Advancing Benzylamine Production: A Green Photocatalytic Hydrogenation Strategy for Industrial Scale
Advancing Benzylamine Production: A Green Photocatalytic Hydrogenation Strategy for Industrial Scale
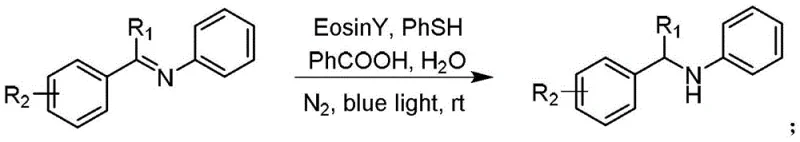
The landscape of organic synthesis is undergoing a paradigm shift towards sustainability, driven by the urgent need to reduce environmental footprints and operational costs in chemical manufacturing. Patent CN110305025B introduces a groundbreaking methodology for the synthesis of benzylamine compounds, a class of vital intermediates in the pharmaceutical and agrochemical industries. This innovation leverages visible light photocatalysis to achieve the hydrogenation of imines under mild conditions, utilizing water as a solvent and inexpensive organic dyes as catalysts. By replacing traditional high-pressure hydrogenation or stoichiometric hydride reductions with this photo-driven approach, the technology addresses critical pain points regarding safety, cost, and ecological impact. For industry leaders seeking a reliable pharmaceutical intermediates supplier, understanding this technological leap is essential for future-proofing supply chains and enhancing product purity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of imines to amines has relied heavily on stoichiometric reducing agents such as sodium borohydride or lithium aluminum hydride, which pose significant safety hazards due to their reactivity and the generation of hazardous waste streams. Alternatively, transition metal-catalyzed hydrogenation using rhodium, ruthenium, or palladium has been the standard for high-value synthesis, yet these methods are plagued by the high cost of precious metals and the stringent requirement for their complete removal from the final active pharmaceutical ingredient (API). The presence of trace metal residues can lead to costly purification steps and regulatory compliance issues, creating bottlenecks in the commercial scale-up of complex organic intermediates. Furthermore, traditional methods often require high pressures of hydrogen gas or elevated temperatures, increasing the energy consumption and operational risk profile of the manufacturing process.
The Novel Approach
In stark contrast, the method disclosed in CN110305025B utilizes a metal-free organocatalytic system driven by visible light, specifically employing catalysts like Eosin Y or Rhodamine B. This approach operates at room temperature and atmospheric pressure, utilizing thiophenol as a hydrogen source and weak acids as co-catalysts to facilitate the transformation. The ability to use water as a reaction solvent represents a monumental shift towards green chemistry, eliminating the need for large volumes of volatile organic solvents. This novel pathway not only simplifies the reaction setup by allowing for arbitrary feeding sequences but also ensures that the resulting benzylamine derivatives are free from toxic metal contaminants, thereby streamlining downstream processing and quality control measures for high-purity benzylamine derivatives.
Mechanistic Insights into Visible Light Photocatalytic Hydrogenation
The core of this innovation lies in the photophysical properties of the organic catalyst, such as Eosin Y, which absorbs blue light to reach an excited state capable of engaging in single electron transfer (SET) processes. Upon irradiation, the excited catalyst interacts with the imine substrate and the thiophenol hydrogen donor, initiating a radical cascade that ultimately results in the reduction of the carbon-nitrogen double bond. This mechanism avoids the formation of metal-hydride species typical of traditional catalysis, instead relying on proton-coupled electron transfer (PCET) pathways that are highly efficient under mild conditions. The use of a weak acid co-catalyst further modulates the redox potentials involved, ensuring high conversion rates while maintaining the integrity of sensitive functional groups present on the aromatic rings of the substrate.
