Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Intermediates for Global Pharma Supply Chains
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Intermediates for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These specific molecular architectures are of immense value due to the prevalence of the 1,2,4-triazole ring in numerous bioactive frameworks and its utility as a bidentate ligand in coordination chemistry and organic light-emitting diodes (OLEDs). The disclosed technology represents a paradigm shift from laborious, low-yielding traditional syntheses to a streamlined, metal-free oxidative cyclization process. By leveraging a catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), this innovation enables the direct coupling of readily available 2-methylquinolines with trifluoroacetamidine hydrazides. For R&D directors and procurement specialists alike, this patent offers a compelling value proposition: a route that eliminates the need for expensive transition metal catalysts, avoids stringent anhydrous or oxygen-free operating conditions, and delivers high-purity intermediates suitable for immediate downstream application in drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the methodology described in patent CN113307790B, the synthesis of quinolyl-substituted 1,2,4-triazoles was fraught with significant inefficiencies that hindered commercial viability. The state-of-the-art traditional approach typically relied on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to achieve the final target molecule. This multi-step lineage not only compounded material losses at each stage but also subjected the intermediates to severe reaction conditions that often degraded sensitive functional groups. Consequently, the overall yield of this conventional pathway was abysmally low, reported at merely 17%, rendering it economically unfeasible for large-scale manufacturing. Furthermore, the reliance on multiple isolation and purification steps between each transformation increased the consumption of solvents and silica gel, driving up both the environmental footprint and the cost of goods sold (COGS). For supply chain managers, such a fragile process posed substantial risks regarding batch-to-batch consistency and the ability to meet tight production deadlines, as any deviation in one of the five steps could compromise the entire batch.
The Novel Approach
In stark contrast to the arduous traditional routes, the novel approach presented in the patent utilizes a direct, one-pot oxidative cyclization strategy that dramatically simplifies the synthetic landscape. By employing cheap and easily obtainable 2-methylquinoline derivatives and trifluoroacetamidine hydrazides as starting materials, the method bypasses the need for pre-functionalized carboxylic acids. The core of this innovation lies in the use of a TBAI/TBHP catalytic system promoted by diphenylphosphoric acid in a polar aprotic solvent like DMSO. This system facilitates the in situ oxidation of the methyl group on the quinoline ring to an aldehyde, which subsequently undergoes condensation with the hydrazide followed by oxidative cyclization to form the triazole ring. 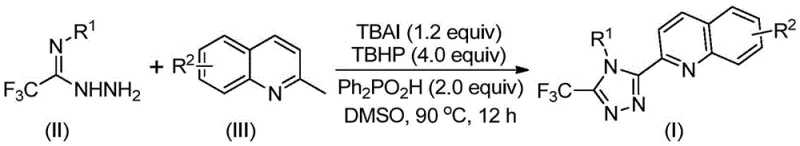 This telescoped process reduces the synthetic timeline from five discrete steps to a single thermal operation conducted at moderate temperatures between 80°C and 100°C. The elimination of intermediate isolations not only boosts the overall mass balance but also significantly reduces the operational complexity, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations aiming for high throughput and minimal waste generation.
This telescoped process reduces the synthetic timeline from five discrete steps to a single thermal operation conducted at moderate temperatures between 80°C and 100°C. The elimination of intermediate isolations not only boosts the overall mass balance but also significantly reduces the operational complexity, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations aiming for high throughput and minimal waste generation.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The mechanistic underpinning of this transformation is a sophisticated interplay of radical oxidation and electrophilic substitution, orchestrated by the iodine/peroxide couple. The reaction initiates with the activation of tert-butyl hydroperoxide (TBHP) by tetrabutylammonium iodide (TBAI), generating reactive iodine species and tert-butoxyl radicals. These active species are responsible for the selective oxidation of the benzylic methyl group on the 2-methylquinoline substrate, converting it first to an alcohol and subsequently to the corresponding 2-quinolinecarbaldehyde in situ. This aldehyde intermediate then undergoes a condensation reaction with the trifluoroacetamidine hydrazide to form a dehydrated hydrazone species. Following this condensation, the system promotes an oxidative iodination event, likely involving the formation of an iodonium intermediate, which triggers an intramolecular electrophilic substitution. This cyclization step closes the five-membered triazole ring, followed by a final aromatization process that expels the iodine species and restores aromaticity to the newly formed heterocycle. The presence of diphenylphosphoric acid is crucial, acting as a proton shuttle and stabilizing agent that enhances the efficiency of the cyclization step while suppressing side reactions.
