Revolutionizing Roxadustat Production: A Palladium-Free Oxidative Strategy for Commercial Scale-Up
Introduction to Next-Generation Roxadustat Synthesis
The pharmaceutical landscape for anemia treatment has been significantly shaped by Roxadustat, a hypoxia-inducible factor prolyl hydroxylase inhibitor. However, the manufacturing of this critical active pharmaceutical ingredient (API) has historically faced hurdles related to complex multi-step syntheses and reliance on costly noble metal catalysts. Patent CN113816904A introduces a transformative synthetic methodology that fundamentally alters the production paradigm for Roxadustat and its key intermediates. This innovation leverages a direct oxidative functionalization strategy using persulfate oxidants and amide solvents to construct the crucial 1-methyl isoquinoline scaffold. By bypassing traditional palladium-catalyzed methylation steps, this technology offers a more sustainable and economically viable pathway. The method is characterized by its use of cheap, easily obtainable raw materials and mild reaction conditions that facilitate high yields and simplified post-treatment processes. For global supply chain stakeholders, this represents a pivotal shift towards more resilient and cost-effective manufacturing capabilities for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
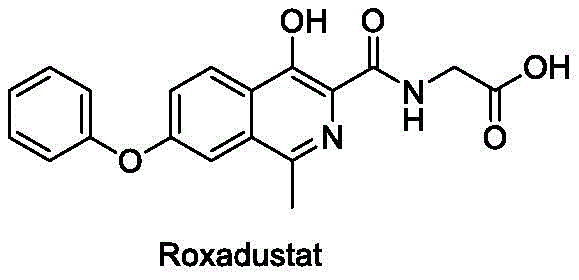
The Limitations of Conventional Methods
Historically, the synthesis of Roxadustat has been plagued by inefficient routes that impose significant burdens on both R&D and production teams. Existing literature, such as the routes disclosed in CN201310302822.0, typically involves a cumbersome sequence where an intermediate 4-hydroxy-7-phenoxyisoquinoline-3-carboxylate must undergo methylation at the 1-position. This critical step often necessitates the use of tetramethylethylenediamine and acetic anhydride, followed by hydrogenation using palladium on carbon. The reliance on noble metal palladium not only inflates raw material costs but also introduces stringent requirements for heavy metal removal to meet regulatory standards. Furthermore, these conventional pathways frequently utilize high-boiling point reagents like morpholine and acetic anhydride, which are notoriously difficult to remove during purification. The presence of these reagents often leads to the formation of stubborn by-products, necessitating additional reaction steps to convert impurities, thereby extending the overall process timeline and reducing overall throughput efficiency.
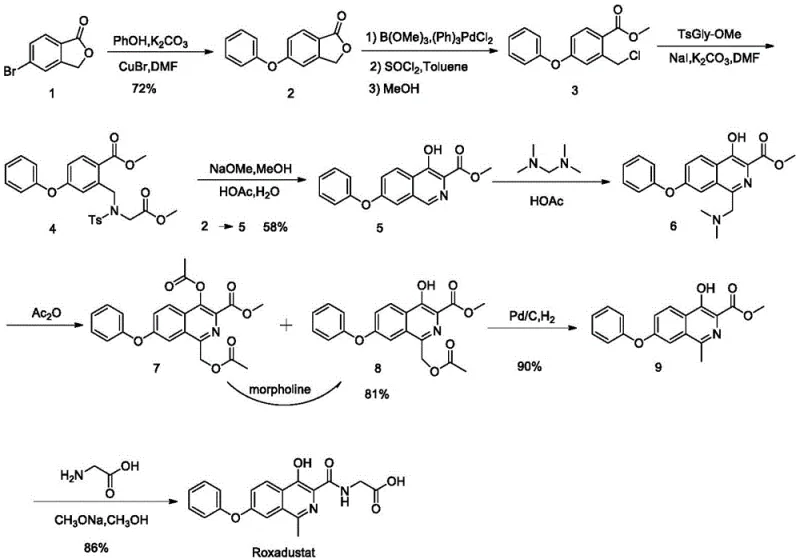
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent utilizes a direct oxidative coupling reaction to install the necessary side chain functionality in a single step. This method reacts a starting isoquinoline compound (SM) with an amide derivative (SM-A) under the influence of a persulfate oxidant. This innovative transformation effectively merges the methylation and functionalization steps, drastically shortening the synthetic route. The reaction proceeds under mild thermal conditions, typically between 60°C and 70°C, eliminating the energy-intensive high-temperature steps required in other methodologies. By avoiding the use of palladium catalysts and hazardous oxidants like hydrogen peroxide in dangerous rearrangement steps, this new route significantly enhances operational safety. The resulting intermediates are easier to purify, often requiring only simple filtration or solvent washing, which streamlines the workflow and minimizes waste generation. This streamlined architecture makes the process inherently more suitable for the commercial scale-up of complex pharmaceutical intermediates.
