Revolutionizing Statin Intermediate Production via Advanced Kinetic Resolution and Scalable Cyanation Technology
The pharmaceutical industry continuously seeks robust synthetic pathways for critical statin intermediates, specifically targeting the efficient production of Ethyl (R)-4-cyano-3-hydroxybutyrate. This compound serves as a pivotal chiral building block for HMG-CoA reductase inhibitors, such as Atorvastatin, which has historically demonstrated massive market demand. Patent CN102295579B introduces a transformative synthesis process that overcomes the limitations of traditional chiral pool approaches by employing hydrolytic kinetic resolution technology. This method utilizes non-chiral raw materials to directly synthesize high-value chiral compounds, marking a significant shift towards more sustainable and cost-effective manufacturing paradigms. By leveraging advanced Salen metal complex catalysis, the process achieves exceptional stereocontrol under mild reaction conditions, thereby addressing the critical needs of R&D directors focused on purity and impurity profiles. The strategic implementation of this technology not only enhances the chemical efficiency of the synthesis but also aligns with modern green chemistry principles by reducing reliance on expensive, naturally derived chiral starting materials that often suffer from supply volatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ethyl (R)-4-cyano-3-hydroxybutyrate has relied heavily on biological methods or chemical transformations starting from expensive chiral pools. Conventional biological methods often involve the biotransformation of ethyl 4-chloroacetoacetate or the use of cyanohydrolase on racemic epoxides, which can introduce complexities regarding enzyme stability, substrate specificity, and downstream processing. Chemical methods frequently utilize chiral starting materials like L-(-)-malic acid or chiral epichlorohydrin, which are inherently costly and subject to fluctuating market availability. Furthermore, many prior art cyanation methods rely on aprotic polar solvents that complicate post-reaction workup and solvent recovery, leading to increased environmental burdens and operational costs. These traditional routes often struggle to balance high enantiomeric purity with economic viability, creating bottlenecks for procurement managers seeking reliable [precise industry noun] supplier partnerships. The reliance on multi-step sequences from natural chiral sources also limits the scalability of production, making it difficult to meet the surging demands of the global cardiovascular therapeutic market without incurring prohibitive expenses.
The Novel Approach
The innovative process detailed in CN102295579B circumvents these challenges by initiating synthesis from racemic ethyl epoxybutyrate, a readily accessible and inexpensive non-chiral precursor. Through the application of hydrolytic kinetic resolution using specific Salen catalysts, the process selectively hydrolyzes one enantiomer while leaving the desired (S)-epoxybutyrate intact with high optical purity. This approach effectively decouples the production of chiral intermediates from the constraints of the chiral pool, offering a more predictable and controllable supply chain. The subsequent ring-opening reaction utilizes aqueous sodium cyanide, a departure from the hazardous and difficult-to-remove organic solvents used in legacy methods. This modification drastically simplifies the isolation of the final product, as the use of water facilitates easier phase separation and reduces the load on waste treatment facilities. For supply chain heads, this represents a tangible improvement in commercial scale-up of complex [precise industry noun plural], as the process is designed with industrial feasibility in mind, minimizing unit operations and maximizing throughput efficiency without compromising on the stringent quality standards required for pharmaceutical applications.
Mechanistic Insights into Salen-Catalyzed Hydrolytic Kinetic Resolution
The core of this technological breakthrough lies in the precise mechanistic action of the Salen-metal complex catalyst during the hydrolytic kinetic resolution step. The catalyst, typically a Cobalt-Salen complex modified with Lewis acids such as SnCl2, SnCl4, FeCl3, or ZnCl2, coordinates with water molecules to activate the epoxide ring of the racemic starting material. Due to the chiral environment created by the Salen ligand, the activation energy for the hydrolysis of the (R)-enantiomer is significantly lower than that of the (S)-enantiomer. Consequently, the (R)-epoxide is rapidly converted into the corresponding diol, while the (S)-epoxide remains largely unreacted, allowing for its recovery with exceptional enantiomeric excess. This differentiation in reaction rates is the fundamental principle of kinetic resolution, and the specific tuning of the catalyst's electronic and steric properties ensures that the selectivity factor remains high throughout the reaction duration of 12 to 24 hours at temperatures between 0°C and 20°C.
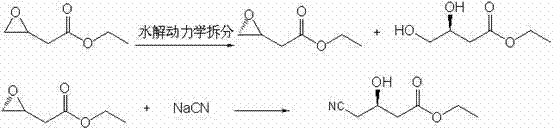
Following the resolution, the control of impurities becomes paramount to ensure the suitability of the intermediate for downstream API synthesis. The process incorporates a quenching step using L-ascorbic acid, which serves to deactivate the metal catalyst and prevent potential metal-catalyzed side reactions that could degrade the product quality. The separation of the unreacted (S)-epoxybutyrate from the formed diol is achieved through vacuum distillation, a physical separation method that avoids the introduction of additional chemical impurities. The subsequent cyanation step proceeds via a nucleophilic attack by the cyanide ion on the less hindered carbon of the epoxide ring, resulting in regioselective ring opening. This mechanism ensures that the cyano group is installed at the correct position (C4) while retaining the stereochemical integrity at C3. The use of water as the solvent in this step not only aids in heat dissipation during the exothermic ring-opening but also ensures that inorganic salts remain in the aqueous phase, facilitating the extraction of the organic product with high purity levels exceeding 99.5%.
