Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles: A Breakthrough for Commercial API Manufacturing
Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles: A Breakthrough for Commercial API Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN111978265B discloses a highly efficient preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives, addressing critical limitations in existing literature. This technology leverages a ferric chloride-promoted cyclization strategy that operates under mild, air-tolerant conditions, eliminating the need for stringent anhydrous or oxygen-free environments. The introduction of the trifluoromethyl group into the 1,2,4-triazole scaffold is particularly valuable, as it significantly enhances the electronegativity, metabolic stability, and lipophilicity of the resulting drug candidates. As illustrated in the structural diversity of known pharmaceuticals, this core motif is essential for next-generation therapeutics.
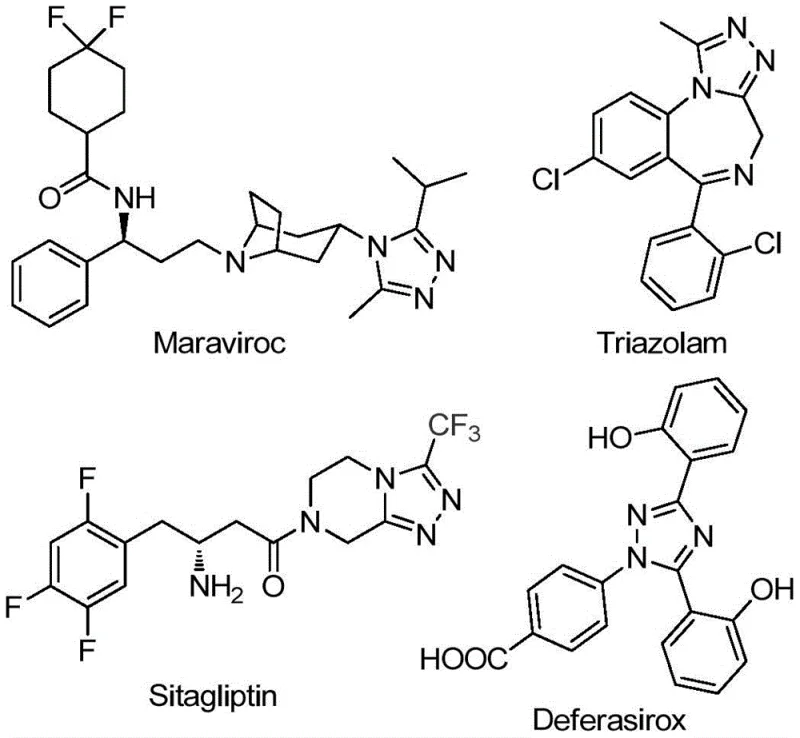
For R&D directors and process chemists, the ability to access these scaffolds reliably is paramount. The disclosed method not only simplifies the synthetic workflow but also expands the accessible chemical space by successfully incorporating alkyl hydrazones, a substrate class that has historically been problematic in similar transformations. This represents a significant leap forward in the cost reduction in pharmaceutical intermediate manufacturing, offering a pathway to high-purity compounds with reduced operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with challenges that hinder industrial adoption. Traditional protocols often rely on the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines. These legacy methods are frequently characterized by harsh reaction conditions, requiring extreme temperatures or pressures that pose safety risks and increase energy consumption. Furthermore, the substrate scope is notoriously narrow; specifically, previous tandem cyclization methods utilizing trifluoroethylimidoyl chlorides failed to react with alkyl hydrazones, severely limiting the structural diversity of accessible products. Such limitations result in lower reaction yields and necessitate complex purification steps to remove unreacted starting materials and side products, thereby inflating the overall cost of goods.
The Novel Approach
In stark contrast, the methodology described in patent CN111978265B introduces a streamlined, two-stage process that overcomes these historical bottlenecks. By utilizing cheap and readily available acyl hydrazides and trifluoroethylimide chlorides as starting materials, the process initiates with a base-promoted intermolecular reaction followed by a metal Lewis acid-promoted cyclization. The use of ferric chloride as a promoter is particularly innovative, enabling the reaction to proceed efficiently at moderate temperatures ranging from 40 to 80°C. This approach not only tolerates a wide range of functional groups but also successfully incorporates alkyl hydrazones, filling a critical gap in synthetic capability. The reaction scheme below highlights the simplicity and elegance of this transformation, showcasing the direct conversion of precursors into the desired triazole core.
