Advanced Electrochemical Synthesis of Selenium-Containing Thiazoline Compounds for Pharmaceutical Applications
The rapid evolution of medicinal chemistry has increasingly highlighted the critical role of selenium-containing heterocycles in drug discovery, particularly for their potent anticancer and antiviral properties. As detailed in the groundbreaking patent CN111235596A, a novel electrochemical methodology has been established for the efficient preparation of selenium-containing thiazoline compounds, addressing the urgent need for greener and more sustainable synthetic routes in the fine chemical industry. This technology leverages constant current electrolysis to drive the cyclization of thioamides with diselenide compounds, bypassing the need for harsh chemical oxidants or extreme thermal conditions that have historically plagued this sector. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this innovation represents a paradigm shift towards safer, more cost-effective manufacturing processes that align with modern environmental standards. The ability to synthesize these complex scaffolds under mild conditions not only enhances the purity profile of the final product but also streamlines the downstream purification steps, offering substantial value to the global supply chain of active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiazoline derivatives has relied heavily on methodologies that impose significant operational burdens and safety risks on chemical manufacturing facilities. Traditional protocols, such as those utilizing N-bromosuccinimide (NBS) in chloroform at elevated temperatures, necessitate the handling of toxic halogenated solvents and strong oxidizing agents, which complicate waste disposal and increase the overall carbon footprint of the production process. Furthermore, alternative thermal approaches often require multi-step sequences involving high-temperature heating, which can lead to thermal degradation of sensitive functional groups and result in lower overall yields due to the formation of complex impurity profiles. These legacy methods frequently demand rigorous safety protocols and specialized equipment to manage exothermic reactions, thereby inflating the capital expenditure required for setting up production lines. The reliance on stoichiometric amounts of chemical oxidants also generates substantial amounts of salt waste, creating a bottleneck for companies aiming to achieve green chemistry certification and reduce their environmental liability in the competitive landscape of agrochemical intermediate and pharmaceutical manufacturing.
The Novel Approach
In stark contrast to these archaic techniques, the electrochemical strategy disclosed in the patent introduces a streamlined, single-step protocol that operates efficiently at room temperature using electricity as the primary driving force for the redox reaction. By employing graphite felt electrodes and a simple electrolyte system comprising lithium perchlorate in acetonitrile, this method eliminates the need for external chemical oxidants, thereby drastically simplifying the reaction workup and minimizing the generation of hazardous byproducts. The core reaction involves the anodic activation of the diselenide species, which subsequently reacts with the thioamide substrate to form the desired thiazoline ring with high regioselectivity and atom economy. This approach not only reduces the reaction time to approximately 4 hours under a constant current of 2 mA but also ensures that the process remains robust across a wide range of substrate variations, including those with electron-withdrawing or electron-donating groups. 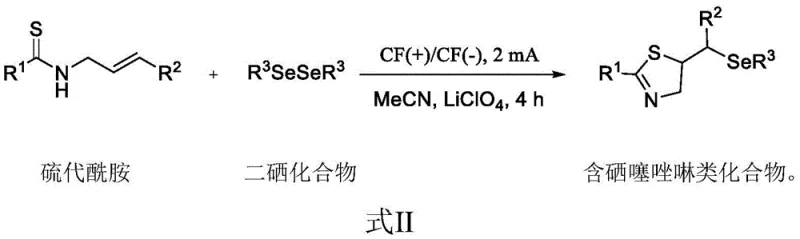
Mechanistic Insights into Electrochemical Selenylation and Cyclization
The mechanistic pathway of this transformation is rooted in the precise control of electron transfer at the electrode surface, which facilitates the generation of reactive selenyl species without the over-oxidation issues common in chemical methods. Upon application of the constant current, the diselenide bond undergoes homolytic or heterolytic cleavage at the anode to generate electrophilic selenium intermediates, which are immediately captured by the nucleophilic sulfur atom of the thioamide. This initial interaction triggers an intramolecular cyclization cascade where the nitrogen atom attacks the activated alkene moiety, closing the five-membered thiazoline ring with exceptional stereochemical fidelity. The use of a divided or undivided cell configuration with inert graphite electrodes ensures that the cathodic reduction processes do not interfere with the anodic oxidation, maintaining a stable reaction environment that prevents the decomposition of the sensitive organoselenium products. This controlled electrochemical environment is crucial for maintaining the integrity of the selenium-carbon bond, which is often susceptible to cleavage under the harsh acidic or basic conditions employed in traditional synthetic routes.
From an impurity control perspective, the mildness of the electrochemical conditions plays a pivotal role in suppressing side reactions such as polymerization of the alkene substrate or oxidative degradation of the thioamide functionality. Since the reaction potential is carefully regulated by the constant current mode, the system avoids the high-energy states that typically lead to non-selective radical coupling or over-oxidation to selenones or selenoxides. Consequently, the crude reaction mixture contains significantly fewer byproducts compared to thermal methods, allowing for a more efficient purification process via standard silica gel column chromatography. This high level of selectivity is particularly advantageous for the production of high-purity OLED material precursors or pharmaceutical intermediates where trace metal contamination or organic impurities can severely impact the efficacy and safety of the final drug product. The ability to tune the reaction parameters, such as current density and electrolyte concentration, provides process chemists with a powerful toolkit for optimizing the quality of the output without altering the fundamental reagent stoichiometry.
