Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Drug Discovery
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Drug Discovery
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable pathways to bioactive scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a robust preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These specific molecular frameworks are of immense interest to the pharmaceutical and material science sectors due to their versatility as ligands in organic light-emitting diodes (OLEDs) and their prevalence in drug intermediates. The patent outlines a novel oxidative cyclization strategy that bypasses the limitations of classical synthesis, offering a streamlined route that is both economically viable and environmentally friendlier. By leveraging a metal-free catalytic system, this technology addresses critical pain points in modern process chemistry, specifically the reliance on toxic heavy metals and complex multi-step sequences.
For R&D directors and process chemists, the ability to access diverse 1,2,4-triazole derivatives with high functional group tolerance is paramount. The methodology described allows for the modular introduction of various substituents on both the quinoline and the triazole rings, enabling the rapid generation of compound libraries for structure-activity relationship (SAR) studies. This flexibility is crucial for optimizing lead candidates in drug discovery programs. Furthermore, the operational simplicity of the reaction—requiring no stringent anhydrous or anaerobic conditions—makes it highly attractive for translation from the laboratory bench to pilot plant operations. As we delve deeper into the technical specifics, it becomes clear that this patent represents a substantial leap forward in the manufacturing of complex nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been fraught with inefficiencies that hinder large-scale application. Traditional protocols often rely on quinoline-2-carboxylic acid as the primary starting material. This precursor necessitates a cumbersome five-step reaction sequence to achieve the final target molecule. Such multi-step syntheses inherently suffer from cumulative yield losses; in the case of the conventional method cited in the background of the patent, the total yield is a dismal 17%. Beyond the poor atom economy, these older routes typically demand severe reaction conditions, including the use of hazardous reagents and strict environmental controls that drive up operational costs. The reliance on carboxylic acid derivatives also limits the structural diversity that can be easily accessed, as functionalizing the quinoline ring prior to triazole formation can be synthetically challenging. For procurement managers, these factors translate into higher raw material costs and longer lead times, making the conventional approach unsustainable for commercial supply chains.
The Novel Approach
In stark contrast, the invention disclosed in CN113307790B introduces a direct and efficient oxidative cyclization pathway. This novel approach utilizes readily available and inexpensive 2-methylquinoline and trifluoroacetohydrazide derivatives as the foundational building blocks. The reaction is promoted by a synergistic system of tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), facilitating the transformation in a single pot. This drastic reduction in step count—from five steps down to essentially one major transformation—significantly enhances the overall process efficiency. The method tolerates a wide range of functional groups, allowing for the synthesis of diversified products with substituents such as methyl, methoxy, halogens, and nitro groups at various positions on the aromatic rings. This modularity is a game-changer for medicinal chemists seeking to optimize pharmacokinetic properties.
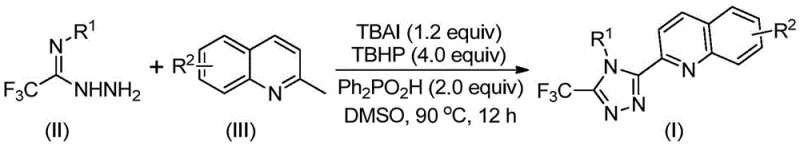
The visual representation of this transformation highlights the elegance of the new route. As shown in the reaction scheme, the coupling of the hydrazide component (Formula II) with the methylquinoline component (Formula III) proceeds smoothly under thermal conditions to yield the target triazole (Formula I). This direct assembly not only simplifies the workflow but also minimizes waste generation, aligning with green chemistry principles. For a reliable pharmaceutical intermediate supplier, adopting such a streamlined process means faster time-to-market for clients and a more robust supply chain capable of meeting fluctuating demands without the bottlenecks associated with multi-step syntheses.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
Understanding the mechanistic underpinnings of this reaction is essential for R&D teams aiming to replicate or further optimize the process. The transformation is believed to proceed through a radical-mediated oxidative pathway initiated by the TBAI and TBHP system. Initially, the methyl group on the 2-methylquinoline undergoes oxidation to generate a reactive 2-quinolinecarbaldehyde intermediate in situ. This aldehyde then undergoes a condensation reaction with the trifluoroacetohydrazide to form a dehydrated hydrazone intermediate. This step is critical as it sets the stage for the subsequent ring closure. The presence of diphenylphosphinic acid (Ph2PO2H) plays a pivotal role as an additive, likely stabilizing intermediates or facilitating proton transfer events that lower the activation energy of the cyclization step.