How to Synthesize Ethyl (R)-4-cyano-3-hydroxybutyrate Efficiently
To implement this synthesis route effectively, operators must adhere to strict control parameters regarding temperature and molar ratios to maximize yield and optical purity. The process begins with the preparation of the reaction mixture in tetrahydrofuran, where the precise addition of water and catalyst initiates the kinetic resolution. Detailed standard operating procedures dictate the specific stoichiometry required to balance the conversion rate against the enantiomeric excess, ensuring that the unreacted epoxide is recovered in optimal quantities. Following the resolution and distillation, the cyanation step requires careful handling of the aqueous cyanide solution to maintain safety and reaction efficiency. The detailed standardized synthesis steps are outlined in the guide below, providing a clear roadmap for technical teams to replicate the high-performance results documented in the patent literature.
- Perform hydrolytic kinetic resolution of ethyl epoxybutyrate using a Salen-Co catalyst and water in THF at 0-20°C to obtain (S)-epoxybutyrate.
- Separate the unreacted (S)-epoxybutyrate from the diol byproduct via vacuum distillation under controlled temperature.
- React the purified (S)-epoxybutyrate with aqueous sodium cyanide solution to effect ring-opening and yield the final cyano-hydroxy ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers profound advantages that directly address the pain points of procurement and supply chain management in the fine chemical sector. By shifting away from expensive chiral starting materials to commodity chemicals, the raw material cost base is significantly reduced, providing a buffer against market volatility. The simplification of the workup procedure, particularly the elimination of difficult-to-recycle aprotic solvents in the cyanation step, leads to substantial cost savings in utility consumption and waste disposal. These efficiencies translate into a more competitive pricing structure for the final intermediate, enabling pharmaceutical manufacturers to optimize their overall cost of goods sold. Furthermore, the robustness of the chemical process ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification materials.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in the final product and the use of water as a reaction medium drastically simplify the purification process. This reduction in downstream processing complexity means fewer unit operations are required, leading to lower energy consumption and reduced labor costs per kilogram of product. Additionally, the ability to recover and potentially recycle the THF solvent further enhances the economic viability of the process. By avoiding the premium pricing associated with chiral pool starting materials, manufacturers can achieve significant margin improvements without compromising on the quality of the high-purity [precise industry noun] delivered to clients.
- Enhanced Supply Chain Reliability: Sourcing non-chiral raw materials like ethyl epoxybutyrate is inherently more stable than relying on fermentation-derived chiral acids or specialized enzymatic preparations. This shift mitigates the risk of supply disruptions caused by agricultural variables or bioprocess failures. The chemical synthesis route is less susceptible to biological contamination issues, ensuring a continuous and predictable flow of materials. For supply chain planners, this reliability is crucial for reducing lead time for high-purity [precise industry noun plural], as it allows for more accurate forecasting and inventory management. The scalability of the process ensures that supply can be ramped up quickly to meet unexpected spikes in demand from downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing reaction conditions that are easily manageable in large-scale reactors. The mild temperature range (0-20°C) reduces the cooling load requirements, making the process energy-efficient even at multi-ton scales. Moreover, the use of water in the cyanation step aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. By minimizing the use of hazardous organic solvents, the process reduces the environmental footprint of the manufacturing facility. This compliance advantage is critical for maintaining operational licenses and meeting the sustainability goals of global pharmaceutical partners who prioritize green chemistry in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific beneficial effects and background technical details provided in the patent documentation, offering clarity on the operational advantages and chemical performance of the route. Understanding these aspects is essential for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement specialists assessing the quality assurances provided by suppliers utilizing this technology.
Q: What is the enantiomeric excess (ee) achievable with this Salen-catalyzed process?
A: According to patent data CN102295579B, the hydrolytic kinetic resolution step consistently achieves an ee value of 99.8% to 99.9% for the (S)-epoxybutyrate intermediate, ensuring high stereochemical purity for the final API intermediate.
Q: How does the use of water in the cyanation step benefit industrial production?
A: Using water as the solvent for the sodium cyanide ring-opening reaction eliminates the need for expensive aprotic polar solvents. This significantly simplifies post-reaction workup, reduces solvent recovery costs, and minimizes environmental pollution associated with volatile organic compound emissions.
Q: Can this process be scaled for commercial API manufacturing?
A: Yes, the process utilizes non-chiral, readily available starting materials and mild reaction conditions (0-20°C). The elimination of complex chiral pool precursors and the use of robust Salen catalysts make it highly suitable for commercial scale-up of complex chiral building blocks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl (R)-4-cyano-3-hydroxybutyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Ethyl (R)-4-cyano-3-hydroxybutyrate meets the exacting standards required for statin API synthesis. Our infrastructure is designed to support the complex chemical transformations described in patent CN102295579B, leveraging our expertise in kinetic resolution and hazardous chemistry handling to deliver superior products.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your long-term strategic goals. Let us be your partner in delivering high-performance chemical solutions that drive innovation in the pharmaceutical industry.
