Advanced Ruthenium Complex Production: Enhancing Catalytic Efficiency and Commercial Viability
Introduction to Next-Generation Ruthenium Catalyst Manufacturing
The landscape of homogeneous catalysis is undergoing a significant transformation driven by the demand for higher atomic efficiency and simplified purification protocols in the synthesis of fine chemicals. Patent CN108137637B introduces a groundbreaking methodology for the production of [bis(2-phosphinoethyl)amine]carbonylhalohydride ruthenium(II) complexes, widely known in the industry as Ru-MACHO type catalysts. This intellectual property addresses critical bottlenecks associated with traditional synthetic routes, specifically targeting the inefficiencies of ruthenium source utilization and the challenging removal of phosphine by-products. By leveraging a novel reaction pathway involving dinuclear ruthenium precursors, primary alcohols, and bases, this technology enables the direct formation of single-isomer catalytic species with exceptional purity profiles. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and economically viable manufacturing paradigms for high-value hydrogenation catalysts used extensively in pharmaceutical and agrochemical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ru-MACHO catalysts has relied on the reaction of tris(triphenylphosphine)ruthenium(II) chloride carbonyl hydride with bis[2-(diphenylphosphino)ethyl]amine ligands in solvents such as toluene. As illustrated in the reaction scheme below, this conventional pathway suffers from inherently poor atom economy, calculated at merely 17.3 mass%. A major operational drawback is the stoichiometric generation of three equivalents of triphenylphosphine (PPh3) as a by-product. This phosphine waste is notoriously difficult to separate from the final catalyst product, often requiring extensive chromatographic purification or multiple recrystallization steps that drastically reduce overall yield. Furthermore, the product obtained through this legacy route is typically a mixture of coordination isomers, necessitating additional washing procedures with n-hexane and ethanol that can degrade the yield from 85% down to 55%, thereby inflating production costs and extending lead times.
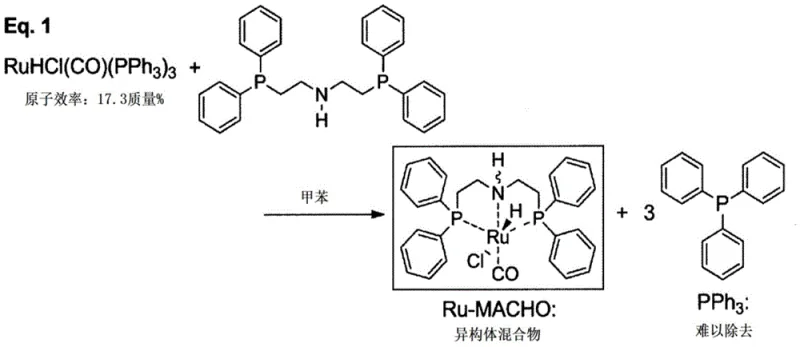
The Novel Approach
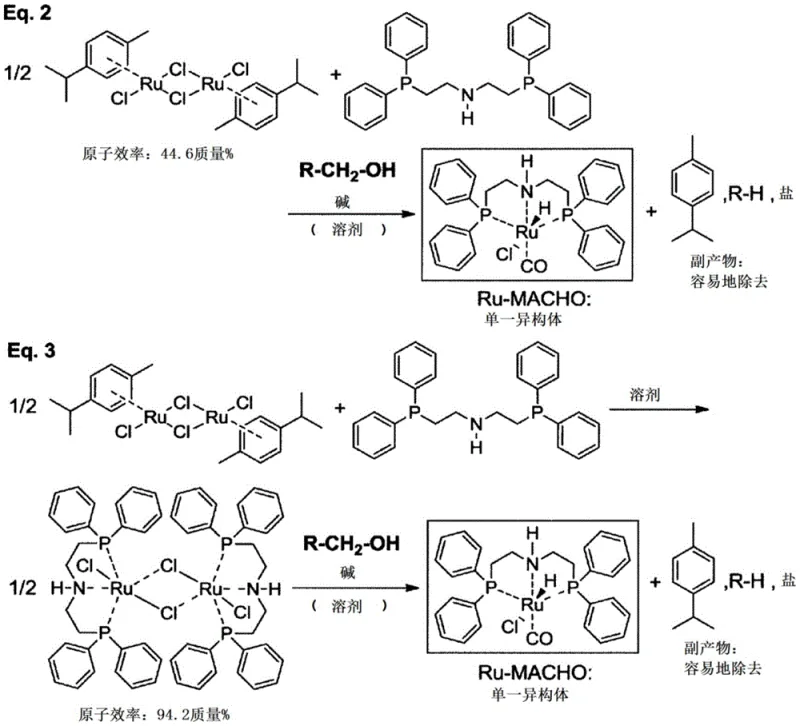