Impurity control is inherently superior in this system due to the high chemoselectivity of the photocatalytic cycle. Unlike harsh hydride reagents that may reduce other reducible functionalities indiscriminately, the visible light-driven process targets the imine bond with precision. The radical intermediates generated during the reaction are short-lived and efficiently quenched by the hydrogen source, minimizing side reactions such as polymerization or over-reduction. This selectivity is crucial for R&D directors focusing on purity and impurity profiles, as it reduces the burden on purification teams and ensures a cleaner crude product profile before final isolation. The robustness of the catalyst under aqueous conditions further prevents hydrolysis side reactions that might occur in acidic or basic traditional reduction environments.
How to Synthesize Benzylamine Derivatives Efficiently
The practical implementation of this synthesis route is designed for ease of operation, requiring standard laboratory equipment equipped with a blue LED light source and a nitrogen inlet. The protocol involves mixing the imine starting material with thiophenol, the organic photocatalyst, and a carboxylic acid co-catalyst in water or a compatible organic solvent. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the imine substrate, thiophenol as the hydrogen source, an organic visible light catalyst such as Eosin Y, and a weak acid co-catalyst in a reaction vessel.
- Purge the system with nitrogen gas to establish an inert atmosphere and add water or an organic solvent as the reaction medium.
- Irradiate the reaction mixture with blue LED light at room temperature for 24 to 48 hours, followed by extraction and purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers transformative benefits that extend beyond mere technical feasibility. The elimination of expensive transition metals like palladium and iridium directly translates to substantial cost savings in raw material procurement, shielding the production budget from the volatility of precious metal markets. Additionally, the use of water as a primary solvent drastically reduces the costs associated with solvent purchase, recovery, and hazardous waste disposal, contributing to a leaner and more sustainable manufacturing model. The simplified reaction conditions also mean lower energy consumption, as there is no need for high-pressure reactors or extensive heating, further driving down the operational expenditure (OPEX) for cost reduction in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with abundant organic dyes like Eosin Y removes a major cost driver from the bill of materials. This shift eliminates the need for specialized metal scavenging resins and extensive analytical testing for metal residues, which are traditionally expensive and time-consuming. Consequently, the overall production cost per kilogram is significantly optimized, allowing for more competitive pricing strategies in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reagents required for this process, including thiophenol and benzoic acid, are commodity chemicals with stable and robust supply chains, reducing the risk of production delays caused by raw material shortages. The simplicity of the reaction setup, which does not require specialized high-pressure equipment, allows for greater flexibility in manufacturing site selection and capacity expansion. This resilience ensures consistent delivery schedules and reduces lead time for high-purity benzylamine derivatives, fostering stronger relationships with downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the use of flow chemistry principles compatible with LED irradiation, ensuring uniform light penetration and reaction efficiency. The aqueous nature of the reaction aligns perfectly with increasingly stringent environmental regulations regarding VOC emissions and heavy metal discharge. By adopting this green chemistry approach, manufacturers can future-proof their operations against regulatory changes and enhance their corporate social responsibility (CSR) profiles through demonstrable reductions in environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the primary advantages of using Eosin Y over transition metal catalysts for imine reduction?
A: Eosin Y is an organic dye that eliminates the risk of heavy metal contamination in the final product, which is critical for pharmaceutical applications. It is also significantly more cost-effective and environmentally benign compared to precious metals like palladium or rhodium.
Q: Can this photocatalytic method tolerate diverse functional groups on the imine substrate?
A: Yes, the method demonstrates broad substrate scope, successfully reducing imines with electron-donating groups like methoxy and electron-withdrawing groups like bromo and iodo, as well as bulky naphthyl substituents.
Q: Is water a viable solvent for this hydrogenation reaction?
A: Absolutely. The patent specifically highlights the use of water as a primary solvent, which drastically reduces volatile organic compound (VOC) emissions and simplifies waste treatment processes compared to traditional organic solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic technologies in modernizing the production of key pharmaceutical building blocks. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the absence of metal residues and confirm the structural integrity of every batch of benzylamine intermediates we produce.
We invite forward-thinking partners to collaborate with us to leverage this green synthesis route for their specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive efficiency and value for your supply chain.
