Advanced Manufacturing Strategy for Roxadustat Intermediates via Optimized Cyclization
Advanced Manufacturing Strategy for Roxadustat Intermediates via Optimized Cyclization
The pharmaceutical industry is constantly seeking more efficient pathways for the production of critical small molecule inhibitors, particularly for treating chronic conditions like anemia associated with kidney disease. Patent CN109400528B introduces a groundbreaking synthesis method for Roxadustat (also known as Cobumei), a potent hypoxia-inducible factor prolyl hydroxylase inhibitor (HIF-PHI). This technology represents a significant leap forward in process chemistry, moving away from the hazardous and convoluted routes of the past toward a streamlined, six-step sequence that prioritizes safety and scalability. The core innovation lies in the strategic use of a classical Bischler-Napieralski isoquinoline synthesis followed by a selective dehydroaromatization, enabling the construction of the complex isoquinoline scaffold with remarkable precision. 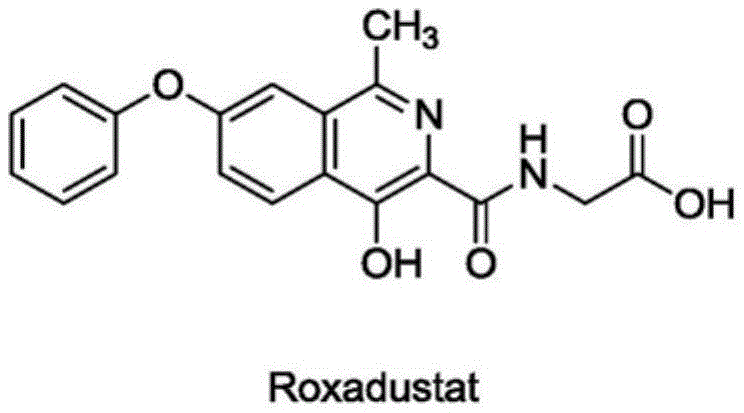
For R&D directors and process chemists, the implications of this patent are profound, offering a robust alternative to legacy methods that often struggle with impurity profiles and harsh reaction conditions. The disclosed method starts from 2-amino-3-hydroxy-3-(4-phenoxyphenyl)propionic acid, a readily accessible chiral building block, and transforms it through acetic anhydride protection, esterification, cyclization, oxidation, hydrolysis, and finally glycine condensation. This logical progression not only simplifies the synthetic tree but also ensures that each intermediate is stable and easily isolable, facilitating rigorous quality control throughout the manufacturing chain. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is key to delivering high-purity Roxadustat to the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Roxadustat has been plagued by significant technical hurdles that hindered cost-effective commercialization. Early approaches, such as those described in patent WO2004108681, relied on 4-nitrophthalonitrile as a starting material, necessitating a lengthy multi-step sequence involving substitution, hydrolysis, and rearrangement to construct the isoquinoline ring. These routes often suffered from low overall yields and required extensive purification efforts, including column chromatography, which is notoriously difficult to scale. Furthermore, other documented methods, such as those in CN201280036322, utilized phosphorus oxychloride not just as a reagent but as a bulk solvent, creating severe safety hazards and corrosion issues for reactor vessels. 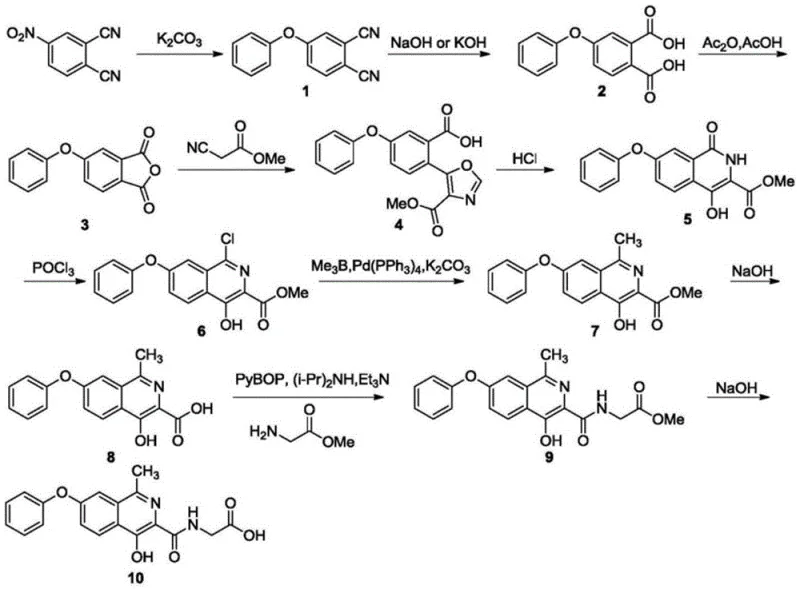
Additionally, literature routes involving sodium or lithium metal reagents imposed stringent requirements on reaction equipment, demanding strictly anhydrous conditions and specialized handling protocols that drive up operational costs. The use of Grignard reagents for methylation, as seen in patent CN108424388, frequently led to uncontrollable side reactions, resulting in complex impurity spectra that were challenging to separate. These factors combined to create a supply chain bottleneck, where the cost reduction in API manufacturing was stifled by the inherent dangers and inefficiencies of the chemistry itself. For supply chain heads, these legacy processes represented a continuous risk of batch failure and extended lead times.
