Advanced Synthesis of Vitamin E Intermediates Using Novel Lewis Acid Catalysis for Commercial Scale
Advanced Synthesis of Vitamin E Intermediates Using Novel Lewis Acid Catalysis for Commercial Scale
The global demand for high-purity vitamins and nutraceuticals continues to drive innovation in synthetic chemistry, particularly for complex molecules like Vitamin E (tocopherol). Patent CN1449393A introduces a groundbreaking methodology for the preparation of novel beta-olefinic compounds that serve as critical intermediates in the synthesis of phytone and ultimately Vitamin E. This technology represents a significant departure from traditional condensation methods, leveraging specific Lewis acid catalysts to achieve superior regioselectivity and yield under remarkably mild conditions. For R&D directors and procurement specialists, understanding this mechanistic shift is crucial for optimizing supply chains and reducing the cost of goods sold (COGS) in vitamin manufacturing. The patent details a versatile route that accommodates various hydrocarbon groups, allowing for the precise construction of the side chain required for tocopherol activity.
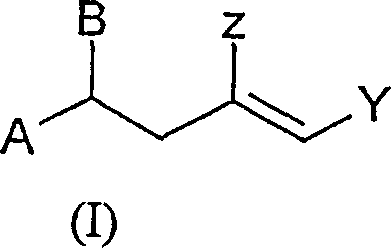
The core innovation lies in the structural flexibility of the general formula (I) compounds, where substituents A, Y, and Z can be tailored to specific downstream requirements. By utilizing a catalytic system involving metal halides or triflates, the process avoids the harsh conditions often associated with older synthetic routes, such as extreme temperatures or stoichiometric amounts of hazardous reagents. This not only improves the safety profile of the manufacturing facility but also significantly reduces the formation of unwanted by-products, thereby simplifying the purification workflow. As a reliable vitamin E intermediate supplier, recognizing the value of such patented methodologies allows for the strategic positioning of high-quality raw materials that align with modern green chemistry principles and regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Vitamin E intermediates has relied on condensation reactions that often suffer from poor atom economy and limited control over stereochemistry. Traditional methods frequently require stoichiometric amounts of strong acids or bases, leading to significant waste generation and challenging downstream processing. Furthermore, conventional routes often struggle with the coupling of bulky side chains, resulting in low yields and the formation of difficult-to-separate isomers. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for procurement managers tasked with maintaining consistent inventory levels. The reliance on multi-step sequences with low overall convergence further exacerbates these issues, making the supply chain vulnerable to disruptions at any single stage of the synthesis.
The Novel Approach
In contrast, the approach detailed in CN1449393A utilizes a direct catalytic condensation between a specific olefinic compound and an acetal or imine precursor. This method dramatically streamlines the synthetic pathway by reducing the number of discrete unit operations required to reach the target intermediate. The use of Lewis acids such as Ferric Chloride or rare earth triflates allows the reaction to proceed at temperatures ranging from -50°C to +25°C, which is a substantial improvement over the high-temperature conditions of legacy processes. This mild thermal profile preserves the integrity of sensitive functional groups and minimizes thermal degradation, ensuring a cleaner reaction profile. For supply chain heads, this translates to a more robust process that is easier to control and scale, offering a distinct competitive advantage in cost reduction in vitamin manufacturing.
Mechanistic Insights into Lewis Acid-Catalyzed Condensation
The mechanistic foundation of this invention rests on the activation of the electrophilic species—typically an acetal or an aldehyde derivative—by the Lewis acid catalyst. The metal center coordinates with the oxygen atoms of the acetal group, increasing the electrophilicity of the adjacent carbon and facilitating nucleophilic attack by the electron-rich double bond of the olefinic partner. This coordination complex lowers the activation energy of the rate-determining step, allowing the reaction to proceed rapidly even at sub-ambient temperatures. The choice of catalyst is critical; experimental data within the patent highlights that while aluminum chloride is effective, transition metal triflates like Scandium Triflate and Ytterbium Triflate offer enhanced turnover frequencies and better tolerance to functional groups. This mechanistic precision ensures that the coupling occurs exclusively at the desired position, preventing the formation of branched isomers that could compromise the biological activity of the final Vitamin E product.
Furthermore, the presence of a base, such as pyridine or triethylamine, plays a nuanced role in scavenging protons generated during the reaction cycle, thereby preventing acid-catalyzed polymerization of the olefinic starting materials. This dual-catalyst system (Lewis acid plus weak base) creates a buffered environment that maximizes the lifespan of the active catalytic species. From an impurity control perspective, this mechanism is superior because it avoids the generation of free carbocations that typically lead to rearrangement by-products. The result is a reaction mixture with a simplified impurity profile, which significantly reduces the burden on purification teams. For R&D directors focused on quality by design (QbD), understanding this catalytic cycle provides the necessary leverage to optimize reaction parameters for maximum purity and yield without extensive trial-and-error experimentation.