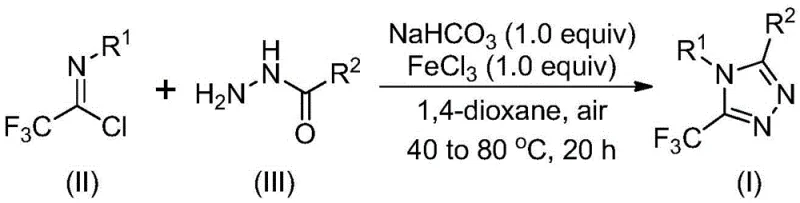
From a supply chain perspective, the reliance on commodity chemicals like sodium bicarbonate and ferric chloride ensures that raw material availability is never a bottleneck. The elimination of sensitive reagents means that the process can be executed in standard reactor vessels without specialized inert atmosphere equipment, drastically simplifying the engineering requirements for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
A deep understanding of the reaction mechanism is crucial for optimizing process parameters and ensuring consistent quality. The proposed mechanism involves an initial base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimide chloride and the hydrazide, generating a trifluoroacetamidine derivative intermediate. This step is facilitated by sodium bicarbonate, which acts as a mild base to scavenge the generated hydrochloric acid without degrading sensitive functional groups. Subsequently, the addition of the metal Lewis acid, specifically ferric chloride, triggers an intramolecular dehydration condensation. The Lewis acidity of the iron center activates the imine nitrogen, facilitating the nucleophilic attack by the adjacent hydrazine nitrogen and subsequent ring closure to form the 1,2,4-triazole system.
This mechanistic pathway offers distinct advantages regarding impurity control. The mild basic conditions in the first stage prevent the hydrolysis of the imidoyl chloride, a common side reaction in aqueous or strongly basic media. Furthermore, the specific activation by ferric chloride in the second stage ensures high regioselectivity, minimizing the formation of isomeric byproducts. The broad substrate tolerance is evidenced by the successful synthesis of various derivatives, including those with electron-donating methyl groups, electron-withdrawing chloro or trifluoromethyl groups, and bulky naphthyl substituents. The structural variety shown in the following examples underscores the versatility of this catalytic system for generating diverse chemical libraries.
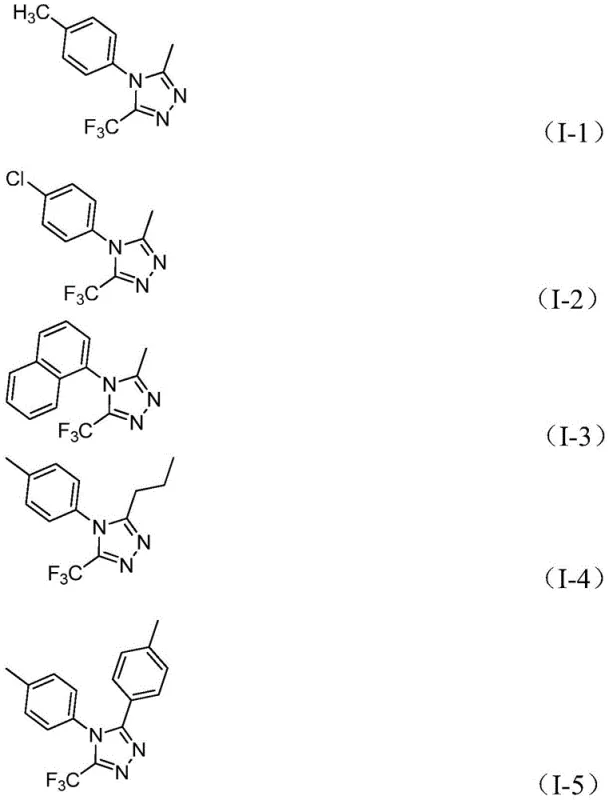
For quality assurance teams, the predictability of this mechanism means that impurity profiles are consistent and manageable. The absence of transition metals that are difficult to remove, combined with the use of iron which is generally less regulated than precious metals like palladium or platinum, simplifies the downstream purification process. This results in high-purity 1,2,4-triazole derivatives that meet the stringent specifications required for active pharmaceutical ingredient (API) production.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Derivatives Efficiently
The operational simplicity of this synthesis makes it an ideal candidate for technology transfer from the laboratory to the pilot plant. The procedure involves a sequential addition of reagents in a common aprotic solvent, followed by a temperature ramp to drive the cyclization to completion. Detailed standard operating procedures regarding stoichiometry, solvent selection, and workup protocols are critical for maximizing yield and purity. For a comprehensive guide on executing this synthesis with optimal efficiency, please refer to the standardized protocol outlined below.
