Advanced Biaryl Synthesis: Scalable Reductive Cross-Coupling for Commercial Production
Advanced Biaryl Synthesis: Scalable Reductive Cross-Coupling for Commercial Production
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for more robust and economically viable pathways to access privileged structural motifs. Among these, biaryl structures stand out as fundamental scaffolds found in a vast array of bioactive molecules, agrochemicals, and advanced materials. A significant breakthrough in this domain is documented in patent CN113402350B, which discloses a novel preparation method for biaryl compounds utilizing a direct reductive cross-coupling strategy. This technology addresses long-standing challenges in traditional cross-coupling methodologies by employing readily available aryl quaternary ammonium salts and aryl bromides as coupling partners. By leveraging a palladium-catalyzed system enhanced with magnesium and lithium chloride, this approach offers a streamlined route that operates under remarkably mild conditions, typically between 25°C and 60°C. The implications for industrial manufacturing are profound, as it promises to reduce the reliance on sensitive organometallic reagents while maintaining high yields and exceptional substrate universality.
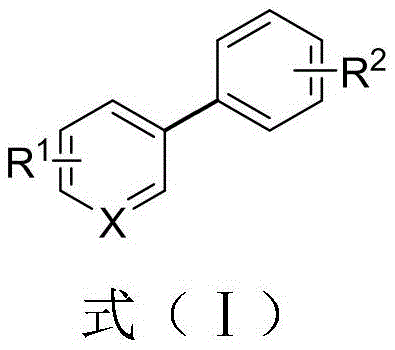
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biaryl linkages has relied heavily on established cross-coupling reactions such as Suzuki-Miyaura or Kumada couplings, which, while effective, impose significant logistical and operational burdens on large-scale production facilities. Traditional methods often necessitate the use of pre-formed organometallic nucleophiles, such as aryl boronic acids or Grignard reagents, which are inherently unstable and sensitive to environmental factors like moisture and oxygen. The preparation and storage of these reagents require stringent anhydrous conditions and specialized infrastructure, leading to increased operational costs and safety hazards in a commercial setting. Furthermore, the presence of sensitive functional groups on the substrate often limits the scope of these reactions, requiring extensive protection and deprotection strategies that add unnecessary steps and reduce overall atom economy. The generation of stoichiometric amounts of metal waste, particularly boron or magnesium salts, also presents substantial environmental compliance challenges and waste disposal costs that modern green chemistry initiatives strive to minimize.
The Novel Approach
In stark contrast to these conventional limitations, the methodology outlined in CN113402350B introduces a paradigm shift by utilizing aryl quaternary ammonium salts as stable electrophilic coupling partners. These salts are not only inexpensive and easy to prepare from ubiquitous amines but also exhibit remarkable stability under ambient conditions, effectively eliminating the need for cold chain storage or inert atmosphere handling prior to the reaction. The direct cross-coupling with aryl bromides, mediated by a catalytic amount of palladium and activated magnesium, allows for a "one-pot" synthesis that significantly simplifies the process workflow. This novel approach bypasses the requirement for pre-generated nucleophiles, thereby reducing the number of unit operations and minimizing the potential for human error during reagent transfer. The result is a more resilient manufacturing process that is less susceptible to batch-to-batch variability caused by reagent degradation, ensuring consistent product quality and reliability for downstream applications in drug discovery and development.
Mechanistic Insights into Pd-Catalyzed Reductive Cross-Coupling
The success of this synthetic route hinges on a sophisticated interplay between the palladium catalyst and the activating agents, specifically the magnesium-lithium chloride complex. The mechanism likely involves the in situ generation of a highly reactive low-valent palladium species, facilitated by the reducing power of the activated magnesium. The addition of lithium chloride is critical, as it is known to enhance the reactivity of magnesium metal, potentially forming soluble organomagnesium species or modifying the surface of the magnesium to allow for efficient electron transfer at mild temperatures. This activation step ensures that the catalytic cycle proceeds smoothly without the need for harsh thermal conditions that could degrade sensitive substrates. The aryl quaternary ammonium salt serves as an excellent leaving group precursor, undergoing oxidative addition with the palladium center more readily than traditional halides in certain contexts, thus driving the catalytic turnover efficiently.
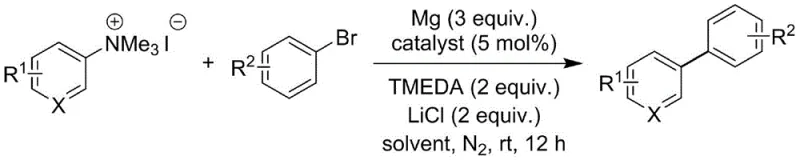
Furthermore, the system demonstrates exceptional chemoselectivity, which is paramount for the synthesis of complex pharmaceutical intermediates where multiple reactive sites may be present. The use of TMEDA (N,N,N',N'-tetramethylethylenediamine) as a ligand or additive further stabilizes the magnesium species and solubilizes the inorganic salts in the organic phase, creating a homogeneous reaction environment that promotes consistent kinetics. This careful balancing of reagents allows the reaction to tolerate a wide array of functional groups, including esters, nitriles, ethers, and even halogens, without competitive side reactions such as homocoupling or dehalogenation. The ability to control the impurity profile through precise modulation of the catalyst loading and reaction temperature provides R&D teams with a robust tool for optimizing purity specifications, ensuring that the final biaryl products meet the rigorous standards required for clinical grade materials.