In stark contrast, the patented innovation utilizes dichloro(p-cymene)ruthenium(II) dimer as the ruthenium source, reacting it with the amine ligand in the presence of a primary alcohol and a base. This strategic change elevates the atomic efficiency of the ruthenium source to greater than or equal to 44.6 mass%, representing a substantial improvement in resource utilization. The reaction mechanism elegantly bypasses the formation of stubborn phosphine impurities; instead, the by-products are limited to hydrocarbons and inorganic salts which can be effortlessly removed via simple filtration or aqueous washing. Crucially, this method delivers the target ruthenium complex as a single isomer directly from the reaction vessel, eliminating the yield losses associated with isomer separation. This streamlined approach not only enhances the chemical purity but also simplifies the downstream processing infrastructure required for commercial scale-up.
Mechanistic Insights into Alcohol-Mediated Hydride Formation
The core of this technological advancement lies in the sophisticated interplay between the dinuclear ruthenium precursor and the primary alcohol solvent system. Detailed mechanistic studies suggest that the reaction proceeds through a well-defined sequence involving the initial formation of a ruthenium alkoxide intermediate. Upon treatment with a base, this intermediate undergoes a critical beta-hydride elimination step, which serves as the internal source for the hydride ligand essential for catalytic activity. Subsequently, a decarbonylation event occurs, likely facilitated by the thermal conditions of the reflux, installing the carbonyl ligand onto the divalent ruthenium center. This intramolecular rearrangement ensures that both the hydride and carbonyl functionalities are integrated with high fidelity, avoiding the stochastic distribution of ligands seen in less controlled environments. The result is a highly defined coordination sphere around the metal center that is prerequisite for the catalyst's renowned performance in carbonyl hydrogenation reactions.
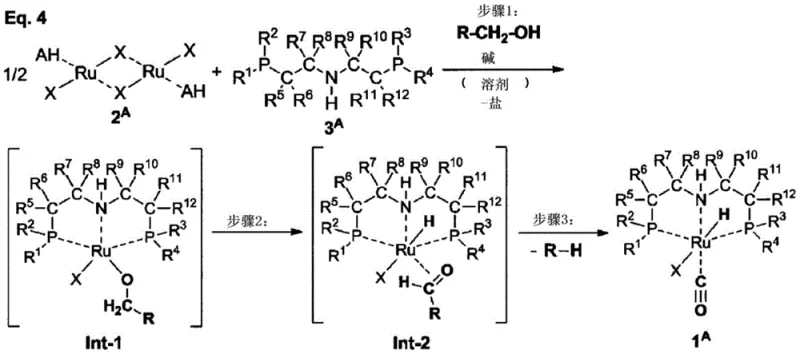
From an impurity control perspective, the choice of primary alcohol plays a dual role as both a reactant and a solvent, which significantly influences the purity profile of the final batch. The mechanism dictates that the alkyl group of the alcohol is eliminated as a hydrocarbon by-product (R-H). By selecting alcohols where the resulting hydrocarbon has low coordinating ability and high volatility, such as 3-methoxy-1-butanol or 1-butanol, the process ensures that these by-products do not interfere with the catalyst's active site or remain trapped in the crystal lattice. This mechanistic understanding allows process engineers to fine-tune reaction parameters, such as temperature and base equivalents, to maximize the conversion of the dinuclear precursor while minimizing the formation of off-cycle species. The robustness of this mechanism across various ligand substitutions further underscores its versatility for generating a library of tailored ruthenium catalysts.
