Advanced Synthesis of Phosphine Chiral Center Compounds for Asymmetric Catalysis
Introduction to Patent CN113087742B Technology
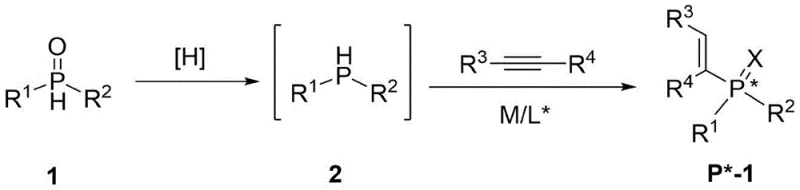
The landscape of asymmetric catalysis is continually evolving, driven by the demand for highly enantioselective transformations in the synthesis of active pharmaceutical ingredients and fine chemicals. Patent CN113087742B introduces a groundbreaking methodology for the preparation of compounds containing a phosphine chiral center, addressing long-standing challenges in the field of organophosphorus chemistry. Traditionally, the direct catalytic asymmetric synthesis of chiral trivalent phosphine compounds has been hindered by the instability and toxicity of the requisite starting materials. This patent discloses a robust two-step protocol that utilizes stable secondary phosphine oxides as precursors, which are subsequently reduced and coupled with alkynes under nickel catalysis. This approach not only enhances the safety profile of the synthesis but also expands the structural diversity of accessible chiral ligands, offering a reliable pathway for producing high-purity chiral phosphines essential for modern homogeneous catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral trivalent phosphine compounds has relied heavily on the use of secondary phosphines (R2PH) as direct substrates for metal-catalyzed coupling reactions. However, secondary phosphines are notoriously difficult to handle due to their extreme air sensitivity, high toxicity, and unpleasant odor, which necessitates rigorous inert atmosphere techniques and specialized storage facilities. Furthermore, the limited availability of diverse secondary phosphine starting materials restricts the structural variety of the resulting ligands, often forcing researchers to rely on a narrow subset of commercially available options. Previous methods reported by groups such as Toste, Glueck, and Hayashi, while effective, often suffer from these substrate limitations and may require harsh conditions or expensive noble metal catalysts like Palladium or Platinum, which can complicate downstream purification and increase the overall cost of goods for large-scale manufacturing processes.
The Novel Approach
The methodology outlined in CN113087742B represents a paradigm shift by employing secondary phosphine oxides (SPOs) as the primary feedstock. SPOs are bench-stable, non-volatile, and significantly less toxic than their trivalent counterparts, drastically simplifying the supply chain and handling requirements. The process involves an initial reduction step where the SPO is converted into the reactive secondary phosphine hydride in situ or as a discrete intermediate using mild reducing agents such as silanes or boranes. This generated hydride is then immediately engaged in a nickel-catalyzed asymmetric hydrophosphination reaction with various alkynes. By decoupling the stability issues of the starting material from the reactivity required for bond formation, this novel approach allows for the efficient and highly selective construction of P-chiral centers. The use of nickel, a more abundant and cost-effective metal compared to palladium or rhodium, further underscores the economic advantages of this synthetic route for industrial applications.
Mechanistic Insights into Nickel-Catalyzed Asymmetric Hydrophosphination
The core of this technology lies in the nickel-catalyzed addition of the P-H bond across the carbon-carbon triple bond of an alkyne. The reaction mechanism typically involves the oxidative addition of the secondary phosphine hydride to a low-valent nickel(0) species, which is generated in situ from precursors like Ni(COD)2 in the presence of a chiral ligand. This step forms a nickel(II) hydrido-phosphido complex that retains the chirality induced by the ligand environment. Subsequent coordination and insertion of the alkyne into the Ni-P or Ni-H bond determines the regioselectivity and stereoselectivity of the final product. The chiral ligand, such as ferrocenyl bisphosphines or binaphthyl derivatives, creates a sterically defined pocket around the metal center, effectively differentiating the enantiotopic faces of the alkyne substrate. This precise control is critical for achieving high enantiomeric excess (ee) values, with the patent reporting examples reaching up to 96% ee, ensuring that the resulting ligands are suitable for demanding asymmetric catalytic applications where optical purity is paramount.
Impurity control is inherently managed through the choice of starting materials and reaction conditions. Since the secondary phosphine oxide precursors are stable solids or liquids that can be purified by standard crystallization or distillation prior to use, the baseline purity of the reaction input is high. The reduction step is monitored to ensure complete conversion, minimizing the carryover of unreacted oxide which could act as a poison in subsequent catalytic cycles. Furthermore, the reaction is typically quenched with protecting reagents such as sulfur powder or borane dimethyl sulfide to convert the sensitive trivalent phosphine product into a more stable pentavalent species (e.g., phosphine sulfide or phosphine-borane adduct) before purification. This protection step prevents oxidation during workup and column chromatography, thereby preserving the optical integrity of the chiral center and ensuring a clean impurity profile in the final isolated material.
