Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Intermediates for Advanced Drug Discovery
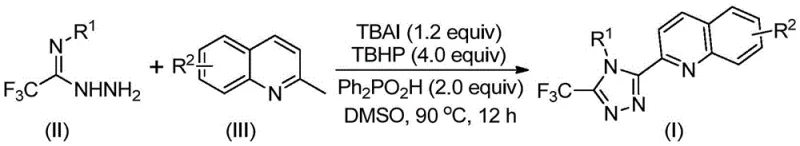
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-rich heterocycles, particularly 1,2,4-triazoles, which serve as critical scaffolds in bioactive molecules and functional materials. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. This innovation addresses long-standing challenges in heterocyclic synthesis by replacing complex, multi-step sequences with a streamlined oxidative cyclization protocol. By leveraging a metal-free catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), the process achieves remarkable conversion rates under relatively mild thermal conditions. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiency and operational complexity. Traditional literature methods typically rely on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to arrive at the target structure. This legacy approach suffers from a dismal total yield of approximately 17%, rendering it economically unviable for large-scale production. Furthermore, these conventional routes often demand severe reaction conditions, including strict temperature controls and the use of hazardous reagents, which escalate both safety risks and waste disposal costs. The reliance on such inefficient pathways creates a bottleneck in the supply chain for drug candidates requiring this specific heterocyclic motif, forcing manufacturers to absorb high production costs and extended lead times.
The Novel Approach
In stark contrast, the methodology described in patent CN113307790B introduces a paradigm shift by utilizing readily available 2-methylquinoline and trifluoroacetimidohydrazide as starting materials. This novel route condenses the synthesis into a direct oxidative cyclization, bypassing the need for pre-functionalized carboxylic acid derivatives. The reaction is promoted by a synergistic combination of TBAI and TBHP, facilitated by diphenylphosphinic acid in a polar aprotic solvent like DMSO. This approach not only drastically improves atom economy but also eliminates the need for toxic heavy metal catalysts, which are notoriously difficult to remove from final API intermediates. The result is a process that is operationally simple, does not require anhydrous or oxygen-free environments, and delivers superior yields, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core of this technological advancement lies in the intricate mechanistic pathway driven by the TBAI/TBHP catalytic system. The reaction initiates with the oxidation of the methyl group on the 2-methylquinoline substrate, effectively converting it in situ into a reactive 2-quinolinecarbaldehyde equivalent. This transient aldehyde species then undergoes a condensation reaction with the trifluoroacetimidohydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination and intramolecular electrophilic substitution facilitate the closure of the triazole ring. Finally, aromatization yields the stable 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole product. The presence of diphenylphosphinic acid is believed to stabilize key intermediates and enhance the overall reaction kinetics, ensuring high conversion efficiency even at moderate temperatures between 80°C and 100°C.
From an impurity control perspective, this metal-free mechanism offers distinct advantages over transition-metal catalyzed alternatives. The absence of copper, palladium, or iron catalysts means there is no risk of heavy metal leaching into the product stream, a critical parameter for regulatory compliance in pharmaceutical manufacturing. The reaction profile suggests a radical-mediated process that is highly selective for the desired cyclization, minimizing the formation of side products associated with over-oxidation or polymerization. This inherent selectivity simplifies the downstream purification process, often allowing for straightforward filtration and column chromatography to achieve high-purity specifications. For quality assurance teams, this translates to a cleaner impurity profile and reduced analytical burden during batch release testing.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis route requires precise control over reagent stoichiometry and thermal parameters to maximize yield and reproducibility. The patent data indicates that the molar ratio of the oxidant TBHP to the catalyst TBAI is critical, with optimal results observed when using a slight excess of the oxidant to drive the reaction to completion. The choice of solvent is equally important, with DMSO demonstrating superior performance in dissolving the polar intermediates and facilitating the ionic radical mechanisms involved. Operators should note that while the reaction is tolerant to ambient conditions, maintaining a consistent temperature range of 80-100°C for 8 to 14 hours is essential for full conversion. Detailed standardized operating procedures for this transformation are outlined below to ensure consistent batch-to-batch quality.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphinic acid, trifluoroacetimidohydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and maintain stirring for 8 to 14 hours to ensure complete conversion without requiring inert atmosphere.
- Perform post-treatment by filtering the mixture, mixing with silica gel, and purifying via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers transformative economic and logistical benefits. The elimination of expensive transition metal catalysts removes a significant cost center associated with both raw material acquisition and specialized waste treatment. Furthermore, the use of commodity chemicals like 2-methylquinoline and TBHP ensures a stable and resilient supply chain, reducing vulnerability to market fluctuations affecting exotic reagents. The operational simplicity of the process, which foregoes the need for inert atmosphere reactors, allows for utilization of standard glass-lined or stainless steel equipment, thereby lowering capital expenditure requirements for scale-up. These factors collectively contribute to a substantial reduction in the cost of goods sold (COGS) for the final intermediate.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the synthetic route. By collapsing a five-step sequence into a single pot operation, manufacturers save significantly on labor, energy, and solvent consumption. The avoidance of heavy metals eliminates the costly and time-consuming scavenging steps typically required to meet ppm-level residual metal specifications. Additionally, the high yields reported in the patent examples, reaching up to 97% for certain substrates, mean that less raw material is wasted, directly improving the overall material throughput and profitability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk-grade starting materials enhances the robustness of the supply chain. Unlike specialized catalysts that may have long lead times or single-source dependencies, reagents like TBAI and diphenylphosphinic acid are commercially accessible from multiple global vendors. This diversity in sourcing options mitigates the risk of production stoppages due to raw material shortages. Moreover, the tolerance of the reaction to non-anhydrous conditions reduces the logistical burden of storing and handling sensitive moisture-sensitive reagents, further streamlining warehouse operations and inventory management.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this metal-free protocol aligns perfectly with modern green chemistry principles. The absence of toxic heavy metals simplifies effluent treatment and reduces the environmental footprint of the manufacturing facility. The process is inherently scalable, as demonstrated by its successful application across a diverse range of substrates with varying electronic properties. This scalability ensures that the method can be seamlessly transferred from pilot plant trials to multi-ton commercial production without encountering the nonlinear scale-up issues often associated with complex catalytic cycles, ensuring continuous supply continuity for downstream customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-carboxylic acid and five synthetic steps with only 17% total yield, this novel approach utilizes cheap 2-methylquinoline and achieves yields up to 97% in a single pot without heavy metal catalysts.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, a key operational advantage of this patent technology is that the reaction proceeds efficiently in standard organic solvents like DMSO without the need for rigorous exclusion of water or oxygen, significantly simplifying reactor requirements.
Q: Is the catalyst system suitable for large-scale industrial production?
A: Yes, the use of organocatalysts like TBAI and common oxidants like TBHP avoids toxic heavy metal residues, eliminating expensive removal steps and making the process highly amenable to commercial scale-up and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocyclic synthesis in accelerating drug development timelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN113307790B can be successfully translated into industrial reality. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in metal-free catalytic processes allows us to offer clients a cleaner, more sustainable supply option for complex triazole derivatives.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
