turning complex chemical challenges into commercial successes.
