Scalable Synthesis of Novel N-N Bridged Beta-Carboline Isatin Derivatives for Oncology Applications
Scalable Synthesis of Novel N-N Bridged Beta-Carboline Isatin Derivatives for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds to overcome drug resistance in oncology, and recent intellectual property disclosures have highlighted a promising class of hybrid molecules. Specifically, patent CN116253732A details the invention of a beta-carboline derivative featuring an N-N bridged isatin unit, representing a significant advancement in medicinal chemistry for antitumor applications. This novel compound class is obtained by chemically connecting a beta-carboline core with an isatin derivative through a hydrazone linkage, resulting in structures that demonstrate superior biological activity compared to their individual components. The strategic combination of these two privileged heterocyclic systems offers a unique opportunity for drug discovery teams to explore new mechanisms of action while leveraging established synthetic pathways. As a leading manufacturer, we recognize the immense potential of these intermediates in the development of next-generation kinase inhibitors and cytotoxic agents.
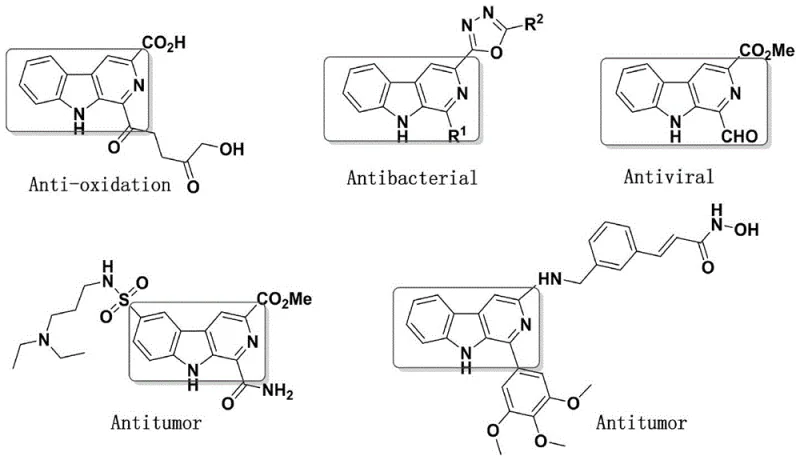
The structural versatility of these derivatives allows for extensive structure-activity relationship (SAR) studies, particularly through modifications at the 1, 3, and 9 positions of the beta-carboline ring. The patent explicitly outlines that introducing suitable substituents at these positions can further enhance antitumor efficacy, providing a clear roadmap for optimization. For R&D directors focused on purity and impurity profiles, understanding the precise connectivity and stereochemistry of these hybrids is crucial. The N-N bridge serves not only as a linker but also as a potential metabolic stability modulator, which is a critical parameter in early-stage drug design. By integrating the planar aromatic system of the beta-carboline with the hydrogen-bonding capability of the isatin oxindole ring, these molecules are engineered to interact more effectively with biological targets involved in cell proliferation and apoptosis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex heterocyclic hybrids often suffers from multi-step sequences that require harsh reaction conditions, expensive transition metal catalysts, and difficult purification protocols. Conventional methods for linking indole-based scaffolds frequently rely on cross-coupling reactions that generate significant amounts of heavy metal waste, posing challenges for environmental compliance and cost control in large-scale manufacturing. Furthermore, older synthetic routes to functionalized beta-carbolines often involve low-yielding cyclization steps or require protecting group strategies that add unnecessary complexity and reduce overall atom economy. These inefficiencies lead to higher production costs and longer lead times, which are critical bottlenecks for procurement managers aiming to secure reliable supplies of high-purity pharmaceutical intermediates. The reliance on toxic solvents and difficult-to-remove impurities in legacy processes also complicates the regulatory filing process for new drug applications.
