Advanced One-Pot Synthetic Strategy for High-Purity 2-Hydroxypyridine Derivatives
Introduction to Next-Generation Pyridine Synthesis
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A pivotal advancement in this domain is detailed in patent CN101857566B, which discloses a revolutionary synthetic method for 2-hydroxypyridine compounds. These compounds serve as critical building blocks in the pharmaceutical industry, acting as key intermediates for biologically active molecules, catalysts, and even monomers for liquid crystal displays. The traditional pathways to access these valuable scaffolds have often been plagued by multi-step sequences, harsh reaction conditions, and prohibitive costs, creating bottlenecks for both R&D teams and supply chain managers. This new methodology addresses these pain points head-on by utilizing a readily available 2-chloropyridine compound as the initiation raw material.
The core innovation lies in the ability to prepare the target 2-hydroxypyridine compound in a single reaction container through a continuous two-step reaction, completely bypassing the need for the complete processing and isolation of intermediate steps. This "one-pot" strategy represents a paradigm shift in process chemistry, offering a streamlined approach that remarkably improves the yield of the product while effectively shortening the overall reaction time. By enabling milder reaction conditions, the process not only enhances safety profiles but also saves significant production costs, thereby ensuring that the resulting 2-hydroxypyridine compounds possess substantial industrial application value. For procurement specialists and technical directors alike, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering high-quality materials at competitive margins.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-hydroxypyridine derivatives has relied on methodologies that are increasingly viewed as obsolete in the context of modern green chemistry and cost-efficient manufacturing. Prior art, such as methods documented in older literature like the Journal of Organic Chemistry or Tetrahedron Letters, often necessitates the use of expensive starting materials that drive up the initial cost of goods. Furthermore, these traditional routes frequently require multistep syntheses to achieve the target compound, each step introducing potential yield losses, increased solvent consumption, and additional purification burdens. A significant drawback of these conventional techniques is the requirement for harsh reaction conditions, often involving extremely high temperatures that demand specialized equipment and pose safety risks.
For instance, certain prior art methods involve the cleavage of alkoxy groups under severe thermal stress, as illustrated in historical precedents where temperatures reaching 150-180°C were necessary to effect the transformation. Such energy-intensive processes are economically inadequate for large-scale production and simultaneously waste valuable energy resources, rendering them unsuitable for suitability for industrialized production in a competitive market. The cumulative effect of these inefficiencies is a supply chain that is fragile, expensive, and unable to meet the rigorous demands of high-volume API manufacturing. Consequently, there is a pressing need for a method that can overcome these inherent limitations of low productivity and high material costs.
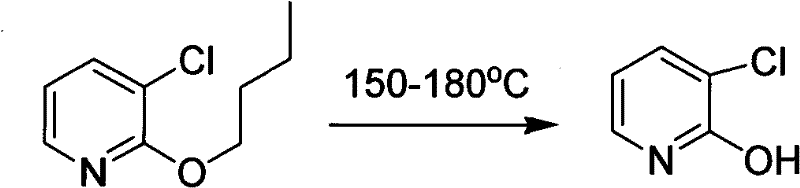
The Novel Approach
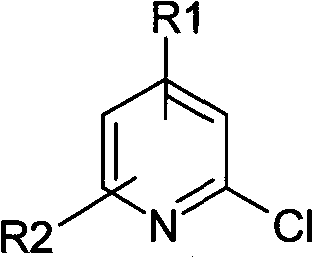
In stark contrast to the cumbersome legacy methods, the novel approach disclosed in the patent leverages the accessibility and reactivity of 2-chloropyridine compounds to forge a direct and efficient path to the desired hydroxylated products. The strategy employs a clever sequence where the 2-chloropyridine compound is first dissolved in a polar aprotic solvent, such as tert-butanol, and treated with an alcoholic solution of sodium tert-butoxide. This initial step activates the system, allowing for a reaction to proceed at a moderate temperature range of 80-120°C over a period of 10-24 hours. Following this, the reaction mixture is processed by filtering, desolvating, and adjusting the pH to a neutral range of 4-7 upon the addition of water, setting the stage for the final transformation without isolating the intermediate species.
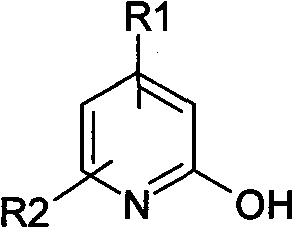
The second phase of this continuous process involves mixing the prepared solution with hydrobromic acid and maintaining a temperature of 60-100°C with stirring for 6-20 hours. This mild acidic treatment effectively converts the intermediate into the final 2-hydroxypyridine compound, which can then be isolated by simple filtration after cooling. The versatility of this method is underscored by the broad scope of compatible starting materials, where R1 and R2 groups on the pyridine ring can vary independently to include hydrogen, alkyl, halogen, or carboxyl substituents. This flexibility allows manufacturers to produce a diverse array of derivatives from a common platform, significantly enhancing the utility of this synthetic route for custom synthesis projects.
Mechanistic Insights into Nucleophilic Substitution and Acidolysis
To fully appreciate the technical robustness of this synthesis, one must delve into the mechanistic underpinnings that drive the transformation from a chloro-substituted pyridine to a hydroxyl-substituted analogue. The first stage of the reaction likely proceeds via a nucleophilic aromatic substitution mechanism, facilitated by the strong base sodium tert-butoxide. In this step, the tert-butoxide anion acts as a nucleophile, attacking the electron-deficient carbon at the 2-position of the pyridine ring, displacing the chloride leaving group. This results in the formation of a 2-tert-butoxypyridine intermediate, although this species is not isolated in the patented process. The use of a polar aprotic solvent environment supports the stability of the ionic species involved and promotes the kinetics of this substitution, ensuring high conversion rates even at the relatively moderate temperatures of 80-120°C.
