Advanced Asymmetric Hydrogenation Technology for High-Purity Optically Active Alcohol Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for the synthesis of chiral building blocks, particularly optically active alcohols which serve as critical intermediates in drug discovery and agrochemical production. Patent CN1926083B introduces a groundbreaking advancement in this domain by detailing a highly efficient preparation method for optically active alcohols utilizing novel ruthenium, rhodium, and iridium metal complexes. Unlike traditional methods that often struggle with substrate compatibility or require harsh conditions, this invention leverages asymmetric hydrogenation under pressurized hydrogen gas to achieve superior yields and stereoselectivity. The core innovation lies in the specific design of the chiral ligands coordinated to the metal center, which facilitates the activation of molecular hydrogen and its subsequent transfer to the ketone substrate with precise stereocontrol. This technology addresses long-standing challenges in synthesizing base-sensitive compounds, offering a versatile platform for generating high-value chiral intermediates.
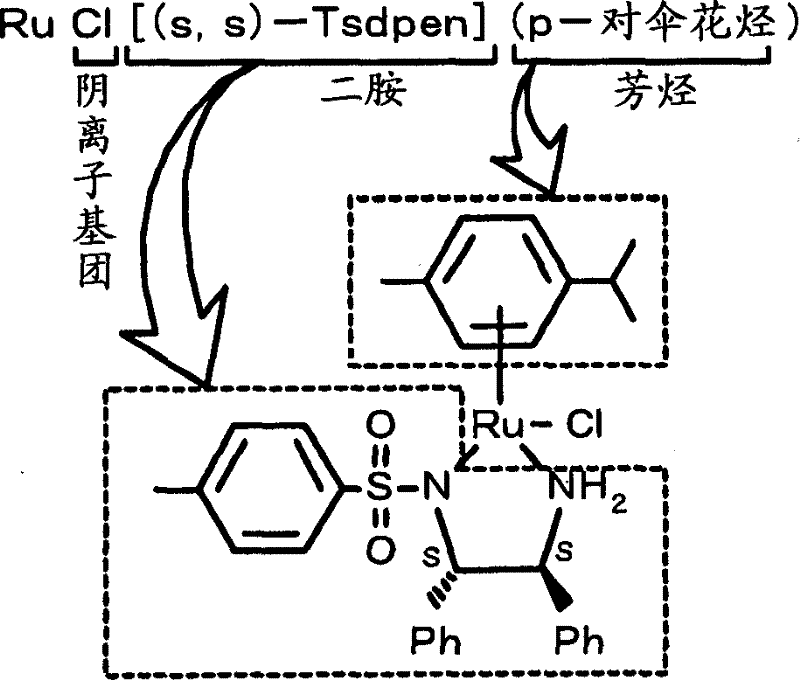
As a reliable pharmaceutical intermediate supplier, understanding the nuances of such catalytic systems is vital for ensuring supply chain continuity and product quality. The patent explicitly highlights the capability to produce optically active alcohols from ketone compounds that were previously difficult to hydrogenate effectively. By operating under pressurized hydrogen conditions ranging from 1 to 200 atmospheres and utilizing polar solvents like methanol, the process eliminates the need for external organic bases that often complicate downstream purification. This methodological shift not only simplifies the reaction workflow but also enhances the overall atom economy of the synthesis. For procurement managers and supply chain heads, this translates to a more predictable manufacturing process with fewer variables that could impact batch consistency or lead times for high-purity chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active alcohols from ketones has relied heavily on asymmetric transfer hydrogenation or hydrogenation methods that necessitate the presence of strong organic bases. For instance, prior art such as Japanese Patent Laid-Open No. 2003-104993 describes using ruthenium-BINAP complexes which, while effective for simple ketones, often result in lower yields or enantiomeric excess when applied to more complex or sensitive substrates. Furthermore, methods disclosed in documents like Japanese Patent Laid-Open No. 11-322649 require the use of triethylamine or similar bases to activate the catalyst or facilitate proton transfer. This reliance on basic conditions presents a significant bottleneck for substrates containing base-labile functional groups, such as alpha-halo ketones or alkynyl ketones, which can undergo decomposition or side reactions like elimination or polymerization. Consequently, manufacturers face increased costs due to lower yields, the need for extensive purification to remove base residues, and the inability to process certain valuable intermediates efficiently.