- Mix sodium bicarbonate, trifluoroethylimide chloride, and hydrazide in an aprotic organic solvent such as 1,4-dioxane.
- Stir the mixture at 30-50°C for 8-16 hours to facilitate base-promoted intermolecular carbon-nitrogen bond formation.
- Add ferric chloride catalyst and heat to 70-90°C for 6-10 hours to complete the Lewis acid-promoted cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this new synthetic route are profound. The shift from exotic, sensitive reagents to commodity chemicals fundamentally alters the cost structure of producing these valuable intermediates. By removing the dependency on strict anhydrous conditions and expensive catalysts, the process inherently reduces the operational expenditure associated with solvent drying, inert gas purging, and specialized reactor maintenance. This translates directly into substantial cost savings in fine chemical manufacturing, allowing for more competitive pricing in the global market.
- Cost Reduction in Manufacturing: The utilization of ferric chloride and sodium bicarbonate represents a drastic reduction in catalyst and reagent costs compared to precious metal systems. Since these materials are abundant and inexpensive, the raw material cost per kilogram of product is significantly lowered. Furthermore, the simplified workup procedure, which often requires only filtration and standard column chromatography, reduces the consumption of silica gel and solvents during purification. This lean manufacturing approach ensures that the overall production cost is minimized without compromising on the quality of the final output.
- Enhanced Supply Chain Reliability: One of the greatest risks in chemical supply chains is the reliance on single-source or hard-to-source reagents. This method mitigates that risk by utilizing starting materials that are widely available from multiple global suppliers. The robustness of the reaction conditions means that production is less susceptible to delays caused by environmental factors or equipment failures related to moisture sensitivity. Consequently, lead times for high-purity pharmaceutical intermediates can be reduced, ensuring a steady flow of materials to downstream API manufacturers and preventing production stoppages.
- Scalability and Environmental Compliance: The ability to scale this reaction from gram levels to multi-ton production is a key advantage for long-term supply security. The use of 1,4-dioxane or other common solvents allows for established recovery and recycling protocols, aligning with modern green chemistry principles. Additionally, the avoidance of heavy metal contaminants simplifies waste treatment and disposal, reducing the environmental footprint of the manufacturing process. This compliance with environmental regulations is increasingly critical for maintaining operational licenses and meeting the sustainability goals of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for their specific projects. The following questions address common inquiries regarding the scope, scalability, and practical application of this patented technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a reliable basis for decision-making.
Q: What are the primary advantages of this FeCl3-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods that require harsh conditions or fail with alkyl hydrazones, this method operates under mild, air-tolerant conditions with broad substrate scope and high yields.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the process utilizes cheap, commercially available reagents and simple workup procedures like filtration, making it highly amenable to commercial scale-up.
Q: What types of substituents are tolerated on the triazole ring?
A: The method demonstrates excellent functional group tolerance, accommodating various aryl groups with electron-donating or withdrawing substituents, as well as alkyl and alkenyl chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in the development of novel therapeutics. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement advanced catalytic methods like the FeCl3-promoted cyclization allows us to offer cost-effective solutions for complex molecule construction.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug discovery and development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline to market while optimizing your budget.