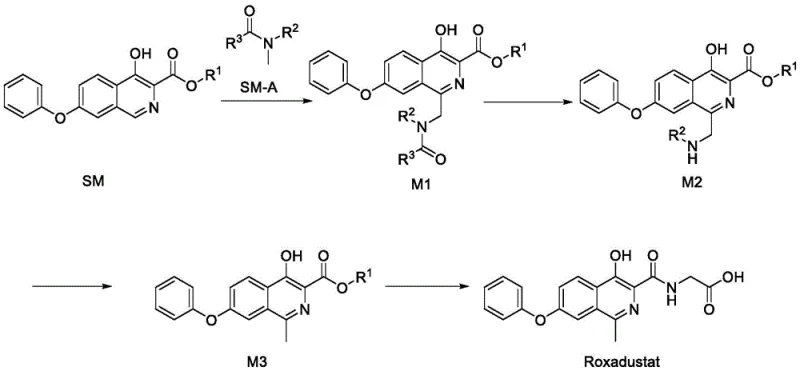
Mechanistic Insights into Persulfate-Mediated Oxidative Functionalization
The core of this technological breakthrough lies in the radical-mediated oxidative functionalization of the isoquinoline ring. The mechanism involves the generation of sulfate radical anions from the persulfate oxidant, which subsequently abstract hydrogen atoms or interact with the amide solvent to generate reactive amidyl radicals. These radicals attack the electron-deficient isoquinoline ring at the 1-position, forming a new carbon-nitrogen or carbon-carbon bond depending on the specific amide used. This direct C-H functionalization bypasses the need for pre-functionalized halogenated intermediates or organometallic reagents. The choice of amide solvent, such as N,N-dimethylacetamide (DMA) or N,N-dimethylformamide (DMF), acts dually as both the reaction medium and the source of the alkyl/amino group. This dual role simplifies the stoichiometry and reduces the volume of waste solvents. The reaction is highly selective, favoring the formation of the desired 1-substituted intermediate M1 with minimal regio-isomers, provided the oxidant ratio is carefully controlled between 1:1 and 1:5 molar equivalents relative to the substrate.
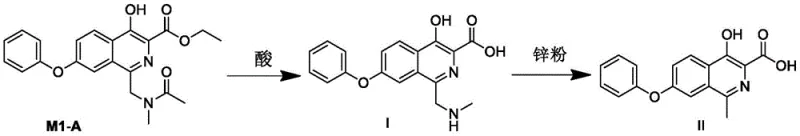
Furthermore, the patent provides critical insights into impurity control, which is paramount for R&D directors focused on purity profiles. A major challenge in Roxadustat synthesis is the formation of impurity Compound I and its downstream derivative, Compound II. These impurities arise from incomplete hydrolysis or side reactions during the reduction phase and share similar polarity and solubility characteristics with the target molecule, making separation via crystallization extremely difficult. The new method addresses this by optimizing the hydrolysis conditions of intermediate M1 to M2. By strictly controlling the acid concentration and temperature during the cleavage of the amide group, the formation of Compound I is minimized. Subsequent reduction steps using zinc powder are also tuned to prevent the carryover of these impurities into the final M3 intermediate. This proactive impurity management ensures that the final API meets stringent ICH guidelines without the need for resource-intensive preparative chromatography.
How to Synthesize Roxadustat Intermediates Efficiently
The synthesis of Roxadustat intermediates via this oxidative route is designed for operational simplicity and robustness. The process begins with the dissolution of the isoquinoline starting material in an amide solvent, followed by the controlled addition of an aqueous persulfate solution. The reaction is monitored via TLC or HPLC until the starting material is fully consumed, typically within 1 to 6 hours. Upon completion, the product can often be isolated by simply adding water to precipitate the solid, followed by filtration and drying. The subsequent hydrolysis step involves dissolving the intermediate in a mixed solvent system of alcohol and ether, treating it with concentrated hydrochloric acid, and heating mildly to effect cleavage. The final reduction to the methyl group utilizes zinc powder in acetic acid, a standard and safe reducing system. For detailed standardized operating procedures and specific parameter optimization, please refer to the comprehensive guide below.
