Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Global Pharma Supply Chains
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a novel preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These nitrogen-containing heterocycles are not merely academic curiosities; they serve as critical building blocks in the development of bioactive molecular frameworks, ranging from potent drug intermediates to functional ligands in organic light-emitting diodes (OLEDs). The structural versatility of the 1,2,4-triazole ring allows it to act as a bidentate ligand in cross-coupling reactions, thereby expanding its utility in medicinal chemistry and material science. As shown in the broader context of bioactive molecules, triazole derivatives are foundational to numerous therapeutic agents, underscoring the commercial imperative for efficient synthesis routes.
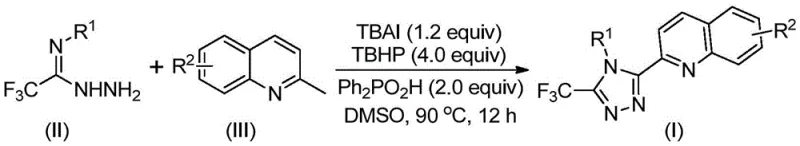
Traditionally, the synthesis of quinolyl-substituted 1,2,4-triazoles has been fraught with inefficiencies that hinder large-scale production. Conventional methods often rely on quinoline-2-carboxylic acid as a starting material, necessitating a cumbersome five-step reaction sequence. This legacy approach suffers from a dismal total yield of approximately 17%, coupled with severe reaction conditions that demand rigorous control and specialized equipment. Such limitations render traditional pathways economically unviable for commercial manufacturing, where margin compression and waste generation are critical concerns. In stark contrast, the novel approach outlined in the patent leverages cheap and easily obtainable starting materials—specifically 2-methylquinoline and trifluoroacetimidohydrazide—to achieve a direct, one-pot oxidative cyclization. This paradigm shift eliminates the need for multi-step isolation and purification, drastically reducing the operational burden on production facilities while enhancing overall atom economy.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core innovation of this technology lies in its elegant mechanistic pathway, which avoids the pitfalls of transition metal catalysis. The reaction initiates with the promotion of tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), which function synergistically to oxidize the methyl group of 2-methylquinoline into a reactive 2-quinoline carbaldehyde intermediate in situ. This aldehyde subsequently undergoes a condensation reaction with trifluoroacetimidohydrazide to form a dehydrated hydrazone intermediate. The process continues through a sophisticated sequence of oxidative iodination, intramolecular electrophilic substitution, and final aromatization to yield the target 3-quinolyl-5-trifluoromethyl-1,2,4-triazole structure. Alternatively, the reaction may proceed via a radical mechanism, facilitated by the peroxide oxidant. This metal-free trajectory is particularly advantageous for pharmaceutical applications, as it inherently mitigates the risk of heavy metal contamination, a stringent regulatory requirement for Active Pharmaceutical Ingredients (APIs).
From an impurity control perspective, the mild nature of this oxidative system offers distinct advantages over harsher traditional methods. By operating at moderate temperatures between 80°C and 100°C, the process minimizes thermal degradation and the formation of complex polymeric byproducts often associated with high-energy activation. The use of diphenylphosphoric acid as an additive further stabilizes the reaction environment, promoting high selectivity for the desired triazole ring closure. Furthermore, the broad substrate tolerance allows for the introduction of diverse functional groups—such as methyl, methoxy, halogens, and nitro groups—at various positions on both the quinoline and the aryl rings without compromising yield. This flexibility ensures that the impurity profile remains manageable and predictable, facilitating easier downstream purification and consistent quality control for high-purity pharmaceutical intermediates.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles Efficiently
Implementing this synthesis in a production environment requires precise adherence to the optimized parameters established in the patent data. The protocol is designed for operational simplicity, utilizing common organic solvents like DMSO, DMF, or dioxane, with DMSO showing superior conversion rates. The reaction does not demand inert atmosphere techniques, allowing it to be conducted in standard reactor vessels without the need for expensive gloveboxes or nitrogen purging systems. This accessibility significantly lowers the barrier to entry for contract manufacturing organizations (CMOs) looking to adopt this technology. For detailed procedural specifics regarding stoichiometry, addition rates, and workup procedures, please refer to the standardized synthesis guide below.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphoric acid, trifluoroacetimidohydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature between 80°C and 100°C and maintain stirring for 8 to 14 hours to ensure complete oxidative cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-quinolyl-5-trifluoromethyl-1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route represents a strategic opportunity to optimize cost structures and enhance supply reliability. The elimination of precious metal catalysts removes a significant variable cost component and simplifies the supply chain by reducing dependency on volatile metal markets. Moreover, the use of commodity chemicals like 2-methylquinoline and tert-butyl hydroperoxide ensures that raw material sourcing remains stable and resilient against market fluctuations. The robustness of the reaction conditions also translates to reduced downtime and higher throughput in manufacturing plants, directly addressing the industry-wide challenge of cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the complete avoidance of expensive transition metal catalysts such as palladium or copper. In traditional cross-coupling or cyclization reactions, these metals not only incur high upfront material costs but also necessitate costly downstream purification steps to meet residual metal specifications (often in the ppm range). By utilizing an organocatalytic system based on TBAI and TBHP, the process inherently bypasses these expenses. Additionally, the one-pot nature of the reaction consolidates multiple synthetic steps into a single operation, significantly reducing solvent consumption, labor hours, and energy usage associated with intermediate isolations. This streamlined workflow results in substantial operational expenditure (OPEX) savings without sacrificing product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for global pharmaceutical production, and this method strengthens resilience by relying on widely available, commodity-grade starting materials. Unlike specialized reagents that may have long lead times or single-source suppliers, 2-methylquinoline and trifluoroacetimidohydrazide are commercially accessible from multiple vendors worldwide. The reaction's tolerance to ambient conditions (no strict anhydrous or anaerobic requirements) further reduces logistical complexities, as materials do not require specialized packaging or transport under inert gas. This ease of handling minimizes the risk of production delays caused by material degradation or shipping constraints, ensuring a steady flow of reliable pharmaceutical intermediate supplier outputs to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to tonnage levels often introduces unforeseen engineering challenges, but this oxidative cyclization is inherently scalable due to its thermal stability and lack of hazardous reagents. The reaction operates safely at temperatures between 80°C and 100°C, avoiding the extreme pressures or cryogenic conditions that complicate scale-up. From an environmental standpoint, the absence of heavy metals simplifies waste treatment protocols, reducing the burden on effluent processing facilities and lowering disposal costs. The high atom efficiency and reduced solvent usage align with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals while maintaining high production volumes for commercial scale-up of complex heterocyclic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is crucial for assessing the feasibility of integrating this technology into existing production lines.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method utilizes an organocatalytic system involving tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), completely eliminating the need for toxic or expensive heavy metal catalysts like palladium or copper.
Q: What is the substrate scope for the quinoline component?
A: The method demonstrates excellent tolerance for various substituents on the quinoline ring, including hydrogen, methyl, methoxy, halogens (Cl, Br), and nitro groups at positions 6, 7, or 8, allowing for diverse library synthesis.
Q: Are anhydrous or oxygen-free conditions necessary for this reaction?
A: No, one of the key advantages of this protocol is its operational simplicity; it does not require strict anhydrous or anaerobic conditions, making it highly suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free oxidative cyclization technologies in modernizing the supply chain for critical pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole conforms to the highest international standards required for drug substance manufacturing.
We invite global partners to collaborate with us to leverage this advanced synthetic route for their specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive landscape of fine chemical manufacturing.
