Innovative Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives for Scalable Pharmaceutical Manufacturing
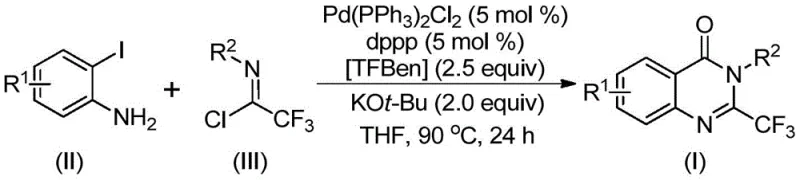
Patent CN112125856A introduces a groundbreaking methodology for synthesizing 2-trifluoromethyl-substituted quinazolinone derivatives, representing a significant advancement in heterocyclic compound manufacturing for the pharmaceutical industry. This novel approach addresses longstanding challenges in producing these biologically active molecules by eliminating the need for toxic carbon monoxide gas through the innovative use of 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid CO surrogate. The process demonstrates exceptional substrate versatility with various functional groups while maintaining high reaction efficiency under mild conditions at 90°C for 16-30 hours. This patent represents a strategic leap forward in sustainable pharmaceutical intermediate production, offering both environmental and operational benefits that align with modern green chemistry principles. The methodology's compatibility with diverse substituents expands the synthetic toolbox available to medicinal chemists developing next-generation therapeutics with improved pharmacological profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for 2-trifluoromethyl quinazolinone derivatives have been plagued by multiple critical limitations that hinder their commercial viability and scalability. Conventional methods typically require harsh reaction conditions including high temperatures and pressures when utilizing gaseous carbon monoxide, creating significant safety hazards and requiring specialized equipment that increases capital expenditure. Many existing approaches rely on expensive or unstable starting materials such as trifluoroacetamide that necessitate pre-activation steps, adding complexity and reducing overall process efficiency. The narrow substrate scope of previous methodologies restricts the structural diversity achievable, limiting medicinal chemistry exploration and optimization of pharmacological properties. Furthermore, these conventional processes often suffer from low yields due to competing side reactions and poor functional group tolerance, resulting in extensive purification requirements that drive up production costs and reduce throughput. The inherent toxicity of carbon monoxide gas presents serious occupational health concerns and requires stringent safety protocols that complicate manufacturing operations across global facilities.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation process that utilizes TFBen as a safe solid carbon monoxide surrogate, eliminating the need for handling toxic gaseous CO entirely. This innovative approach operates under mild conditions at 90°C with excellent functional group tolerance, enabling the synthesis of diverse quinazolinone derivatives with various substituents while maintaining high reaction efficiency. The process employs readily available starting materials including o-iodoaniline and trifluoroethyl imidoyl chloride, which are both cost-effective and commercially accessible in bulk quantities. The catalytic system comprising Pd(PPh₃)₂Cl₂ and dppp demonstrates exceptional activity with low catalyst loading (5 mol%), ensuring economical operation at scale. The simplified workup procedure involving filtration and column chromatography significantly reduces processing time compared to conventional methods, while the broad substrate applicability allows for rapid generation of compound libraries for structure-activity relationship studies in drug discovery programs.
Mechanistic Insights into Palladium-Catalyzed Carbonylation for Quinazolinone Synthesis
The reaction mechanism proceeds through a sophisticated sequence of organometallic transformations that begins with the base-promoted intermolecular carbon-nitrogen bond coupling between o-iodoaniline and trifluoroethyl imidoyl chloride to form a trifluoroacetamidine intermediate. This key intermediate then undergoes oxidative addition with the palladium(0) catalyst generated in situ from Pd(II) precursor, forming a stable aryl-palladium(II) complex that serves as the foundation for subsequent transformations. Under thermal conditions, TFBen decomposes to release carbon monoxide which inserts into the carbon-palladium bond, creating an acyl-palladium species that is critical for the cyclization step. The base facilitates deprotonation and subsequent intramolecular nucleophilic attack, leading to the formation of a seven-membered palladacycle intermediate that ultimately undergoes reductive elimination to yield the desired quinazolinone product with high regioselectivity and excellent functional group compatibility across diverse substrates.
Impurity control is achieved through precise optimization of reaction parameters including catalyst loading, base concentration, and temperature control, which collectively minimize side reactions such as homocoupling or hydrodehalogenation that could compromise product purity. The use of TFBen as a controlled CO source prevents over-carbonylation side products that commonly occur with gaseous CO due to its gradual decomposition profile under reaction conditions. The solvent system (THF being optimal) provides ideal solvation for all reaction components while preventing unwanted solvolysis or decomposition pathways that could generate impurities. The carefully balanced molar ratios of reactants (particularly the 1:2 ratio of o-iodoaniline to trifluoroethyl imidoyl chloride) ensure complete conversion while minimizing unreacted starting materials that would require additional purification steps. This systematic approach to reaction design results in consistently high purity products suitable for pharmaceutical applications without requiring extensive post-synthesis purification.