- Oxidative Functionalization: React the isoquinoline starting material (SM) with an amide solvent (SM-A) and a persulfate oxidant at 60-70°C to form the 1-substituted intermediate M1.
- Acid Hydrolysis: Treat intermediate M1 with concentrated hydrochloric acid in an alcohol/ether solvent mixture at 30-40°C to hydrolyze the amide group, yielding intermediate M2.
- Reductive Methylation & Coupling: Reduce intermediate M2 using zinc powder in acetic acid to form the methyl-substituted intermediate M3, followed by coupling with glycine to finalize Roxadustat.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The most significant advantage is the drastic reduction in raw material costs associated with the elimination of noble metal catalysts. Palladium is not only expensive but also subject to volatile market pricing and supply constraints; removing it from the bill of materials stabilizes cost projections and mitigates supply risk. Additionally, the simplified purification protocols mean that manufacturing cycles are shorter, allowing for faster turnover and improved responsiveness to market demand. The avoidance of high-boiling point solvents reduces the energy load required for solvent recovery and distillation, contributing to lower utility costs and a smaller environmental footprint. These factors combined create a more resilient supply chain capable of sustaining high-volume production without the bottlenecks typical of complex catalytic processes.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the reduction in solvent usage directly lower the cost of goods sold (COGS). By replacing complex multi-step sequences with a concise oxidative pathway, labor and equipment utilization time are significantly optimized. The ability to isolate intermediates through simple precipitation rather than column chromatography further reduces consumable costs and waste disposal fees. This economic efficiency allows for substantial cost savings in pharmaceutical intermediate manufacturing, making the final API more competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like persulfates, zinc powder, and common amides ensures a stable and diversified supply base. Unlike specialized catalysts that may have single-source suppliers, these reagents are widely available globally, reducing the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in input quality, ensuring consistent output and reliable delivery schedules for downstream partners.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of hazardous high-pressure hydrogenation steps make this process inherently safer and easier to scale from pilot plant to commercial production. The reduction in heavy metal waste simplifies effluent treatment and helps facilities maintain strict environmental compliance standards. This alignment with green chemistry principles not only reduces regulatory burden but also enhances the sustainability profile of the manufacturing operation, which is increasingly important for corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These answers are derived directly from the patent specifications and experimental data to provide accurate guidance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing frameworks. We encourage technical teams to review these points closely to appreciate the depth of process optimization achieved.
Q: How does this new synthesis route improve upon traditional palladium-catalyzed methods?
A: Traditional routes often rely on expensive noble metal palladium catalysts for methylation and require complex purification steps to remove high-boiling point reagents like morpholine. This novel oxidative method eliminates the need for palladium entirely, utilizing cheap persulfate oxidants and common amide solvents, which drastically simplifies downstream processing and reduces heavy metal contamination risks.
Q: What specific impurity challenges does this patent address in Roxadustat manufacturing?
A: The patent identifies critical impurities, specifically Compound I and Compound II, which possess polarities and solubilities similar to the final API, making them notoriously difficult to separate. The new method includes specific control strategies during the hydrolysis of M1 to M2 and the reduction to M3 to minimize the formation of these persistent impurities, ensuring higher final purity without extensive chromatography.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial scalability. It operates under mild reaction temperatures (30-70°C), avoids hazardous high-temperature steps found in prior art, and uses readily available, non-toxic reagents. The simplified workup procedures, such as direct filtration or simple solvent washing, make it highly robust for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical industry. Our team of expert chemists has thoroughly analyzed this patented technology and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity pharmaceutical intermediates that meet the most rigorous quality standards. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure stringent purity specifications are met for every batch. By leveraging our expertise in oxidative functionalization and impurity control, we can offer a reliable supply of Roxadustat intermediates that support your drug development and commercialization timelines.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive value for your organization. Let us be your partner in achieving cost-effective and sustainable production of high-quality pharmaceutical ingredients.