How to Synthesize Chiral Phosphine Compounds Efficiently
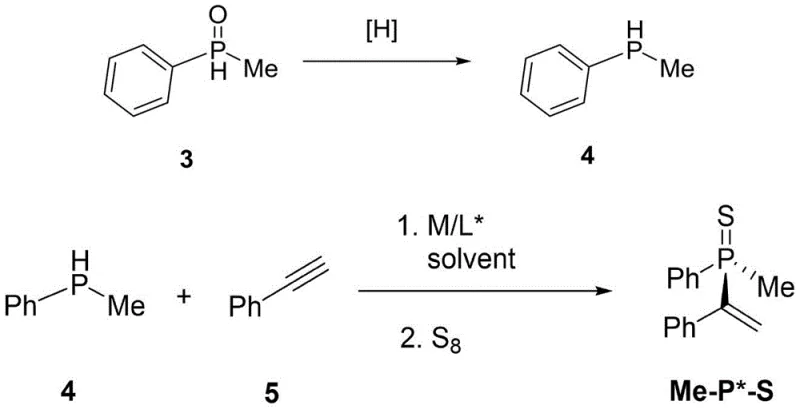
The synthesis protocol described in the patent offers a streamlined workflow that balances operational simplicity with high chemical performance. The process begins with the reduction of the secondary phosphine oxide, followed by the nickel-catalyzed coupling with an alkyne. Detailed operational parameters, including specific molar ratios, solvent choices, and temperature profiles, are critical for reproducing the high yields and selectivities reported in the examples. For instance, maintaining the reaction temperature between -30°C and 50°C during the coupling step is essential to balance reaction rate against potential racemization or side reactions. The following guide outlines the standardized steps derived from the patent data to assist process chemists in implementing this technology.
- Reduce secondary phosphine oxide to secondary phosphine hydride using a silane or borane reducing agent at 20-100°C for 12-120 hours.
- React the resulting secondary phosphine with an alkyne using a Nickel catalyst and chiral ligand in organic solvent at -30 to 50°C.
- Quench with a protecting reagent like sulfur or borane, then purify via column chromatography to isolate the chiral phosphine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic methodology offers tangible benefits that extend beyond mere chemical efficiency. The primary advantage stems from the substitution of hazardous secondary phosphines with stable secondary phosphine oxides. This change fundamentally alters the logistics of raw material management, eliminating the need for specialized cold storage or strict inert gas blanketing during warehousing. Consequently, this leads to significant cost savings in terms of infrastructure and safety compliance, as the hazards associated with transportation and handling are drastically reduced. Moreover, the use of nickel catalysts instead of precious metals like palladium or rhodium reduces the exposure to volatile commodity prices of noble metals, providing greater budget predictability for long-term manufacturing contracts.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts in favor of nickel-based systems directly lowers the raw material cost per kilogram of the produced ligand. Additionally, the stability of the oxide precursors reduces waste associated with material degradation during storage, further optimizing the cost structure. The ability to perform the reaction in common organic solvents like toluene or THF without requiring exotic additives simplifies the solvent recovery process, contributing to overall process economy and reducing the environmental footprint of the manufacturing site.
- Enhanced Supply Chain Reliability: Secondary phosphine oxides are commercially available from a broader range of suppliers compared to specialized chiral secondary phosphines, mitigating the risk of single-source dependency. The robust nature of these starting materials ensures consistent quality upon delivery, reducing the frequency of incoming quality control rejections. This reliability translates into more predictable production schedules and shorter lead times for the final chiral ligands, enabling downstream customers to maintain leaner inventory levels without compromising their own production continuity.
- Scalability and Environmental Compliance: The reaction conditions described, operating at moderate temperatures and pressures, are inherently scalable from gram to multi-kilogram batches without requiring significant re-engineering of the process equipment. The reduced toxicity profile of the starting materials simplifies waste stream treatment and disposal, facilitating compliance with increasingly stringent environmental regulations. This ease of scale-up ensures that the technology can meet the demands of commercial production, supporting the transition from laboratory discovery to industrial manufacturing of complex organometallic catalysts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and specifications provided in CN113087742B, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this synthesis route into existing production workflows or new product development pipelines.
Q: Why use secondary phosphine oxides instead of secondary phosphines directly?
A: Secondary phosphine oxides are significantly more stable, less toxic, and have lower odor compared to secondary phosphines, which are air-sensitive and hazardous. This improves safety and storage logistics.
Q: What metals can be complexed with these chiral phosphine ligands?
A: The synthesized ligands can form complexes with a wide range of transition metals including Ruthenium, Rhodium, Iridium, Palladium, Platinum, Gold, and Nickel, suitable for various catalytic applications.
Q: What represents the enantioselectivity of this method?
A: The method achieves high enantioselectivity, with examples showing ee values up to 96-97% depending on the specific substrate and chiral ligand system employed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphine Ligands Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing for advanced chemical intermediates. Leveraging the innovative pathways described in patents like CN113087742B, we possess the technical expertise to scale diverse reaction pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to ensure enantiomeric excess meets the highest industry standards. We understand that the consistency of chiral ligands is critical for the performance of downstream catalytic processes, and our quality management systems are designed to guarantee batch-to-batch reproducibility.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain insights into how switching to this oxide-based route can optimize your bill of materials. We encourage you to contact us for specific COA data and route feasibility assessments to validate the compatibility of these chiral phosphine compounds with your existing catalytic systems. Let us collaborate to drive efficiency and innovation in your supply chain.