The Novel Approach
In contrast, the methodology described in the patent utilizes a streamlined condensation strategy that significantly simplifies the production workflow. The novel approach involves a direct nucleophilic addition-condensation reaction between an isatin hydrazone intermediate and a formyl-substituted beta-carboline. This reaction proceeds efficiently in ethanol, a green and cost-effective solvent, often requiring only mild heating and catalytic amounts of acetic acid. By avoiding the use of precious metal catalysts and minimizing the number of isolation steps, this route offers substantial cost savings and improved environmental sustainability. The high yields reported for the final condensation step indicate a robust process that is well-suited for commercial scale-up. This efficiency translates directly into supply chain reliability, ensuring that downstream drug developers can access these critical intermediates without the delays typically associated with complex custom synthesis projects.
Mechanistic Insights into Hydrazone Condensation and Precursor Synthesis
The core of this synthetic innovation lies in the efficient construction of the beta-carboline aldehyde precursor and its subsequent coupling with the isatin unit. The synthesis of the key 1-substituted-9-substituted-3-formyl-beta-carboline intermediate begins with L-Tryptophan, undergoing a Pictet-Spengler type cyclization followed by aromatization. This sequence is critical for establishing the rigid tricyclic core essential for biological activity. The subsequent functionalization at the 9-position via alkylation allows for the modulation of lipophilicity and solubility, which are key pharmacokinetic parameters. The final oxidation of the hydroxymethyl group to the aldehyde is performed using manganese dioxide, a selective oxidant that preserves the integrity of the sensitive indole nitrogen. This careful selection of reagents ensures a clean reaction profile with minimal byproduct formation, facilitating easier purification and higher final purity.
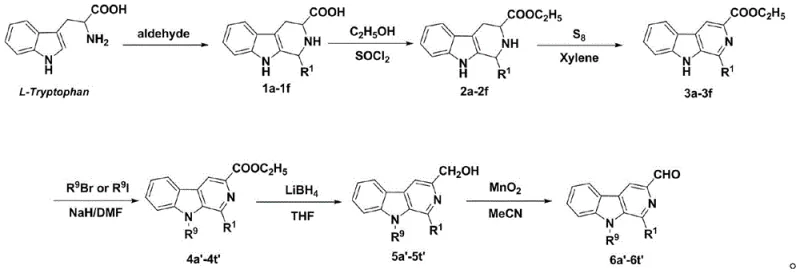
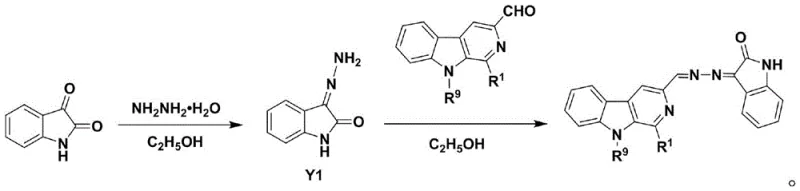
The final coupling mechanism involves the nucleophilic attack of the hydrazine nitrogen on the carbonyl carbon of the beta-carboline aldehyde. This condensation reaction is driven by the elimination of water, forming a stable C=N double bond that links the two pharmacophores. The use of glacial acetic acid as a catalyst protonates the carbonyl oxygen, increasing its electrophilicity and accelerating the reaction rate without degrading the sensitive heterocyclic rings. The resulting hydrazone linkage is planar and conjugated, which may contribute to the molecule's ability to intercalate DNA or inhibit topoisomerases, common mechanisms for beta-carboline-based antitumor agents. Understanding this mechanistic pathway is vital for process chemists aiming to optimize reaction parameters such as temperature and stoichiometry to maximize yield and minimize impurity generation during scale-up operations.
How to Synthesize Beta-Carboline Isatin Derivatives Efficiently
The preparation of these high-value antitumor intermediates follows a logical and scalable sequence that begins with readily available amino acids. The process is designed to be operationally simple, utilizing standard laboratory equipment that can be easily adapted for pilot plant and commercial production. The initial steps focus on building the substituted beta-carboline scaffold, followed by the preparation of the isatin hydrazone, and finally, the convergence of these two fragments. Detailed standard operating procedures for each transformation, including specific molar ratios, solvent volumes, and workup protocols, are essential for ensuring batch-to-batch consistency. For technical teams looking to implement this chemistry, adhering to the specified reaction times and temperatures is critical to achieving the high yields and purity levels reported in the patent literature.
