Advanced Synthesis of Boron Nitrogen Benzocarbazole Derivatives for Commercial OLED Production
Advanced Synthesis of Boron Nitrogen Benzocarbazole Derivatives for Commercial OLED Production
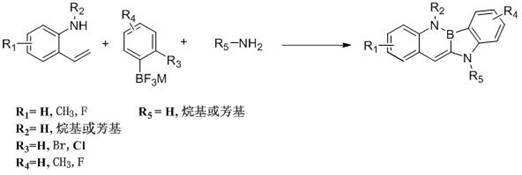
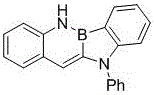
The rapid evolution of the organic electronics sector demands increasingly sophisticated molecular architectures capable of delivering superior charge transport and luminescent properties. In this context, Patent CN112778347B introduces a groundbreaking synthetic methodology for constructing boron nitrogen benzocarbazole derivatives, a class of compounds pivotal for next-generation organic light-emitting diodes (OLEDs) and organic field-effect transistors (OFETs). This technology represents a significant paradigm shift from conventional multi-step syntheses to a streamlined, one-pot three-component coupling strategy. By leveraging a sophisticated palladium-catalyzed cascade reaction, the process enables the precise assembly of complex boron-nitrogen conjugated systems directly from readily available precursors. For R&D directors and procurement specialists alike, this innovation offers a compelling pathway to access high-purity optoelectronic intermediates with enhanced structural diversity and reduced environmental impact, positioning it as a critical asset for reliable OLED material supplier networks seeking to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of boron-nitrogen aromatic compounds has been fraught with significant technical and logistical challenges that hinder commercial scalability. Traditional approaches, such as olefin metathesis ring-closing reactions, often suffer from excessively long synthetic routes and low overall yields, making them economically unviable for mass production. Furthermore, these legacy methods frequently rely on toxic and sensitive reagents that pose severe safety risks and generate substantial hazardous waste, complicating regulatory compliance. Another prevalent technique, electrophilic borylation, typically necessitates the use of highly reactive and unstable boron bromide compounds, requiring stringent anhydrous and anaerobic conditions that are difficult to maintain on a large scale. These conventional pathways also exhibit poor substrate compatibility, limiting the ability to introduce diverse functional groups essential for tuning the electronic properties of the final material. Consequently, the industry has faced a bottleneck in accessing novel boron-nitrogen scaffolds efficiently, driving up costs and extending lead times for high-purity organic semiconductors.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN112778347B utilizes a highly efficient transition metal-catalyzed tandem reaction to construct the boron-nitrogen core in a single operational step. This innovative route combines o-aminostyrene derivatives, aryl fluoroborate derivatives, and amines under mild thermal conditions, typically ranging from 25°C to 100°C. The use of stable aryl fluoroborates eliminates the need for hazardous boron halides, drastically simplifying handling requirements and enhancing workplace safety. Moreover, the reaction demonstrates exceptional specificity and high yields, as evidenced by the successful synthesis of various derivatives without the formation of complex byproduct mixtures. This streamlined approach not only accelerates the discovery of new materials but also facilitates cost reduction in electronic chemical manufacturing by minimizing unit operations and solvent consumption. The versatility of this method allows for the incorporation of various substituents, enabling the precise engineering of HOMO-LUMO levels for specific device applications.
Mechanistic Insights into Pd-Catalyzed Cascade Cyclization
The core of this technological breakthrough lies in the intricate interplay between the palladium catalyst, phosphine ligands, and the multifunctional substrates. The reaction likely proceeds through a complex catalytic cycle involving oxidative addition of the aryl fluoroborate species to the palladium center, followed by coordination and insertion of the alkene moiety from the o-aminostyrene derivative. The presence of bulky and electron-rich phosphine ligands, such as 2-dicyclohexylphosphine-2',4',6'-triisopropylbiphenyl, plays a crucial role in stabilizing the active catalytic species and promoting the reductive elimination step that forms the final C-N and C-B bonds simultaneously. This concerted mechanism ensures the formation of the fused boron-nitrogen heterocyclic ring with high regioselectivity. The base, typically an alkoxide or carbonate, serves to activate the amine nucleophile and neutralize acidic byproducts, driving the equilibrium towards product formation. Understanding these mechanistic nuances is vital for process chemists aiming to further optimize reaction parameters for commercial scale-up of complex optoelectronic intermediates.
From an impurity control perspective, the robustness of this catalytic system is particularly noteworthy. The high specificity of the palladium-mediated transformation minimizes the generation of structural isomers or oligomeric side products that are common in radical-based or harsh electrophilic processes. The mild reaction conditions prevent the decomposition of sensitive functional groups, such as trifluoromethyl or methoxy substituents, which might otherwise degrade under stronger acidic or basic environments. Post-reaction processing is straightforward, involving standard aqueous workups and recrystallization, which effectively removes residual palladium species and inorganic salts. This results in a crude product profile that is significantly cleaner than those obtained from traditional methods, reducing the burden on downstream purification units. For quality assurance teams, this translates to a more consistent impurity profile and easier validation of the manufacturing process, ensuring that the final high-purity boron nitrogen benzocarbazole meets the rigorous specifications required for display and lighting applications.