The Novel Approach
In stark contrast, the methodology outlined in CN109400528B offers a paradigm shift by leveraging a biomimetic-inspired cyclization strategy that is both atom-economical and operationally simple. The new route bypasses the need for dangerous alkali metals and high-pressure autoclaves, instead utilizing mild thermal conditions ranging from 50°C to 80°C for the majority of the transformations. The key breakthrough is the formation of the 3,4-dihydroisoquinoline intermediate via a catalytic cyclization, followed by a highly selective oxidation using 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) to establish the aromatic system. 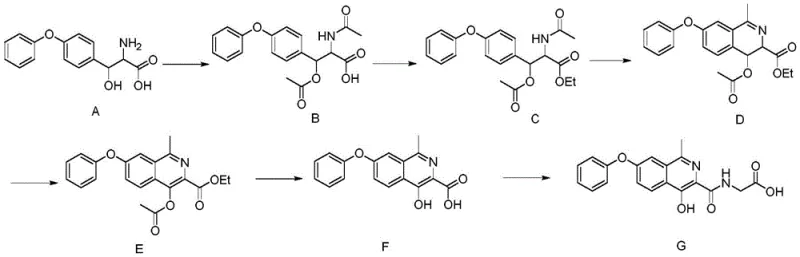
This approach drastically simplifies the downstream processing, as the intermediates precipitate cleanly from reaction mixtures, often requiring only filtration and drying rather than complex chromatographic separation. The use of common organic solvents like acetonitrile, ethanol, and tetrahydrofuran further enhances the commercial viability, allowing for easy solvent recovery and recycling. By condensing the final carboxylic acid intermediate with glycine using standard peptide coupling agents, the process achieves a total yield of approximately 69.5%, a figure that significantly outperforms many historical benchmarks. This streamlined workflow directly addresses the pain points of procurement managers by reducing raw material waste and shortening the production cycle.
Mechanistic Insights into Bischler-Napieralski Cyclization and DDQ Oxidation
The heart of this synthetic innovation lies in the precise execution of the cyclization and subsequent aromatization steps, which dictate the structural integrity of the final isoquinoline core. In step three of the process, the protected amino-ester intermediate undergoes cyclization in the presence of phosphorus oxychloride (POCl3). Unlike traditional methods where POCl3 acts as a corrosive solvent, here it functions as a potent dehydrating catalyst in acetonitrile, promoting the intramolecular condensation to form the dihydroisoquinoline ring with high regioselectivity. This controlled environment minimizes the formation of polymeric byproducts and ensures that the methyl group at the 1-position is correctly oriented, a critical stereochemical feature for biological activity.
Following cyclization, the aromatization step utilizes DDQ as a hydride acceptor to convert the dihydro-species into the fully aromatic isoquinoline system. This oxidation is performed at mild temperatures (20°C to 30°C) in tetrahydrofuran, preventing the degradation of the sensitive phenoxy ether linkage which can occur under harsher oxidative conditions. The mechanism involves the abstraction of hydride ions from the dihydro-ring, driven by the strong electron-withdrawing nature of the cyano and chloro groups on the quinone. This gentle yet effective oxidation strategy is crucial for impurity control, as it avoids the over-oxidation or ring-opening side reactions often seen with stronger oxidants. The result is a high-purity intermediate that sets the stage for the final amide bond formation, ensuring the final API meets stringent regulatory specifications.
How to Synthesize Roxadustat Efficiently
The implementation of this synthesis route requires careful attention to reaction stoichiometry and temperature control to maximize the reported 69.5% overall yield. The process begins with the protection of the amine functionality, followed by esterification to activate the carboxylic acid for the subsequent cyclization. The critical cyclization step must be monitored closely to ensure complete conversion before proceeding to the oxidation phase. Detailed standard operating procedures regarding reagent addition rates and workup protocols are essential for reproducibility. For a comprehensive guide on executing this chemistry, please refer to the standardized synthesis steps provided below.
