Revolutionizing Selenium-Containing Thiazoline Production via Green Electrochemical Cyclization for Global Pharma
Revolutionizing Selenium-Containing Thiazoline Production via Green Electrochemical Cyclization for Global Pharma
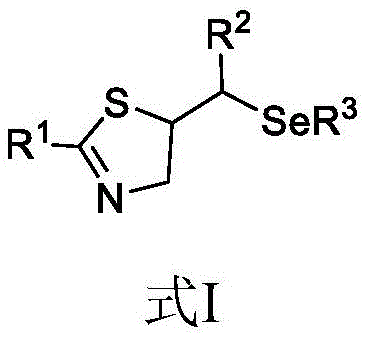
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to access complex heterocyclic scaffolds. A significant breakthrough in this domain is documented in patent CN111235596A, which discloses a novel preparation method for selenium-containing thiazoline compounds. These molecules are of paramount importance due to their diverse biological activities, ranging from anticancer and anti-HIV properties to potent antiviral effects against herpes viruses. The integration of selenium into the thiazoline core enhances pharmacological value, yet traditional synthetic routes have often been plagued by harsh conditions and environmental concerns. This new electrochemical methodology represents a paradigm shift, utilizing electricity as a traceless reagent to drive the cyclization under exceptionally mild conditions. By leveraging constant current electrolysis, this technology offers a robust platform for generating high-purity intermediates, addressing the critical needs of R&D directors focused on impurity control and supply chain managers concerned with scalability and safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiazoline derivatives has relied on methodologies that impose significant burdens on both operational safety and environmental compliance. For instance, prior art from the Pan research group utilized N-bromosuccinimide (NBS) as an oxidant in chloroform solvent at elevated temperatures, introducing toxic halogenated solvents and stoichiometric chemical waste into the process stream. Similarly, approaches developed by the Li group required two-step reactions at 80°C, increasing energy consumption and extending production lead times. Other methods, such as those by the Cazin group, also necessitated heating to 80°C using nitriles and cysteamine hydrochloride. These conventional thermal processes often suffer from poor atom economy, the generation of hazardous byproducts, and the need for rigorous downstream purification to remove residual heavy metals or halogenated impurities. Such factors drastically increase the cost of goods sold (COGS) and complicate the regulatory approval process for active pharmaceutical ingredients (APIs) derived from these intermediates.
The Novel Approach
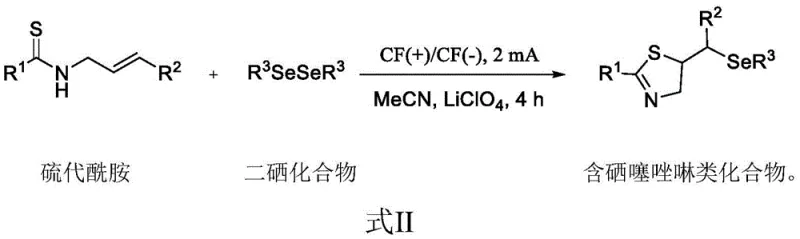
In stark contrast, the invention detailed in patent CN111235596A introduces a streamlined electrochemical protocol that eliminates the need for external chemical oxidants and high-temperature heating. As illustrated in the reaction scheme below, the process involves the direct electrolysis of thioamides and diselenide compounds in acetonitrile using lithium perchlorate as the supporting electrolyte. The reaction proceeds smoothly at room temperature under a constant current of 2 mA, completing within just 4 hours. This transition from thermal/chemical activation to electrochemical activation not only simplifies the operational workflow but also inherently improves the green chemistry profile of the synthesis. By avoiding toxic oxidants like NBS and corrosive acidic conditions, the new method reduces the burden on waste treatment facilities and enhances the overall safety of the manufacturing environment, making it an ideal candidate for modern, sustainable chemical production.
Mechanistic Insights into Electrochemical Oxidative Cyclization
The core innovation of this technology lies in its unique mechanistic pathway, which leverages anodic oxidation to generate reactive selenium species in situ. Under the applied constant current, the diselenide bond undergoes homolytic or heterolytic cleavage at the anode surface, generating electrophilic selenium intermediates or radicals. These activated species then attack the electron-rich alkene moiety of the N-allyl thioamide substrate, initiating a cascade of intramolecular cyclization events. The sulfur atom of the thioamide subsequently participates in the ring closure, forming the stable thiazoline heterocycle while incorporating the selenium functionality at the desired position. This electron-transfer mediated mechanism bypasses the high energy barriers associated with thermal activation, allowing the reaction to proceed efficiently at ambient temperatures. Furthermore, the use of graphite felt electrodes provides a large surface area for electron transfer, ensuring consistent reaction kinetics and minimizing the formation of over-oxidized byproducts that often plague chemical oxidant-based methods.