How to Synthesize Ru-MACHO Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing high-purity ruthenium complexes suitable for industrial application. The process begins with the neutralization of the amine ligand salt using an alkali metal alkoxide in a primary alcohol solvent, followed by the addition of the dinuclear ruthenium dimer. The mixture is then subjected to reflux conditions to drive the ligand exchange and hydride formation to completion. Detailed standardized synthesis steps, including precise molar ratios, temperature profiles, and workup procedures, are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- React dichloro(p-cymene)ruthenium(II) dimer with bis[2-(diphenylphosphino)ethyl]amine in the presence of a primary alcohol and a base to form an intermediate.
- Facilitate beta-hydride elimination and decarbonylation steps under reflux conditions to generate the active hydride species.
- Isolate the single-isomer ruthenium complex product via filtration and washing, removing soluble salt by-products efficiently.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route translates into tangible operational efficiencies and risk mitigation strategies. The elimination of difficult-to-remove triphenylphosphine by-products fundamentally alters the cost structure of catalyst manufacturing by removing the need for expensive chromatographic resins or excessive solvent consumption associated with purification. This simplification of the downstream process directly contributes to cost reduction in pharmaceutical intermediates manufacturing, allowing for more competitive pricing models without compromising on quality standards. Furthermore, the ability to produce the catalyst as a single isomer without yield-degrading separation steps ensures a more predictable and consistent output volume per batch, which is critical for maintaining stable inventory levels in a just-in-time supply chain environment.
- Cost Reduction in Manufacturing: The significant increase in atomic efficiency from 17.3% to over 44.6% means that less raw ruthenium material is wasted per kilogram of finished product. Since ruthenium is a precious metal with volatile market pricing, maximizing its incorporation into the final product is a primary driver for lowering the cost of goods sold. Additionally, the replacement of complex purification workflows with simple filtration and washing steps reduces utility costs, labor hours, and solvent disposal fees, creating a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The starting materials for this process, such as dichloro(p-cymene)ruthenium(II) dimer and common primary alcohols, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of bases and solvents, provides flexibility in sourcing strategies, allowing procurement teams to switch vendors based on availability and price without revalidating the entire process. This resilience ensures continuous supply continuity even during periods of raw material scarcity or logistical disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in multi-gram to potential ton-scale batches without loss of efficiency. The generation of benign by-products like hydrocarbons and salts simplifies waste management and aligns with increasingly stringent environmental regulations regarding heavy metal and phosphine discharge. This environmental compatibility facilitates easier permitting for new production facilities and supports corporate sustainability goals by minimizing the ecological footprint of catalyst manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium complex production technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making.
Q: How does the new synthesis method improve atom efficiency compared to conventional routes?
A: The novel method utilizes dichloro(p-cymene)ruthenium(II) dimer, achieving an atom efficiency of over 44.6 mass%, significantly higher than the 17.3 mass% of traditional methods using tris(triphenylphosphine)ruthenium(II).
Q: What are the primary advantages regarding impurity profiles in this production process?
A: Unlike conventional methods that generate difficult-to-remove triphenylphosphine by-products, this process produces only hydrocarbons and salts which are easily removed by simple filtration or water washing.
Q: Does this method yield a specific isomer of the ruthenium complex?
A: Yes, the process directly yields the ruthenium complex as a single isomer without the need for complex separation techniques required for isomer mixtures in older methodologies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ru-MACHO Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced ruthenium complex synthesis technology for the global fine chemical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in large-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Ru-MACHO catalyst delivered meets the exacting standards required for sensitive hydrogenation applications in API synthesis.
We invite forward-thinking organizations to collaborate with us to leverage this efficient production route for their specific catalytic needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how switching to this optimized supply chain can enhance your operational efficiency and bottom line.