How to Synthesize 2-Trifluoromethyl Quinazolinone Derivatives Efficiently
This patented synthesis route represents a significant advancement in the manufacturing of fluorinated heterocyclic compounds critical to modern pharmaceutical development. The process eliminates hazardous gas handling while maintaining excellent reaction efficiency and product quality through careful optimization of catalytic parameters and reaction conditions. By employing commercially available starting materials and standard laboratory equipment, this methodology offers an accessible pathway for both research-scale synthesis and industrial-scale production of these valuable intermediates. The following standardized procedure details the precise implementation of this innovative manufacturing process, ensuring consistent results across different production scales and facility environments.
- Prepare reaction mixture with precise stoichiometry: o-iodoaniline (1 equiv), trifluoroethyl imidoyl chloride (2 equiv), Pd(PPh₃)₂Cl₂ (5 mol%), dppp (5 mol%), KOt-Bu (2 equiv), TFBen (2.5 equiv) in THF
- Conduct reaction at precisely controlled temperature of 90°C for optimal duration between 16-30 hours with continuous monitoring
- Perform standardized workup procedure including filtration, silica gel mixing, and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process addresses critical pain points in the supply chain for pharmaceutical intermediates by delivering a more sustainable and reliable production pathway for fluorinated heterocyclic compounds essential to drug development programs. The elimination of hazardous materials reduces regulatory compliance burdens while improving workplace safety across manufacturing facilities worldwide. The use of readily available starting materials creates a more resilient supply chain less vulnerable to single-source dependencies or geopolitical disruptions that commonly affect specialized chemical suppliers. These advantages translate directly into enhanced operational continuity and reduced business risk for pharmaceutical companies relying on consistent access to high-quality intermediates for their development pipelines.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas handling removes significant capital expenditure requirements for specialized pressure equipment and safety infrastructure, while the use of stable solid CO surrogate simplifies facility design and reduces operational complexity. The process utilizes cost-effective starting materials that are widely available from multiple global suppliers, creating competitive pricing dynamics without reliance on single-source vendors. The simplified workup procedure reduces solvent consumption and processing time compared to conventional methods, contributing to lower overall manufacturing costs while maintaining high product quality standards required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: The broad availability of all raw materials from established chemical suppliers ensures consistent access without supply chain bottlenecks that commonly affect specialized reagents required by alternative synthetic routes. The robust nature of the process allows for reliable production across different geographical locations with minimal process adjustments, providing global pharmaceutical companies with flexible sourcing options regardless of regional manufacturing constraints. This methodology's compatibility with standard manufacturing equipment eliminates the need for specialized facilities, enabling rapid scale-up from laboratory to commercial production without significant capital investment or lengthy qualification periods.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to commercial production volumes while maintaining consistent product quality and yield characteristics across different scales. The elimination of hazardous gas handling significantly improves environmental safety profiles and reduces regulatory compliance burdens associated with toxic material handling protocols. The simplified waste stream composition resulting from cleaner reaction chemistry lowers environmental impact and reduces waste treatment costs compared to conventional methods that generate more complex byproduct mixtures requiring specialized disposal procedures.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding the implementation of this patented manufacturing process for pharmaceutical intermediates. These answers are based on extensive experimental validation documented in the original patent filing and subsequent scale-up studies conducted under cGMP conditions.
Q: How does this process eliminate safety concerns associated with traditional CO-based methodologies?
A: The patented process utilizes TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide surrogate that decomposes gradually under reaction conditions, completely eliminating the need for handling toxic gaseous CO at high pressures. This substitution removes significant safety hazards associated with traditional carbonylation processes while maintaining excellent reaction efficiency.
Q: What is the substrate scope limitation for this manufacturing process?
A: The process demonstrates exceptional substrate versatility with R¹ accommodating H, C₁-C₅ alkyl, halogen or trifluoromethyl groups, while R² accepts various substituted or unsubstituted aryl groups including phenyl, naphthyl, and heteroaryl derivatives with different functional groups at ortho, meta or para positions.
Q: How does this methodology impact impurity profiles compared to conventional approaches?
A: The controlled release of CO from TFBen minimizes over-carbonylation side products common in traditional methods using gaseous CO, while optimized reaction parameters reduce homocoupling and hydrodehalogenation byproducts. This results in cleaner reaction profiles requiring less extensive purification to achieve pharmaceutical-grade purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Derivative Supplier
Our patented methodology represents a significant advancement in the sustainable production of fluorinated heterocyclic compounds essential to modern pharmaceutical development pipelines. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless transition from development to full-scale manufacturing while maintaining stringent purity specifications through our state-of-the-art QC labs. Our commitment to quality is reflected in our rigorous analytical protocols that exceed industry standards for impurity profiling and structural characterization, providing pharmaceutical partners with complete confidence in material consistency and regulatory compliance across all production scales.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your development programs. Take advantage of our Customized Cost-Saving Analysis service to evaluate how this innovative manufacturing approach can optimize your supply chain economics while ensuring reliable access to high-purity intermediates critical to your drug development timelines.