How to Synthesize Boron Nitrogen Benzocarbazole Efficiently
The practical implementation of this synthesis route is designed for ease of operation while maintaining high standards of reproducibility and safety. The process begins with the careful selection of high-quality starting materials, specifically ensuring that the o-aminostyrene and aryl fluoroborate components are free from moisture and oxygen, which could deactivate the catalyst. The reaction is typically conducted in common organic solvents like toluene, DMF, or THF, which offer excellent solubility for the reactants and facilitate heat transfer during the exothermic phases of the catalytic cycle. Operators must maintain an inert atmosphere, usually nitrogen or argon, throughout the heating period to preserve catalyst activity. Following the reaction completion, the mixture is cooled, and the product is isolated through liquid-liquid extraction followed by drying and recrystallization. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Combine o-aminostyrene derivatives, aryl fluoroborate derivatives, and organic amines in a reaction vessel under inert atmosphere.
- Add a composite catalyst system comprising an organopalladium source, phosphine ligands, and a basic compound in a suitable solvent.
- Heat the mixture to 25-100°C for 1-24 hours, then perform extraction, drying, and recrystallization to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic methodology offers transformative benefits that extend beyond mere technical feasibility. The reliance on commodity chemicals such as aniline, styrenes, and simple inorganic bases means that the raw material supply chain is robust and less susceptible to the volatility associated with specialized reagents. This accessibility ensures a steady flow of inputs, mitigating the risk of production stoppages due to material shortages. Furthermore, the elimination of toxic and sensitive boron halides reduces the need for specialized storage facilities and hazardous waste disposal protocols, leading to substantial cost savings in operational overhead. The simplified workup procedure, which avoids complex chromatographic separations in favor of crystallization, significantly shortens the batch cycle time. These factors collectively contribute to a more resilient and cost-effective supply chain for advanced electronic materials.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, commercially available starting materials and the avoidance of expensive, air-sensitive reagents. By consolidating multiple bond-forming events into a single pot, the method drastically reduces solvent usage, energy consumption, and labor hours associated with intermediate isolation. The high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as byproducts. Additionally, the ability to use standard laboratory glassware and heating equipment without the need for cryogenic cooling or high-pressure reactors lowers the capital expenditure required for setting up production lines. These efficiencies translate directly into a lower cost of goods sold, providing a competitive edge in the pricing of finished optoelectronic components.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the broad availability of the requisite precursors, which are produced by numerous global chemical manufacturers. Unlike proprietary catalysts or custom-synthesized building blocks that may have long lead times, the key components for this reaction can be sourced from multiple vendors, reducing dependency on single suppliers. The mild reaction conditions also imply that the process can be transferred between different manufacturing sites with minimal requalification effort, enhancing flexibility in production planning. Moreover, the stability of the intermediates and the final product under ambient conditions simplifies logistics and warehousing, allowing for safer and cheaper transportation. This reliability is crucial for meeting the tight delivery schedules demanded by the consumer electronics and display industries.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonne quantities is facilitated by the absence of hazardous reagents and the use of common solvents that are easily recovered and recycled. The reduction in toxic waste generation aligns with increasingly stringent environmental regulations, minimizing the risk of compliance violations and associated fines. The simple purification strategy involving recrystallization is inherently scalable and does not require the extensive silica gel or resin columns often needed for chromatography, which can be a bottleneck in large-scale operations. Furthermore, the high selectivity of the reaction reduces the load on wastewater treatment systems by limiting the variety of organic contaminants. This environmental friendliness not only supports corporate sustainability goals but also streamlines the permitting process for new manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boron-nitrogen synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities. They cover aspects ranging from substrate scope and reaction conditions to purification strategies and potential applications. Reviewing these FAQs can help stakeholders assess the suitability of this method for their specific project requirements and identify any necessary adjustments for their unique manufacturing contexts.
Q: What are the primary advantages of this synthesis method over traditional electrophilic borylation?
A: Unlike traditional electrophilic borylation which often requires sensitive boron bromide compounds and harsh conditions, this method utilizes stable aryl fluoroborates and operates under mild temperatures (25-100°C), significantly improving safety and operational simplicity.
Q: Is this process suitable for large-scale manufacturing of optoelectronic materials?
A: Yes, the process employs commercially available raw materials like aniline and styrenes, uses standard solvents such as toluene or DMF, and involves simple post-treatment steps like extraction and recrystallization, making it highly scalable for industrial production.
Q: What types of substituents are compatible with this boron-nitrogen aromatic system construction?
A: The method demonstrates broad substrate tolerance, accommodating various substituents including fluorine, methyl groups, methoxy, and trifluoromethyl groups on the amine and styrene components, allowing for fine-tuning of electronic properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boron Nitrogen Benzocarbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced intermediates like boron nitrogen benzocarbazole derivatives play in the development of cutting-edge display and lighting technologies. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market launch is seamless. We are committed to delivering materials that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical instrumentation to verify every batch. Our dedication to quality and consistency makes us a trusted partner for global innovators seeking to push the boundaries of organic electronics.
We invite you to collaborate with us to leverage this patented technology for your next product development cycle. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient synthesis route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a stable supply of high-performance materials for your future applications.