The Novel Approach
The novel approach detailed in CN1926083B circumvents these limitations by employing specialized metal complexes, such as RuCl[(S,S)-Tsdpen](arene), which are active for asymmetric hydrogenation without the mandatory addition of organic bases. This base-free operational window is a transformative advantage, allowing for the successful hydrogenation of substrates that are unstable in alkaline environments. The process utilizes direct pressurized hydrogen gas rather than hydrogen donors like isopropanol, which drives the reaction equilibrium towards completion and often results in higher conversion rates. Additionally, the versatility of the ligand system allows for fine-tuning the steric and electronic properties of the catalyst to match specific substrate requirements, ensuring high enantiomeric excess even for challenging molecules. This robustness makes the technology ideal for cost reduction in fine chemical manufacturing, as it reduces waste generation and simplifies the workup procedure by eliminating base neutralization steps.
Mechanistic Insights into Asymmetric Hydrogenation with Chiral Metal Complexes
The efficacy of this synthesis method is rooted in the sophisticated coordination chemistry of the catalysts employed. The general formula (1) described in the patent features a ruthenium center coordinated with a chiral sulfonamide diamine ligand and an arene group. This specific arrangement creates a chiral pocket that dictates the facial selectivity of the hydrogen attack on the ketone carbonyl group. The mechanism likely involves the heterolytic cleavage of the hydrogen molecule across the metal-ligand bond, forming a metal-hydride and an ammonium species that cooperatively transfer a hydride and a proton to the substrate. This bifunctional catalysis is key to achieving high turnover numbers and stereoselectivity. The absence of a requirement for external base suggests that the ligand itself participates in the proton shuttle mechanism, stabilizing the transition state through hydrogen bonding interactions. Understanding this mechanistic pathway is crucial for R&D directors aiming to optimize reaction parameters for new substrates.
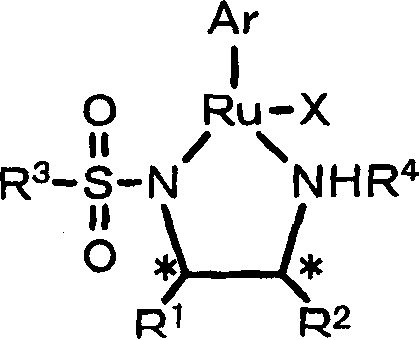
Furthermore, the patent elucidates the structural flexibility of the catalyst system, allowing for variations in the R groups of the diamine ligand and the arene moiety. For example, substituting the arene from p-cymene to mesitylene or hexamethylbenzene can modulate the steric bulk around the metal center, influencing both activity and selectivity. The inclusion of additives such as perchlorate salts (e.g., LiClO4, NaClO4) is also noted to enhance reaction rates and enantioselectivity in certain cases, possibly by affecting the ionization state of the catalyst or stabilizing charged intermediates. This level of tunability ensures that the process can be adapted for the commercial scale-up of complex chiral intermediates across a wide range of chemical spaces. By controlling the stereochemistry at the asymmetric carbon through the choice of (R) or (S) configured ligands, manufacturers can access either enantiomer of the target alcohol with high precision, meeting the stringent purity specifications required for API synthesis.
How to Synthesize Optically Active Alcohols Efficiently
Implementing this technology in a production setting involves a straightforward yet precise protocol that maximizes the potential of the chiral catalysts. The general procedure entails dissolving the ketone substrate and the metal complex in a polar solvent, typically methanol or ethanol, within a pressure-resistant reactor. The system is then purged with inert gas to remove oxygen, which could deactivate the catalyst, before being pressurized with hydrogen. Reaction temperatures are generally mild, ranging from -30°C to 50°C, which helps in preserving sensitive functional groups while maintaining reasonable reaction kinetics. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, pressure settings, and workup procedures validated in the patent examples. This streamlined approach minimizes operational complexity and ensures reproducible results across different batches.
- Prepare the reaction vessel by charging the specific chiral metal complex catalyst (e.g., RuCl[(S,S)-Tsdpen](arene)) and the target ketone substrate into a polar solvent such as methanol or ethanol.