How to Synthesize Selenium-Containing Thiazoline Efficiently
The practical implementation of this synthesis route is designed to be accessible for both laboratory-scale optimization and pilot-plant operations, requiring only standard electrochemical equipment and commercially available reagents. The procedure begins with the dissolution of the thioamide and diselenide substrates in acetonitrile, followed by the addition of the supporting electrolyte to ensure sufficient conductivity for the electrochemical cell. The reaction is then initiated by applying a constant current, during which the progress can be monitored via thin-layer chromatography or HPLC to determine the optimal endpoint, typically achieved within 4 hours for most substrates. 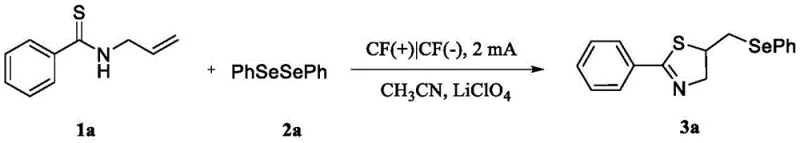
- Prepare the reaction mixture by adding thioamide and diselenide compound in a molar ratio of 1: 0.6 into a reactor containing acetonitrile solvent and lithium perchlorate electrolyte.
- Utilize graphite felt electrodes as both cathode and anode, applying a constant current of 2 mA at room temperature while stirring for 4 hours.
- Upon completion, remove the solvent via rotary evaporation and purify the crude product using silica gel flash column chromatography to isolate the pure target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology offers a compelling value proposition centered around cost efficiency, supply security, and regulatory compliance. By removing the dependency on expensive transition metal catalysts and stoichiometric oxidants, the direct material costs associated with the synthesis are significantly reduced, while the simplified workup procedure lowers the consumption of solvents and stationary phases during purification. This streamlined process translates into shorter batch cycles and higher throughput capacity, enabling manufacturers to respond more agilely to market demands and reduce the lead time for high-purity pharmaceutical intermediates. Furthermore, the elimination of toxic reagents and the use of benign solvents align perfectly with increasingly stringent environmental regulations, mitigating the risk of production shutdowns due to compliance issues and enhancing the long-term sustainability of the supply chain.
- Cost Reduction in Manufacturing: The economic benefits of this method are derived primarily from the substitution of costly chemical oxidants with electricity, which serves as a clean and inexpensive reagent for driving the redox transformation. Additionally, the absence of heavy metal catalysts removes the need for expensive and time-consuming metal scavenging steps, which are often required to meet the strict residual metal limits imposed by pharmacopeial standards. The high atom economy of the reaction ensures that a larger proportion of the starting materials are converted into the desired product, minimizing waste disposal costs and maximizing the return on investment for raw material procurement. Overall, the operational simplicity allows for a reduction in labor hours and utility consumption, contributing to a leaner and more profitable manufacturing model for complex organic synthesis.
- Enhanced Supply Chain Reliability: The robustness of the electrochemical protocol ensures consistent product quality across different batches, which is critical for maintaining uninterrupted supply lines to downstream pharmaceutical customers. Since the starting materials, such as thioamides and diselenides, are readily available from multiple commercial sources, the risk of supply disruption due to raw material scarcity is minimized. The mild reaction conditions also reduce the wear and tear on reactor equipment, extending the lifespan of capital assets and decreasing the frequency of maintenance-related downtime. This reliability fosters stronger partnerships between chemical suppliers and their clients, as it guarantees the timely delivery of critical intermediates needed for drug development and commercial production schedules.
- Scalability and Environmental Compliance: Scaling electrochemical reactions is inherently straightforward, as the process can be intensified by increasing the electrode surface area or using flow chemistry setups, facilitating the transition from gram-scale research to ton-scale commercial production. The green nature of the process, characterized by the absence of hazardous waste streams and the use of recyclable solvents, positions manufacturers favorably in the context of global sustainability initiatives and carbon reduction targets. This environmental stewardship not only enhances the corporate reputation of the supplier but also future-proofs the production process against evolving regulatory landscapes that increasingly penalize polluting manufacturing practices. Consequently, this technology represents a strategic asset for companies aiming to lead the market in sustainable chemical innovation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of selenium-containing thiazolines, providing clarity on the feasibility and benefits of implementing this technology in industrial settings. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring that the information provided is accurate and relevant for decision-makers. Understanding these aspects is essential for evaluating the potential integration of this method into existing production workflows and for assessing its compatibility with specific project requirements.
Q: What are the primary advantages of this electrochemical method over traditional thermal synthesis?
A: Unlike conventional methods requiring high temperatures and toxic solvents like chloroform, this electrochemical approach operates at room temperature with mild conditions, significantly reducing energy consumption and environmental hazards while simplifying the operational workflow.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the use of constant current electrolysis with robust graphite felt electrodes and commercially available electrolytes suggests high scalability, making it suitable for the commercial scale-up of complex pharmaceutical intermediates without the need for expensive transition metal catalysts.
Q: What is the substrate scope for the R1 and R3 groups in this reaction?
A: The method demonstrates excellent tolerance for various substituents, including phenyl, substituted phenyl (with chloro, bromo, trifluoromethyl, methoxy, or methyl groups), and heteroaryl groups like 2-thienyl, allowing for the synthesis of diverse high-purity organoselenides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selenium-Containing Thiazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this electrochemical technology in advancing the synthesis of high-value selenium-containing heterocycles for the global pharmaceutical and fine chemical markets. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality and volume regardless of their project stage. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of selenium-containing thiazoline delivered meets the highest industry standards for safety and efficacy. We are committed to leveraging our technical expertise to optimize this green synthesis route, providing our partners with a competitive edge through superior process efficiency and environmental responsibility.
We invite you to collaborate with us to explore the full capabilities of this innovative manufacturing platform and to discuss how it can be tailored to meet your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis and to obtain specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a reliable supply chain that prioritizes innovation, quality, and sustainability, empowering your organization to bring life-saving therapies to market faster and more efficiently.