- Synthesize 1-substituted-9-substituted-3-formyl-beta-carboline precursors starting from L-Tryptophan via Pictet-Spengler cyclization, esterification, sulfur-mediated aromatization, and selective oxidation.
- Prepare isatin-3-hydrazone intermediate by reacting isatin with hydrazine hydrate in ethanol under reflux conditions.
- Perform the final condensation reaction between the formyl-beta-carboline and isatin-3-hydrazone in ethanol with glacial acetic acid catalyst to yield the target N-N bridged derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route outlined in this patent offers distinct advantages regarding raw material sourcing and cost management. The primary starting material, L-Tryptophan, is a bulk commodity chemical produced on a massive scale for the food and feed industries, ensuring a stable and abundant supply with minimal price volatility. This contrasts sharply with routes that depend on exotic or proprietary building blocks that are subject to supply chain disruptions. Additionally, the use of ethanol as the primary solvent for the key coupling step eliminates the need for expensive, hazardous, or regulated solvents, thereby reducing waste disposal costs and improving the overall safety profile of the manufacturing facility. These factors combine to create a highly cost-effective production model that aligns with the strategic goals of reducing manufacturing expenses in the pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of inexpensive reagents like hydrazine hydrate and manganese dioxide significantly lower the bill of materials. Furthermore, the high conversion rates observed in the condensation step reduce the burden on downstream purification processes, such as chromatography, which are often the most expensive part of fine chemical production. By simplifying the workflow and minimizing the number of unit operations, manufacturers can achieve substantial operational efficiencies that translate into lower unit costs for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard solvents means that the supply chain is less vulnerable to geopolitical risks or single-source supplier failures. The robustness of the chemistry allows for flexible production scheduling, enabling manufacturers to respond quickly to fluctuations in market demand. This reliability is crucial for pharmaceutical companies that require just-in-time delivery of intermediates to maintain their own clinical trial timelines and commercial launch schedules without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot batches and eventually to ton-scale commercial production. The generation of waste is minimized through high atom economy and the use of recyclable solvents, helping companies meet increasingly stringent environmental regulations. The absence of heavy metals in the final product simplifies the regulatory approval process, as there is no need for extensive testing and validation of residual metal limits, accelerating the time to market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel beta-carboline derivatives. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity for stakeholders evaluating this technology for their pipeline. Understanding the nuances of the synthesis and the biological profile of these compounds is essential for making informed decisions about licensing, partnership, or internal development strategies.
Q: What is the primary biological activity of these N-N bridged beta-carboline derivatives?
A: According to patent CN116253732A, these novel compounds exhibit significant in vitro antitumor activity against various cancer cell lines, including A549, BGC-823, and MCF-7, making them promising candidates for oncology drug development.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the synthesis route utilizes widely available commodity chemicals such as L-Tryptophan, Isatin, and common solvents like ethanol and DMF, ensuring a robust and reliable supply chain for large-scale manufacturing.
Q: How does the N-N bridging strategy improve drug efficacy?
A: The N-N bridging connects the pharmacophoric beta-carboline core with the bioactive isatin unit, creating a hybrid structure that leverages the antitumor properties of both moieties, often resulting in enhanced potency compared to single scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Carboline Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure required to bring complex synthetic routes like this from the laboratory bench to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand that the successful development of antitumor drugs depends on the availability of high-purity intermediates, and our rigorous QC labs are equipped to verify stringent purity specifications for every batch we produce. By partnering with us, you gain access to a dedicated resource that prioritizes quality, safety, and regulatory compliance throughout the entire manufacturing lifecycle.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for these novel isatin-beta-carboline hybrids, we are ready to provide the detailed information you need. Let us collaborate to accelerate the development of life-saving oncology therapies by ensuring a secure and efficient supply of these critical pharmaceutical intermediates.
