Advanced Asymmetric Hydrogenation for High-Purity Pharmaceutical Intermediates Manufacturing
Introduction to Breakthrough Asymmetric Hydrogenation Technology
The pharmaceutical and fine chemical industries constantly seek robust methodologies for synthesizing chiral building blocks with exceptional optical purity. Patent CN1926083B introduces a transformative preparation method for optically active alcohols utilizing specialized metal complexes as catalysts. This technology addresses critical limitations in traditional asymmetric hydrogenation by enabling the efficient conversion of ketone compounds that were previously difficult to hydrogenate due to base sensitivity. By employing novel ruthenium, rhodium, or iridium complexes under pressurized hydrogen conditions, manufacturers can achieve high yields and superior enantiomeric excess without the mandatory addition of organic bases. This advancement represents a significant leap forward for the reliable pharmaceutical intermediate supplier market, offering a pathway to produce complex chiral alcohols with reduced process complexity and enhanced environmental profiles.
The core innovation lies in the specific structural design of the metal ligands, which facilitate hydrogen transfer mechanisms that do not rely on external base activation. This capability is paramount for processing substrates containing functional groups that degrade under alkaline conditions, such as halogenated ketones or alkynyl ketones. The patent details a versatile system where the metal center coordinates with sulfonamide diamine derivatives or diphosphine ligands to create a highly active catalytic species. For R&D teams focused on process development, this technology offers a powerful tool to expand the scope of accessible chiral intermediates, ensuring that even the most sensitive molecular architectures can be synthesized with the rigorous purity standards required for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active alcohols from carbonyl compounds has relied heavily on asymmetric metal complexes that often necessitate harsh reaction conditions or specific additives that limit substrate scope. For instance, prior art such as Japanese Patent Laid-Open No. 2003-104993 describes methods using BINAP-type ligands which, while effective for simple ketones, frequently result in low yields or poor enantiomeric excess when applied to more complex or sensitive substrates. Furthermore, other established techniques, like those disclosed in Japanese Patent Laid-Open No. 11-322649, strictly require the presence of organic bases such as triethylamine to activate the catalyst. This dependency on basic conditions creates a significant bottleneck for manufacturing processes involving base-labile compounds, leading to decomposition, side reactions, and costly purification steps to remove residual amines. These limitations severely restrict the commercial viability of producing certain high-value intermediates, forcing manufacturers to resort to less efficient resolution methods or multi-step synthetic routes.
The Novel Approach
In stark contrast, the methodology disclosed in CN1926083B eliminates the strict requirement for base addition, thereby unlocking the potential to hydrogenate a broader range of ketone compounds with remarkable efficiency. By utilizing specific ruthenium complexes coordinated with sulfonamide diamine ligands under pressurized hydrogen, the reaction proceeds rapidly even in neutral or slightly acidic environments. This novel approach allows for the successful hydrogenation of substrates like acetylenic ketones and alpha-halo ketones, which would typically decompose in the presence of traditional organic bases. The process achieves high stereoselectivity and yield by optimizing the electronic and steric properties of the ligand environment around the metal center.  This capability translates directly into cost reduction in fine chemical manufacturing by reducing waste generation and simplifying the workup procedure, making it an ideal solution for the commercial scale-up of complex chiral alcohols needed in the production of active pharmaceutical ingredients.
This capability translates directly into cost reduction in fine chemical manufacturing by reducing waste generation and simplifying the workup procedure, making it an ideal solution for the commercial scale-up of complex chiral alcohols needed in the production of active pharmaceutical ingredients.
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Hydrogenation
The catalytic cycle underpinning this technology involves the formation of a highly active metal-hydride species generated from the precursor complex under hydrogen pressure. The general formula (1) described in the patent features a ruthenium center coordinated with a chiral sulfonamide diamine ligand and an aromatic hydrocarbon group, creating a rigid chiral pocket that dictates the stereochemical outcome of the hydrogen transfer. 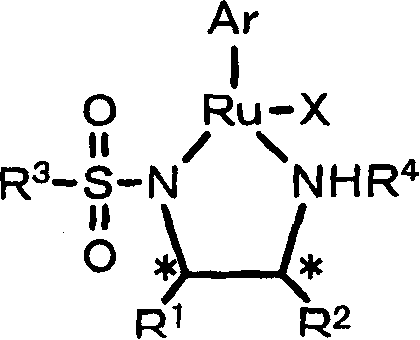 The mechanism likely proceeds through an outer-sphere hydrogen transfer where the hydride and proton are delivered simultaneously to the carbonyl oxygen and carbon, respectively, without the need for substrate coordination to the metal center. This outer-sphere pathway is crucial for maintaining high turnover frequencies and preventing catalyst deactivation by substrate binding. The presence of specific anionic groups and the tunable nature of the aromatic ligands allow for fine-tuning the electronic density at the metal center, optimizing the reaction kinetics for various substrate classes ranging from aromatic ketones to aliphatic diketones.
