Advanced Biphenyl Synthesis: Magnetic Catalyst Technology for Commercial Scale-Up
The chemical industry is currently witnessing a paradigm shift towards sustainable and economically viable catalytic systems, particularly in the synthesis of critical intermediates like biphenyl compounds. Patent CN112939716A, published in June 2021, introduces a groundbreaking preparation method that leverages functionalized magnetic complexes to catalyze the Suzuki-Miyaura coupling reaction. This technology addresses the longstanding challenges associated with traditional noble metal catalysis by utilizing robust, magnetically separable catalysts based on earth-abundant metals such as Nickel, Zinc, Iron, and Tin. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this innovation represents a significant leap forward in process efficiency. The patent details a versatile system where the catalyst, structured as a core-shell magnetic nanoparticle functionalized with specific ligands, facilitates the coupling of brominated aromatic hydrocarbons with arylboronic acids under mild conditions. This approach not only ensures high product yields exceeding 90% in optimized examples but also guarantees exceptional purity levels, often reaching 99.8%, which is paramount for downstream pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of biphenyl derivatives has heavily relied on homogeneous catalysis using precious metals, primarily Palladium. While effective, these conventional methods suffer from severe economic and operational drawbacks that hinder large-scale manufacturing. The primary issue lies in the exorbitant cost of Palladium catalysts, which significantly inflates the raw material expenses for high-volume production runs. Moreover, the homogeneous nature of these catalysts makes separation from the reaction mixture notoriously difficult, often requiring complex purification steps such as column chromatography or extensive washing to meet stringent heavy metal residue limits imposed by regulatory bodies. These additional processing steps not only extend the production lead time but also generate substantial amounts of hazardous waste, complicating environmental compliance. Furthermore, traditional catalysts often lack thermal stability and reusability, leading to single-use scenarios that are neither economically nor environmentally sustainable for a cost reduction in API manufacturing strategy.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a heterogeneous functionalized magnetic complex that fundamentally alters the reaction landscape. By employing a catalyst system such as $NiFe_2O_4@SiO_2@Cu$ or $SnFe_2O_4@SiO_2@Sm$, the process achieves high catalytic efficiency while enabling effortless separation. Upon completion of the reaction, the solid catalyst particles can be instantly retrieved from the liquid phase using a simple external magnetic field, eliminating the need for filtration or centrifugation. This magnetic separation capability drastically simplifies the work-up procedure and ensures that the final product is virtually free from metal contamination. The reaction operates effectively in a green solvent system comprising ethanol and water, further enhancing the environmental profile of the process. As illustrated in the reaction scheme below, the coupling of bromobenzene and phenylboronic acid proceeds smoothly to form biphenyl, demonstrating the robustness of this new catalytic platform.
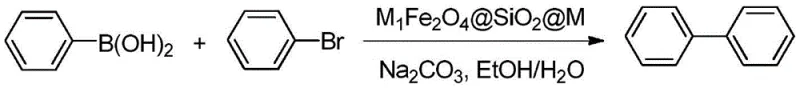
Mechanistic Insights into Functionalized Magnetic Complex Catalysis
The efficacy of this synthesis route lies in the sophisticated architecture of the catalyst, which is designed to maximize active site exposure while ensuring magnetic retrievability. The catalyst consists of a magnetic core, typically composed of ferrites like $M_1Fe_2O_4$ (where M1 is Ni, Zn, Fe, or Sn), encapsulated within a silica ($SiO_2$) shell. This silica layer serves a dual purpose: it protects the magnetic core from acidic degradation during the reaction and provides a stable surface for the grafting of organic ligands. These ligands, often Schiff base complexes coordinating with metals like Copper, Lanthanum, or Samarium, create the active catalytic centers necessary for the oxidative addition and reductive elimination steps of the Suzuki cycle. The structural integrity of this core-shell design is crucial, as it prevents the leaching of magnetic materials into the product stream, thereby maintaining high purity standards essential for a high-purity OLED material or pharmaceutical intermediate.
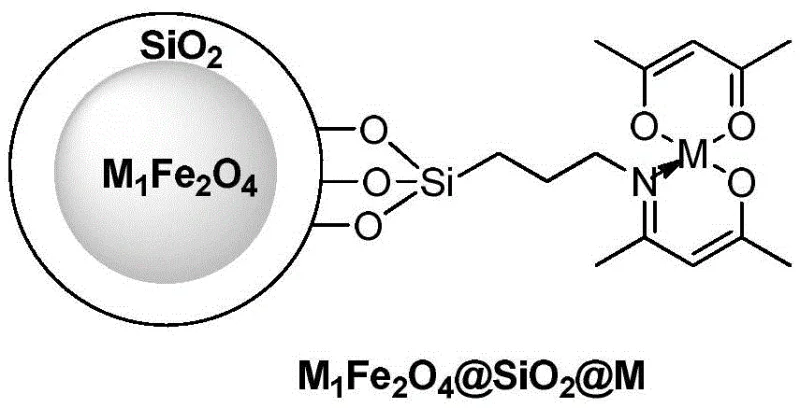
From an impurity control perspective, the heterogeneous nature of the catalyst plays a pivotal role in minimizing side reactions. Unlike homogeneous catalysts that may promote unwanted homocoupling of the boronic acid or dehalogenation of the substrate, the surface-confined active sites on the magnetic nanoparticles offer better selectivity. The steric environment created by the grafted ligands on the silica surface can be tuned to favor the cross-coupling pathway. Additionally, the use of sodium carbonate as a mild base in an ethanol-water mixture creates a buffered environment that suppresses the formation of phenolic byproducts. The patent data indicates that even after multiple recycling runs, the catalyst maintains its structural stability up to 260°C, ensuring that the impurity profile remains consistent batch after batch. This consistency is vital for commercial scale-up of complex polymer additives or fine chemicals where batch-to-batch variability can lead to costly rejection of materials.