The subsequent addition of hydrobromic acid triggers an acidolysis reaction, which is the key to unlocking the final hydroxyl functionality. The strong acid protonates the ether oxygen of the intermediate, making the tert-butyl group a good leaving group in the form of a stable carbocation or isobutylene gas, depending on the specific conditions. This cleavage regenerates the phenolic hydroxyl group on the pyridine ring, yielding the target 2-hydroxypyridine compound. The control of pH during the workup phase is critical; adjusting the solution to a pH of 4-7 ensures that the product precipitates efficiently or remains in a state conducive to the final acid treatment, minimizing the formation of salts or byproducts. This mechanistic elegance allows for precise impurity control, as the continuous nature of the reaction prevents the accumulation of unstable intermediates that could degrade into complex impurity profiles.
How to Synthesize 2-Hydroxypyridine Efficiently
Implementing this synthetic route in a laboratory or pilot plant setting requires careful attention to the specific parameters outlined in the patent embodiments to ensure optimal yield and purity. The process is designed to be operationally simple, yet it demands precision in stoichiometry and thermal management to replicate the success seen in the patent examples. Operators must ensure that the molar ratio of the 2-chloropyridine compound to sodium tert-butoxide is maintained within the preferred range, typically around 1:2, to drive the initial substitution to completion without excessive waste of the base. The choice of solvent is also paramount, with tert-butanol being the preferred medium due to its compatibility with the reagents and its boiling point which aligns well with the required reaction temperatures.
- Dissolve the 2-chloropyridine compound in a polar aprotic solvent such as tert-butanol and react with sodium tert-butoxide at 80-120°C for 10-24 hours.
- Adjust the pH of the reaction mixture to 4-7 after removing the solvent and adding water.
- Mix the solution with hydrobromic acid (30-50% concentration) and stir at 60-100°C for 6-20 hours to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers profound benefits for procurement managers and supply chain heads who are tasked with optimizing the cost structure and reliability of their raw material sourcing. The elimination of intermediate isolation steps translates directly into a drastic simplification of the manufacturing workflow, reducing the number of unit operations required and consequently lowering the labor and equipment overheads associated with production. This streamlined process inherently leads to substantial cost savings, as fewer solvents are consumed, less waste is generated, and the overall throughput of the facility is increased. For buyers seeking cost reduction in fine chemical manufacturing, this technology represents a significant opportunity to lower the total cost of ownership for critical pyridine intermediates.
- Cost Reduction in Manufacturing: The economic impact of this one-pot strategy is multifaceted, primarily driven by the removal of purification and drying steps between the two reaction stages. By avoiding the isolation of the intermediate, the process saves on filtration media, drying energy, and solvent recovery costs, which are often the most expensive components of a chemical bill of materials. Furthermore, the use of commercially available and inexpensive starting materials like 2-chloropyridine derivatives ensures that the input costs remain low and stable, shielding the supply chain from volatility associated with exotic reagents. The milder reaction conditions also reduce energy consumption, contributing to a leaner and more cost-effective production model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Supply continuity is a top priority for any manufacturing organization, and this synthetic route bolsters reliability by relying on commodity chemicals that are widely available in the global market. The robustness of the reaction conditions means that the process is less prone to failure or batch-to-batch variability, ensuring a consistent output of high-quality material. This predictability allows supply chain planners to maintain lower safety stocks and reduce lead times for high-purity pharmaceutical intermediates, as the risk of production delays due to complex synthesis issues is minimized. The ability to operate continuously in a single vessel also simplifies scheduling and capacity planning, further strengthening the resilience of the supply network.
- Scalability and Environmental Compliance: As regulatory pressures mount regarding waste disposal and environmental impact, the greener profile of this synthesis becomes a distinct competitive advantage. The reduction in solvent usage and waste generation aligns with modern sustainability goals, making it easier for facilities to maintain compliance with stringent environmental regulations. The simplicity of the process facilitates easy scale-up from kilogram to tonne quantities without the need for complex engineering modifications, supporting the commercial scale-up of complex pharmaceutical intermediates. This scalability ensures that suppliers can rapidly respond to increasing market demand without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthetic method. These insights are derived directly from the technical specifications and advantageous effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for their own production needs. Understanding these details is crucial for making informed decisions about adopting this route for the manufacture of high-value pyridine derivatives.
Q: What are the primary advantages of this one-pot synthesis method?
A: The primary advantages include the elimination of intermediate isolation steps, significantly reduced reaction times, milder reaction conditions compared to traditional high-temperature methods, and overall lower production costs due to simplified processing.
Q: Can this method be scaled for industrial production?
A: Yes, the method is specifically designed for industrial application value, utilizing commercially available raw materials and standard reaction vessels, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What types of substituents are compatible with this synthetic route?
A: The process is versatile and accommodates various substituents at the R1 and R2 positions on the pyridine ring, including hydrogen, alkyl groups, halogens, and carboxyl groups, allowing for the synthesis of a wide range of derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxypyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the success of our clients' drug development and manufacturing programs. Our team of expert chemists has extensively analyzed patents like CN101857566B to integrate best-in-class processes into our own manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards, guaranteeing that the 2-hydroxypyridine compounds you receive are fit for purpose in even the most sensitive applications.
We invite you to collaborate with us to leverage these advanced synthetic strategies for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume and purity needs, helping you optimize your budget without sacrificing quality. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities can serve as a reliable pharmaceutical intermediates supplier for your long-term growth.