- Protect the amino group of the starting material using acetic anhydride in an organic solvent like acetic acid at 65-70°C.
- Perform esterification with alcohol (e.g., ethanol) and acid catalyst to form the ethyl ester intermediate.
- Execute Bischler-Napieralski cyclization using phosphorus oxychloride as a catalyst in acetonitrile to form the dihydroisoquinoline ring.
- Conduct aromatic ring oxidation using 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) to aromatize the isoquinoline system.
- Hydrolyze the ester group using inorganic base (e.g., NaOH) to obtain the carboxylic acid intermediate.
- Condense the acid intermediate with glycine using a coupling agent (e.g., DIC/HOBt) to yield the final Roxadustat product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers substantial benefits that resonate deeply with procurement managers and supply chain leaders focused on resilience and cost efficiency. The elimination of exotic or hazardous reagents like sodium metal and the reduction of phosphorus oxychloride usage from a solvent to a catalyst dramatically lowers the barrier to entry for manufacturing. This shift not only reduces the cost of goods sold (COGS) by minimizing safety infrastructure requirements but also broadens the pool of qualified suppliers who can safely handle the process. The reliance on commodity chemicals and solvents ensures that raw material availability remains stable, even during global supply chain disruptions.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive transition metal catalysts and complex purification techniques like column chromatography. By utilizing a high-yielding sequence where intermediates can be isolated via simple filtration, the consumption of silica gel and organic solvents for purification is drastically reduced. Furthermore, the high atom economy of the cyclization step means less waste generation, leading to lower disposal costs and a smaller environmental footprint. These efficiencies compound to deliver a much more competitive price point for the final active pharmaceutical ingredient without compromising on quality.
- Enhanced Supply Chain Reliability: The use of robust, non-hazardous starting materials ensures a steady and predictable supply flow, mitigating the risks associated with regulated precursor chemicals. Since the reaction conditions are mild and do not require specialized high-pressure equipment, production can be easily distributed across multiple manufacturing sites, enhancing supply continuity. The shortened production cycle, attributed to the reduced number of unit operations and faster workup times, allows for quicker turnaround on orders, effectively reducing lead time for high-purity pharmaceutical intermediates. This agility is vital for meeting the fluctuating demands of the global anemia treatment market.
- Scalability and Environmental Compliance: The synthetic pathway is inherently designed for scale-up, with each step demonstrating excellent heat transfer characteristics and manageable exotherms. The avoidance of heavy metals and the minimization of chlorinated solvent usage align perfectly with modern green chemistry principles and strict environmental regulations. This compliance reduces the regulatory burden on manufacturing partners and facilitates smoother technology transfer between sites. The ability to run the process in standard glass-lined or stainless steel reactors makes it an ideal candidate for commercial scale-up of complex heterocycles, ensuring long-term sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this specific Roxadustat synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these details helps stakeholders make informed decisions about adopting this superior manufacturing route.
Q: What are the primary safety advantages of this new Roxadustat synthesis route compared to prior art?
A: Unlike previous methods that utilized hazardous sodium or lithium metal reagents or required phosphorus oxychloride as a bulk solvent, this novel process employs phosphorus oxychloride merely as a catalyst in safer organic solvents like acetonitrile. Furthermore, it eliminates the need for high-pressure autoclaves in the final steps, significantly reducing operational risks and equipment requirements for large-scale production.
Q: How does this method improve the purity profile of the final API intermediate?
A: The process avoids the use of Grignard reagents, which are known to generate difficult-to-remove side products in traditional routes. By utilizing a controlled oxidation step with DDQ and mild hydrolysis conditions, the method achieves a final product purity exceeding 99%, minimizing the need for complex column chromatography purification.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the route is specifically designed for industrial viability. It utilizes readily available starting materials, operates under mild temperatures (mostly below 80°C), and achieves a high total yield of approximately 69.5%. The avoidance of extreme conditions and hazardous reagents makes it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this optimized synthesis route for the global supply of anemia therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific solvent systems and mild thermal conditions required for this process, while our rigorous QC labs enforce stringent purity specifications to guarantee product consistency. We are committed to bridging the gap between innovative academic chemistry and robust industrial application.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and safe manufacturing technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to see how our expertise can accelerate your project timelines and enhance your supply chain security. Let us help you secure a reliable source of high-quality Roxadustat intermediates for your next generation of therapies.