Following the formation of the hydrazone, the system undergoes oxidative iodination, where the iodine species generated from TBAI activates the hydrazone carbon. This is followed by an intramolecular electrophilic substitution reaction, where the nitrogen nucleophile attacks the activated carbon center, closing the five-membered triazole ring. Finally, an aromatization step occurs, driven by the oxidant, to yield the stable 3-quinolyl-5-trifluoromethyl-1,2,4-triazole product. The entire sequence is a testament to the power of organocatalysis in constructing complex heterocycles. The mechanism also suggests a degree of radical character, which explains the broad substrate scope and the tolerance for various electronic environments on the aromatic rings. This deep mechanistic understanding assures quality control teams that the process is robust and predictable, minimizing the risk of unexpected impurity profiles that often plague transition metal-catalyzed reactions.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and purity. The patent provides a detailed protocol that balances reagent stoichiometry with thermal conditions to ensure optimal performance. The procedure is designed to be user-friendly, avoiding the need for specialized equipment like gloveboxes or high-pressure reactors. By following the standardized steps outlined below, chemists can reliably produce high-purity intermediates suitable for downstream applications. The use of dimethyl sulfoxide (DMSO) as the preferred solvent ensures excellent solubility of all reactants, promoting homogeneous reaction kinetics.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphinic acid, trifluoroacetohydrazide derivative, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-quinolyl-5-trifluoromethyl-1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers compelling advantages that directly impact the bottom line and supply chain resilience. The shift away from precious metal catalysts to an organic iodide/peroxide system eliminates a major cost driver in fine chemical manufacturing. Furthermore, the simplification of the synthetic route reduces the number of unit operations, thereby lowering labor costs and facility occupancy time. For supply chain heads, the reliability of raw material sourcing is a critical metric, and this method excels by utilizing commodity chemicals that are widely available in the global market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or copper results in substantial cost savings. In traditional cross-coupling or cyclization reactions, the cost of the catalyst and the subsequent purification steps required to remove trace metal residues can account for a significant portion of the total production cost. By utilizing a metal-free system based on TBAI and TBHP, manufacturers can avoid these expenses entirely. Additionally, the high atom economy of the one-pot process reduces solvent consumption and waste disposal costs. The use of cheap starting materials like 2-methylquinoline further drives down the cost of goods sold (COGS), making the final API intermediate more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method contributes significantly to supply chain stability. Because the reaction does not require strict anhydrous or oxygen-free conditions, it is less susceptible to failures caused by minor deviations in operational parameters. This tolerance makes the process easier to scale and manage across different manufacturing sites. Moreover, the starting materials are commercially available in bulk quantities, reducing the risk of supply disruptions associated with specialized or custom-synthesized precursors. This reliability ensures consistent delivery schedules for downstream pharmaceutical clients, fostering long-term partnerships and trust.
- Scalability and Environmental Compliance: The method is inherently scalable, having been demonstrated to work effectively on gram scales with the potential for expansion to kilogram and ton levels. The absence of heavy metals simplifies the regulatory compliance landscape, as there is no need for rigorous testing and validation of metal clearance in the final product. This aligns well with increasingly stringent environmental regulations regarding heavy metal discharge and residual limits in pharmaceuticals. The simpler work-up procedure, involving filtration and standard column chromatography, facilitates easier scale-up compared to complex extraction or crystallization processes often required for metal-catalyzed reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of these valuable triazole intermediates. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for potential partners and technical stakeholders.
Q: What are the key advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-carboxylic acid which suffer from low yields (around 17%) and harsh multi-step conditions, this novel approach utilizes cheap 2-methylquinoline and operates under mild, metal-free conditions with significantly higher efficiency.
Q: Does this process require expensive transition metal catalysts?
A: No, the process described in patent CN113307790B is entirely metal-free. It utilizes an organic catalytic system involving tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), eliminating the need for costly palladium or copper catalysts and subsequent heavy metal removal steps.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It does not require strict anhydrous or oxygen-free conditions, uses commercially available and inexpensive raw materials, and has been demonstrated to work effectively on gram scales, indicating strong potential for kilogram to ton-level manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced heterocyclic intermediates in the development of next-generation therapeutics and functional materials. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN113307790B and is fully equipped to leverage this metal-free oxidative cyclization route for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to market is seamless. Our state-of-the-art facilities are designed to handle complex organic syntheses with precision, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality standards for every batch produced.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your project specifics, obtain specific COA data for our available intermediates, and receive comprehensive route feasibility assessments. Let us be your trusted partner in delivering high-purity pharmaceutical intermediates that drive innovation and efficiency in your drug development pipeline.