The mechanism likely proceeds through an outer-sphere hydrogen transfer where the hydride and proton are delivered simultaneously to the carbonyl oxygen and carbon, respectively, without the need for substrate coordination to the metal center. This outer-sphere pathway is crucial for maintaining high turnover frequencies and preventing catalyst deactivation by substrate binding. The presence of specific anionic groups and the tunable nature of the aromatic ligands allow for fine-tuning the electronic density at the metal center, optimizing the reaction kinetics for various substrate classes ranging from aromatic ketones to aliphatic diketones.
Impurity control is inherently managed through the high chemoselectivity of these catalysts, which preferentially reduce the ketone functionality while leaving other sensitive groups intact. The patent highlights that the reaction can be conducted in polar solvents like methanol or ethanol, which stabilize the transition state and facilitate proton transfer. Additionally, the optional addition of inorganic salts, such as perchlorates or tetrafluoroborates, can further accelerate the reaction rate without compromising optical purity. This mechanistic robustness ensures that the resulting optically active alcohols possess stringent purity specifications, minimizing the formation of diastereomers or over-reduced byproducts. For quality assurance teams, this means a more predictable impurity profile and a streamlined analytical validation process, essential for meeting the regulatory requirements of global pharmaceutical markets.
How to Synthesize Optically Active Alcohol Efficiently
The implementation of this asymmetric hydrogenation process follows a straightforward operational protocol designed for scalability and reproducibility in industrial settings. The synthesis begins with the preparation of the catalyst solution, where the specific chiral metal complex is dissolved in a dry, degassed polar solvent under an inert atmosphere to prevent oxidation. Subsequently, the ketone substrate is introduced into the reaction vessel, and the mixture is subjected to pressurized hydrogen gas within the optimized range of 1 to 200 atmospheres. The reaction temperature is carefully maintained between -30°C and 50°C to balance reaction rate and selectivity, with typical completion times ranging from several hours to a day depending on the substrate concentration and catalyst loading.
- Prepare the reaction vessel by charging the specific chiral ruthenium, rhodium, or iridium metal complex catalyst into a pressure-resistant autoclave under inert atmosphere.
- Introduce the target ketone substrate and a polar solvent such as methanol or ethanol into the vessel, optionally adding specific inorganic salts to enhance catalytic activity.
- Pressurize the system with hydrogen gas to the specified range (typically 1-200 atm) and maintain the reaction temperature between -30°C and 50°C until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented hydrogenation technology offers substantial strategic benefits regarding cost efficiency and supply continuity. The elimination of organic bases from the reaction mixture drastically simplifies the downstream processing, removing the need for extensive washing steps or chromatographic separations to remove amine residues. This simplification leads to significant cost savings in manufacturing by reducing solvent consumption, waste disposal costs, and overall processing time. Furthermore, the high catalytic activity allows for low catalyst loadings with high substrate-to-catalyst ratios, reducing the reliance on expensive precious metals and lowering the raw material cost per kilogram of the final product. These factors combined create a more resilient supply chain capable of delivering high volumes of intermediates without the bottlenecks associated with complex purification workflows.
Enhanced supply chain reliability is another critical advantage, as the robustness of the catalyst system ensures consistent batch-to-batch quality and high yields even with varying substrate qualities. The ability to process base-sensitive substrates expands the portfolio of manufacturable intermediates, reducing the risk of supply disruptions caused by the inability to source specific chiral building blocks from external vendors. Moreover, the process is amenable to both batch and continuous flow operations, providing flexibility in production planning and capacity management. From an environmental compliance perspective, the reduction in hazardous waste and the use of greener solvents like alcohols align with increasingly strict global sustainability mandates, enhancing the corporate social responsibility profile of the manufacturing operation and ensuring long-term regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this asymmetric hydrogenation technology based on the detailed disclosures within the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into existing production lines or new product development pipelines. The answers provided reflect the specific experimental data and mechanistic explanations found in the source material, ensuring accuracy and relevance for decision-makers.
Q: What types of ketone substrates are compatible with this hydrogenation method?
A: This method is particularly effective for ketone compounds that are unstable in basic conditions, such as acetylenic ketones, alpha-halo ketones, and cyclic ketones, which traditionally suffer from low yields or decomposition.
Q: Is the addition of an organic base required for the reaction to proceed?
A: No, unlike conventional methods that rely on organic bases like triethylamine, this patented process operates efficiently without mandatory base addition, significantly simplifying the downstream purification process.
Q: Which metal centers are utilized in the described catalyst systems?
A: The patent discloses the use of asymmetric complexes based on Ruthenium (Ru), Rhodium (Rh), and Iridium (Ir), coordinated with specific sulfonamide diamine or diphosphine ligands to achieve high stereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Alcohol Supplier
At NINGBO INNO PHARMCHEM, we leverage cutting-edge technologies like the asymmetric hydrogenation methods described in CN1926083B to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of optically active alcohol meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence allows us to tackle complex synthetic challenges, providing our clients with reliable access to high-quality chiral intermediates that drive their drug development programs forward.
We invite you to collaborate with us to explore how this advanced catalytic technology can optimize your specific manufacturing needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating the potential economic benefits of switching to this base-free hydrogenation route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain efficiency and product quality. Partner with NINGBO INNO PHARMCHEM to secure a sustainable and cost-effective source for your critical chiral building blocks.