How to Synthesize Biaryl Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural nuances to maximize yield and safety. The process begins with the careful activation of magnesium scraps, a step that is crucial for generating the necessary reducing equivalents without inducing exothermic runaways. Following activation, the reaction is conducted in ultra-dry tetrahydrofuran under an inert nitrogen atmosphere to prevent quenching of the active species by atmospheric moisture. The sequential addition of the aryl quaternary ammonium salt, the palladium catalyst, and the aryl bromide must be managed to maintain optimal concentration gradients that favor the cross-coupling pathway over competing decomposition routes. While the general procedure is straightforward, attention to detail regarding the stoichiometry of the magnesium and lithium chloride is essential for reproducibility. For a comprehensive breakdown of the standardized operating procedures, please refer to the detailed synthesis guide below.
- Activate magnesium scraps with lithium chloride under reduced pressure at 310-330°C to generate the active reducing species.
- Combine aryl quaternary ammonium salt, aryl bromide, Pd catalyst, and TMEDA in ultra-dry THF under nitrogen atmosphere.
- Stir the reaction mixture at 25-60°C for 6-12 hours, followed by aqueous workup and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this technology offers tangible benefits that extend beyond mere chemical efficiency, directly impacting the bottom line and supply chain resilience. The primary advantage lies in the substitution of expensive and unstable organometallic reagents with commodity chemicals that are readily available from multiple global suppliers. This shift reduces the risk of supply disruptions caused by the limited availability of specialized reagents and mitigates the price volatility associated with niche chemical markets. Additionally, the simplified workflow reduces the demand for specialized equipment capable of handling pyrophoric materials, allowing for the utilization of standard glass-lined reactors that are common in most multipurpose chemical plants. This compatibility with existing infrastructure accelerates the timeline for technology transfer and commercial scale-up, enabling faster time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the elimination of costly pre-activation steps and the reduction in waste disposal fees associated with heavy metal byproducts. By avoiding the use of stoichiometric amounts of toxic tin or zinc reagents often found in alternative coupling methods, the process significantly lowers the environmental compliance burden and the associated costs of wastewater treatment. The high atom economy and the ability to run reactions at near-room temperature also translate into substantial energy savings, as there is no need for prolonged heating or cryogenic cooling. These cumulative efficiencies result in a lower cost of goods sold (COGS), providing a competitive edge in pricing negotiations for bulk active pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: Stability is a key metric for supply chain continuity, and the use of shelf-stable aryl quaternary ammonium salts ensures that raw materials can be stocked in advance without degradation concerns. This inventory flexibility allows procurement managers to buffer against market fluctuations and secure raw materials during periods of low demand, ensuring uninterrupted production schedules. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, reducing the frequency of batch failures and the need for costly rework. This reliability fosters stronger partnerships between chemical manufacturers and their clients, as delivery timelines become more predictable and trustworthy.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges related to heat transfer and mixing, but the mild exothermic nature of this reductive coupling makes it inherently safer for large-volume production. The absence of hazardous reagents simplifies the safety assessment protocols and reduces the regulatory hurdles required for plant approval. Moreover, the green chemistry attributes of this method, such as low pollution and high efficiency, align perfectly with the sustainability goals of modern multinational corporations. By adopting this pathway, companies can demonstrate a commitment to environmental stewardship while simultaneously achieving operational excellence, satisfying both regulatory bodies and corporate social responsibility mandates.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific projects, we have compiled a set of frequently asked questions based on the detailed experimental data provided in the patent documentation. These inquiries address common concerns regarding catalyst selection, substrate scope, and process optimization, offering clarity on how this method compares to industry standards. Understanding these technical nuances is essential for R&D directors and process chemists who are tasked with selecting the most appropriate synthetic route for complex molecule assembly. The following answers are derived directly from the empirical results observed during the development of this patented methodology.
Q: Why are aryl quaternary ammonium salts preferred over boronic acids in this synthesis?
A: Aryl quaternary ammonium salts offer superior stability against air and moisture compared to organoboron reagents, eliminating the need for stringent anhydrous storage and simplifying the supply chain logistics for large-scale manufacturing.
Q: What is the role of Lithium Chloride in the magnesium activation step?
A: Lithium chloride acts as a crucial additive that facilitates the formation of highly reactive 'Rieke Magnesium' species at lower temperatures, ensuring efficient reduction of the palladium catalyst and consistent reaction initiation.
Q: Can this method tolerate sensitive functional groups like esters or nitriles?
A: Yes, the patent data demonstrates excellent functional group tolerance, successfully accommodating esters, nitriles, and halogens without side reactions, which is critical for synthesizing complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biaryl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercially viable manufacturing process requires deep technical expertise and state-of-the-art facilities. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN113402350B can be realized on an industrial scale. Our team of expert process chemists is well-versed in optimizing palladium-catalyzed reactions to meet stringent purity specifications, utilizing our rigorous QC labs to monitor every critical parameter from raw material intake to final product release. We are committed to delivering high-purity biaryl compounds that adhere to the highest quality standards, supporting your pipeline from early-stage development through to commercial launch.
We invite you to explore how this innovative synthesis route can optimize your supply chain and reduce overall project costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecular targets. We are prepared to provide specific COA data and route feasibility assessments to help you make informed decisions about your sourcing strategy. Let us collaborate to bring your next generation of pharmaceutical intermediates to market with speed, efficiency, and confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