From an impurity control perspective, this metal-free mechanism offers distinct advantages over transition-metal catalyzed alternatives. The absence of heavy metals such as palladium, copper, or iron eliminates the risk of metal contamination in the final API intermediate, a critical quality attribute for regulatory compliance. Furthermore, the radical nature of the oxidation ensures high chemoselectivity, tolerating a wide range of functional groups including halogens, nitro groups, and ethers without causing undesired side reactions like dehalogenation or over-oxidation. The use of DMSO as the solvent further aids in stabilizing polar intermediates and ensuring homogeneous reaction conditions, which is vital for maintaining consistent kinetics across large batches. This robust mechanistic profile ensures that the resulting 3-quinolyl-5-trifluoromethyl-1,2,4-triazole compounds possess a clean impurity profile, reducing the burden on downstream purification teams and facilitating faster release testing for commercial batches.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a production environment requires precise adherence to the optimized stoichiometry and thermal parameters outlined in the patent data. The process is designed to be operationally simple, requiring standard glass-lined or stainless steel reactors capable of handling mild oxidative conditions. The key to success lies in the molar ratios of the oxidant and catalyst; specifically, maintaining a molar ratio of TBAI to TBHP to diphenylphosphoric acid at approximately 1.0-1.5 : 4 : 2 ensures maximal conversion of the starting materials. The reaction is typically run with a slight excess of the hydrazide component relative to the quinoline to drive the equilibrium towards the product.  Post-reaction workup is straightforward, involving filtration to remove insoluble salts followed by silica gel mixing and standard column chromatography, although on a multi-ton scale, crystallization or extraction protocols would be developed to replace chromatography for cost efficiency. The detailed standardized synthesis steps for replicating this high-yield process are provided in the guide below.
Post-reaction workup is straightforward, involving filtration to remove insoluble salts followed by silica gel mixing and standard column chromatography, although on a multi-ton scale, crystallization or extraction protocols would be developed to replace chromatography for cost efficiency. The detailed standardized synthesis steps for replicating this high-yield process are provided in the guide below.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphoric acid, trifluoroacetamidine hydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80°C to 100°C and maintain stirring for a duration of 8 to 14 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the high-purity 3-quinolyl-5-trifluoromethyl-1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple yield improvements. The primary driver of value is the drastic simplification of the supply chain for raw materials; 2-methylquinolines and trifluoroacetic acid derivatives are commodity chemicals available from multiple global vendors, mitigating the risk of single-source dependency. Moreover, the elimination of expensive transition metal catalysts removes a significant line item from the bill of materials, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. The process does not require specialized equipment for handling air-sensitive or moisture-sensitive reagents, meaning it can be executed in standard multipurpose reactors without the need for nitrogen blanketing or rigorous drying protocols, thereby lowering capital expenditure and operational overheads.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the convergence of several efficiency factors. By collapsing a five-step synthesis into a single pot, the method eliminates the labor, time, and solvent costs associated with four intermediate isolation and purification stages. The avoidance of precious metal catalysts not only saves on the initial purchase price but also removes the costly and time-consuming step of heavy metal scavenging required to meet strict ppm limits in pharmaceutical products. Additionally, the high atom economy of the oxidative cyclization minimizes waste generation, leading to lower disposal costs and a more sustainable manufacturing footprint. These cumulative savings allow for a significantly reduced cost of goods, providing a competitive pricing advantage in the global market for complex heterocyclic intermediates.
- Enhanced Supply Chain Reliability: Operational resilience is greatly enhanced by the robustness of the reaction conditions. Since the protocol functions effectively without anhydrous or anaerobic environments, the risk of batch failure due to minor leaks or moisture ingress is virtually eliminated. This tolerance translates to higher first-pass yields and more predictable production schedules, ensuring that delivery commitments to downstream API manufacturers are met consistently. The broad substrate scope demonstrated in the patent, accommodating various electron-donating and electron-withdrawing groups, means that a single platform technology can be used to produce a diverse library of analogues. This flexibility allows suppliers to rapidly pivot production to meet changing demand for specific drug candidates without requalifying entirely new synthetic routes, thereby securing the continuity of supply for critical therapeutic programs.
- Scalability and Environmental Compliance: The transition from laboratory benchtop to commercial scale is facilitated by the use of common solvents like DMSO and stable reagents that do not pose extreme safety hazards. The reaction temperatures of 80°C to 100°C are easily manageable with standard heating systems, avoiding the energy intensity of cryogenic or high-pressure processes. From an environmental standpoint, the metal-free nature of the catalysis aligns perfectly with green chemistry principles, reducing the toxic load in wastewater streams. The high yields reported, reaching up to 97% in optimized examples, mean that less raw material is wasted per kilogram of product, further enhancing the sustainability profile. This combination of safety, scalability, and environmental friendliness makes the process highly attractive for regulatory approval and long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating the feasibility of this route for their specific projects. Understanding these nuances is essential for making informed decisions about process adoption and vendor selection.
Q: What are the primary advantages of this metal-free synthesis over traditional methods?
A: Unlike traditional multi-step routes requiring harsh conditions and yielding only 17%, this novel method operates in a single pot without heavy metal catalysts or strict anhydrous conditions, significantly simplifying purification and reducing operational costs.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the protocol utilizes cheap, commercially available starting materials like 2-methylquinoline and avoids sensitive reagents, making it highly amenable to scale-up from gram-scale laboratory synthesis to multi-ton industrial manufacturing.
Q: What is the substrate scope for the R1 and R2 positions?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents such as methyl, methoxy, halogens (Cl, Br), nitro, and trifluoromethyl groups on both the quinoline and phenyl rings with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of life-saving medications. Our technical team has thoroughly analyzed the methodology disclosed in CN113307790B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative chemistry to the global market. We are committed to delivering these complex scaffolds with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the exacting standards required by top-tier pharmaceutical companies. Our facility is equipped to handle the specific solvent and thermal requirements of this oxidative cyclization, ensuring a seamless transition from pilot plant to full-scale manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact us today to request specific COA data for our reference standards and comprehensive route feasibility assessments. Let us collaborate to accelerate your drug development timelines with a reliable, cost-effective, and scalable source of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole intermediates.