How to Synthesize Vitamin E Intermediates Efficiently
The synthesis of these critical intermediates involves a carefully orchestrated sequence of mixing, catalysis, and workup steps designed to maximize efficiency and safety. The process begins with the preparation of the reaction vessel under an inert atmosphere to prevent moisture interference, followed by the sequential addition of the olefinic substrate and the acetal or aldehyde partner in a suitable organic solvent such as dichloromethane or toluene. Once the substrates are homogenized, the Lewis acid catalyst is introduced at a controlled rate to manage the exotherm, maintaining the reaction temperature within the optimal window of -30°C to 20°C. Detailed standard operating procedures for this synthesis, including specific molar ratios and quenching protocols, are essential for reproducibility.
- Prepare the reaction mixture by combining a general formula (II) olefinic compound with a formula (III) aldehyde/imine or formula (IV) acetal in an organic solvent.
- Introduce a Lewis acid catalyst such as Ferric Chloride (FeCl3), Scandium Triflate, or Ytterbium Triflate at temperatures ranging from -50°C to +25°C.
- Allow the catalytic condensation to proceed for 30 minutes to 6 hours, followed by aqueous workup and purification to isolate the novel intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers tangible benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the raw material portfolio; by utilizing widely available commodity chemicals like citral derivatives and simple acetals, manufacturers can reduce dependency on exotic or monopolized precursors. This diversification of the supply base enhances resilience against market volatility and ensures continuity of supply even during global disruptions. Additionally, the mild reaction conditions eliminate the need for specialized high-pressure or high-temperature reactors, allowing production to be shifted to multipurpose facilities with lower capital expenditure requirements. This flexibility is a key driver for cost reduction in vitamin manufacturing, enabling producers to respond agilely to fluctuating market demands.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in favor of abundant metal salts like Iron(III) Chloride significantly lowers the direct material costs associated with the catalytic system. Moreover, the high selectivity of the reaction reduces the volume of solvents and adsorbents required for purification, leading to substantial savings in waste disposal and utility consumption. By shortening the synthetic route and improving overall yield, the cost per kilogram of the final intermediate is drastically reduced, improving margin potential for downstream vitamin producers.
- Enhanced Supply Chain Reliability: The robustness of the Lewis acid catalyzed process ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by out-of-specification results. The use of stable, non-pyrophoric catalysts simplifies logistics and storage requirements, reducing the administrative burden of handling hazardous materials. This reliability allows supply chain heads to optimize inventory levels and reduce safety stock, freeing up working capital while maintaining high service levels for pharmaceutical and nutraceutical customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram scale to multi-kilogram batches without loss of efficiency. The reduced generation of hazardous waste and the ability to recycle solvents align with increasingly stringent environmental regulations, mitigating compliance risks. This environmental stewardship not only protects the company's reputation but also future-proofs the manufacturing asset against tightening global emissions and waste disposal standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: What are the primary advantages of the Lewis acid catalyzed method for Vitamin E intermediates?
A: The method described in Patent CN1449393A offers significant advantages including mild reaction conditions (often near room temperature), high regioselectivity, and the ability to use readily available starting materials like citral derivatives and acetals, reducing overall process complexity.
Q: Which catalysts are most effective for this synthesis according to the patent data?
A: Experimental data indicates that Ferric Chloride (FeCl3), Scandium Triflate (Sc(OTf)3), and Ytterbium Triflate (Yb(OTf)3) provide superior yields compared to traditional aluminum chloride, with FeCl3 being particularly noted for its efficiency and cost-effectiveness in large-scale applications.
Q: Can this process be scaled for industrial Vitamin E production?
A: Yes, the process is highly amenable to scale-up. It operates effectively at atmospheric pressure and moderate temperatures, avoiding the need for extreme high-pressure equipment, which simplifies reactor design and enhances safety profiles for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin E Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex vitamins relies on the availability of high-quality, consistently supplied intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Vitamin E intermediate meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of Lewis acid catalysis and deliver products that facilitate efficient downstream synthesis for our global partners.
We invite you to collaborate with us to explore how this advanced synthetic route can optimize your production costs and enhance your supply chain stability. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing strategies, ensuring a seamless partnership driven by innovation and mutual growth.