- Purge the system with inert gas followed by pressurized hydrogen gas, ensuring the pressure is maintained within the optimal range of 1 to 200 atmospheres depending on substrate reactivity.
- Stir the mixture at a controlled temperature between -30°C and 50°C for 5 to 24 hours, then isolate the product via standard purification methods like column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric hydrogenation technology offers substantial strategic benefits beyond mere technical performance. The ability to synthesize optically active alcohols without the need for stoichiometric amounts of organic bases significantly simplifies the downstream processing workflow. Eliminating base neutralization and salt removal steps reduces the consumption of auxiliary chemicals and water, leading to a greener process with a smaller environmental footprint. This efficiency directly contributes to cost reduction in manufacturing by lowering waste disposal costs and shortening the overall production cycle time. Moreover, the high catalytic activity allows for low catalyst loadings, with substrate-to-catalyst ratios reaching up to 100,000 in some embodiments, which drastically reduces the cost associated with precious metals like ruthenium and rhodium.
- Cost Reduction in Manufacturing: The elimination of organic bases and the high efficiency of the catalyst system mean fewer raw materials are consumed per kilogram of product. By avoiding the use of expensive chiral auxiliaries or resolution agents often required in racemic synthesis, the overall material cost is significantly optimized. The process operates under relatively mild conditions, reducing energy consumption for heating or cooling compared to high-temperature alternatives. Additionally, the high selectivity minimizes the formation of by-products, thereby increasing the yield of the desired isomer and reducing the loss of valuable starting materials during purification.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system against various functional groups ensures a consistent supply of intermediates even when substrate sources vary slightly in quality. The ability to use common solvents like methanol and ethanol simplifies logistics and storage requirements, as these are readily available globally. Furthermore, the scalability of the reaction from gram to multi-kilogram scales without significant loss in performance provides confidence in meeting large-volume demands. This reliability is critical for maintaining continuous production schedules and avoiding delays caused by process failures or inconsistent batch quality.
- Scalability and Environmental Compliance: The process generates minimal waste streams, primarily consisting of the solvent which can often be recovered and recycled. The absence of heavy metal contamination issues often associated with other catalytic systems simplifies compliance with strict regulatory standards for residual metals in pharmaceutical products. The high atom economy of direct hydrogenation aligns with green chemistry principles, making it easier to obtain environmental permits and maintain a sustainable manufacturing profile. This scalability ensures that the technology can grow with the demand, supporting the transition from clinical trial materials to commercial production seamlessly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these aspects helps stakeholders make informed decisions about integrating this method into their existing manufacturing workflows.
Q: Why is this base-free hydrogenation method superior for base-sensitive substrates?
A: Conventional transfer hydrogenation often requires organic bases like triethylamine which can degrade base-sensitive functional groups such as alpha-halo ketones or alkynes. This patented method utilizes direct pressurized hydrogenation with specialized metal complexes that operate efficiently without added base, preserving substrate integrity while achieving high enantiomeric excess.
Q: What represents the typical catalyst loading for commercial scale-up?
A: The patent demonstrates exceptional catalytic efficiency with substrate-to-catalyst (S/C) molar ratios ranging from 10 to 100,000. For industrial applications, ratios between 1,000 and 5,000 are frequently achievable while maintaining high yields and stereoselectivity, significantly reducing precious metal costs.
Q: Can this technology handle complex substrates like alkynyl ketones?
A: Yes, the technology is specifically designed to hydrogenate difficult substrates including alkynyl ketones, cyclic ketones, and halogenated ketones. The specific ligand environment around the ruthenium or rhodium center prevents over-reduction of triple bonds while selectively reducing the carbonyl group to the desired chiral alcohol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of production. Our expertise in asymmetric catalysis allows us to adapt the methodologies described in patents like CN1926083B to suit specific client needs, optimizing yields and minimizing costs while maintaining the highest standards of quality and safety.
We invite you to collaborate with us to leverage this advanced technology for your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your target molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and enhance your supply chain resilience. Let us be your partner in turning complex chemical challenges into commercial successes.