From an impurity control perspective, this electrochemical approach offers distinct advantages for producing high-purity pharmaceutical intermediates. Traditional methods involving strong chemical oxidants often lead to non-selective oxidation of sensitive functional groups on the aromatic rings, such as methoxy or methyl substituents, resulting in complex impurity profiles that are difficult to separate. In this electrochemical system, the potential is controlled by the current density, providing a level of selectivity that chemical reagents cannot match. The mild conditions prevent the degradation of the thiazoline ring and preserve the integrity of the selenium-carbon bond, which is crucial for maintaining the biological activity of the final molecule. Consequently, the crude reaction mixture is significantly cleaner, reducing the load on downstream purification steps like silica gel chromatography and enabling the isolation of products with superior purity specifications required for clinical applications.
How to Synthesize Selenium-Containing Thiazoline Efficiently
The practical implementation of this synthesis route is designed for ease of adoption in both laboratory and pilot-scale settings. The procedure begins with the precise weighing of the thioamide substrate and the diselenide reagent, typically in a molar ratio of 1:0.6, ensuring optimal stoichiometry for the cyclization. These reagents are dissolved in acetonitrile along with lithium perchlorate, which serves as the conductive medium for the electrochemical cell. The reaction vessel is equipped with graphite felt electrodes, chosen for their chemical inertness and conductivity, and the system is subjected to a constant current of 2 mA. The entire process is conducted at room temperature, eliminating the need for energy-intensive heating mantles or cooling baths. After the designated reaction time of 4 hours, the solvent is removed under reduced pressure, and the residue is purified via standard flash column chromatography.
- Prepare the reaction mixture by adding thioamide and diselenide compound in a 1: 0.6 molar ratio into a reactor with acetonitrile solvent and lithium perchlorate electrolyte.
- Utilize graphite felt electrodes as both cathode and anode, applying a constant current of 2 mA at room temperature for 4 hours to drive the cyclization.
- Upon completion, remove the solvent via rotary evaporation and purify the crude product using silica gel flash column chromatography to obtain the pure target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology translates into tangible strategic benefits beyond mere technical feasibility. The elimination of expensive and hazardous chemical oxidants like NBS directly impacts the raw material cost structure, removing the volatility associated with specialty reagent pricing. Furthermore, the shift to room temperature operations significantly reduces utility costs related to heating and cooling, contributing to a lower overall carbon footprint for the manufacturing site. The simplicity of the workup procedure, involving basic solvent evaporation and chromatography, shortens the batch cycle time, thereby enhancing the throughput capacity of existing production facilities without the need for major capital investment in new reactors. This efficiency gain is critical for meeting tight delivery schedules in the fast-paced pharmaceutical supply chain.
- Cost Reduction in Manufacturing: The removal of stoichiometric chemical oxidants and the reduction in energy consumption through ambient temperature processing lead to substantial cost savings. By replacing expensive reagents with electricity, which is generally cheaper and more stable in price, the variable cost per kilogram of the intermediate is significantly optimized. Additionally, the simplified purification process reduces the consumption of silica gel and organic solvents, further driving down the operational expenditure associated with waste disposal and material handling.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically thioamides and diselenides, are commercially available and stable, reducing the risk of supply disruptions. The robustness of the electrochemical method means that the process is less sensitive to minor fluctuations in reaction parameters, ensuring consistent batch-to-bquality. This reliability allows supply chain planners to forecast production timelines with greater accuracy, minimizing the need for safety stock and reducing inventory holding costs for high-value intermediates.
- Scalability and Environmental Compliance: Scaling electrochemical reactions is increasingly straightforward with modern flow chemistry technologies, allowing for seamless transition from gram-scale R&D to multi-ton commercial production. The green nature of this process, characterized by the absence of heavy metal catalysts and toxic halogenated solvents, ensures compliance with stringent environmental regulations such as REACH and EPA guidelines. This compliance mitigates the risk of regulatory fines and facilitates smoother audits from global pharmaceutical clients who prioritize sustainable sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of selenium-containing thiazolines. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this electrochemical method over traditional thermal synthesis?
A: Unlike conventional methods requiring high temperatures (80°C+) and toxic oxidants like NBS, this electrochemical approach operates at room temperature with mild conditions, significantly reducing energy consumption and hazardous waste generation while simplifying operational complexity.
Q: How does this process impact the purity profile of the final pharmaceutical intermediate?
A: The mild reaction conditions and the use of electricity as a clean reagent minimize side reactions and thermal degradation, resulting in a cleaner crude reaction mixture that facilitates easier purification and higher final purity specifications essential for API manufacturing.
Q: Is this synthesis method scalable for industrial production of organoselenides?
A: Yes, the process utilizes simple graphite felt electrodes and standard electrolytes without expensive transition metal catalysts, making it highly adaptable for commercial scale-up with reduced equipment corrosion risks and lower overall manufacturing costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selenium-Containing Thiazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN111235596A can be successfully translated into industrial reality. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications for complex organoselenium compounds. We are committed to delivering high-purity selenium-containing thiazolines that meet the exacting standards of the global pharmaceutical industry, supporting your drug development programs with reliable and compliant supply chains.
We invite you to collaborate with us to explore the full commercial potential of this green synthesis technology. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data for our selenium-containing thiazoline portfolio and to discuss route feasibility assessments for your custom synthesis projects. Together, we can accelerate the development of next-generation therapeutics while optimizing your manufacturing costs and environmental impact.