How to Synthesize Biphenyl Compounds Efficiently
The synthesis protocol outlined in the patent offers a straightforward and scalable route for producing biphenyl derivatives, making it highly attractive for industrial adoption. The process begins by charging a reactor with the requisite molar ratios of brominated aromatic hydrocarbon and arylboronic acid, typically ranging from 1:1 to 1:1.6, along with sodium carbonate as the base. The choice of solvent is critical, with an ethanol-to-water volume ratio of 3:1 to 8:1 proving optimal for solubilizing reactants while maintaining catalyst suspension. Once the functionalized magnetic complex catalyst is added at a loading of 1-25% relative to the boronic acid mass, the mixture is heated to a moderate temperature of 40-80°C. The detailed standardized synthetic steps, including specific work-up procedures and purification techniques to achieve >99% purity, are provided in the technical guide below.
- Charge the reactor with brominated aromatic hydrocarbon, arylboronic acid, sodium carbonate, and ethanol-water mixed solvent.
- Add the functionalized magnetic complex catalyst (e.g., NiFe2O4@SiO2@Cu) and stir the mixture at 25-80°C for 1-8 hours.
- Separate the catalyst using an external magnetic field, recover the solvent, and recrystallize the crude product with ethanol to obtain pure biphenyl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this magnetic catalysis technology offers profound strategic benefits that extend beyond mere technical performance. The most immediate impact is seen in the drastic simplification of the supply chain for catalysts; shifting from scarce, geopolitically sensitive noble metals like Palladium to abundant base metals like Iron and Nickel mitigates supply risk and price volatility. This substitution results in substantial cost savings on raw materials, allowing for more competitive pricing of the final biphenyl intermediates. Furthermore, the elimination of complex filtration and heavy metal scavenging steps reduces the consumption of auxiliary chemicals and processing time, directly lowering the operational expenditure per kilogram of product. The ability to recycle the catalyst for multiple batches without regeneration further amplifies these economic advantages, creating a leaner and more efficient manufacturing process.
- Cost Reduction in Manufacturing: The replacement of expensive Palladium catalysts with functionalized magnetic complexes based on Nickel, Zinc, or Iron leads to a significant decrease in catalyst procurement costs. Since the catalyst can be magnetically separated and reused for at least 8 cycles without loss of activity, the effective cost per batch is drastically reduced compared to single-use homogeneous systems. Additionally, the simplified downstream processing eliminates the need for expensive metal scavengers and reduces solvent usage, contributing to overall lower production costs.
- Enhanced Supply Chain Reliability: Relying on earth-abundant metals for catalysis insulates the production process from the supply chain disruptions often associated with precious metals. The raw materials required for the catalyst synthesis, such as iron salts and silica precursors, are globally available and commoditized, ensuring a stable and continuous supply. This reliability is crucial for maintaining uninterrupted production schedules for critical pharmaceutical intermediates and agrochemical building blocks, reducing the risk of stockouts.
- Scalability and Environmental Compliance: The use of an ethanol-water solvent system aligns perfectly with green chemistry principles, reducing the environmental footprint of the manufacturing process. The heterogeneous nature of the reaction minimizes waste generation, and the magnetic separation technique avoids the creation of filter cakes laden with heavy metals. This ease of waste management facilitates smoother regulatory approvals and supports the commercial scale-up of complex fine chemicals in facilities with strict environmental discharge limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this magnetic catalysis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific product portfolios.
Q: What are the advantages of using magnetic complex catalysts over traditional Palladium catalysts?
A: Magnetic complex catalysts offer significant cost reductions as they utilize base metals like Nickel, Zinc, or Iron instead of expensive noble metals like Palladium. Furthermore, their heterogeneous nature allows for easy separation via an external magnetic field, eliminating complex filtration steps and reducing heavy metal residues in the final API intermediate.
Q: Can the functionalized magnetic catalyst be recycled for multiple batches?
A: Yes, the patent data demonstrates excellent catalyst stability. The functionalized magnetic complex can be recovered and reused for at least 8 consecutive cycles without significant loss of catalytic activity, maintaining yields between 85-92% and purity above 98% throughout the recycling process.
Q: What represent the optimal reaction conditions for this Suzuki coupling process?
A: The optimal conditions involve reacting brominated aromatics with arylboronic acids in an ethanol-water mixed solvent (volume ratio 3-8:1) using sodium carbonate as the base. The reaction typically proceeds efficiently at temperatures between 40-80°C for 2-5 hours, providing a balance between reaction rate and energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the magnetic complex-catalyzed Suzuki coupling technology described in patent CN112939716A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory-scale method into robust, industrial-grade processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications demonstrated in the patent are maintained at full manufacturing scale. We operate rigorous QC labs equipped with advanced analytical instruments to verify the absence of heavy metal residues and to guarantee the stringent purity specifications required by global pharmaceutical and electronic material clients.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your biphenyl derivative needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this technology can optimize your bill of materials. Please contact us today to request specific COA data for our pilot batches and to discuss route feasibility assessments for your target molecules, ensuring a secure and efficient supply chain for your critical intermediates.
