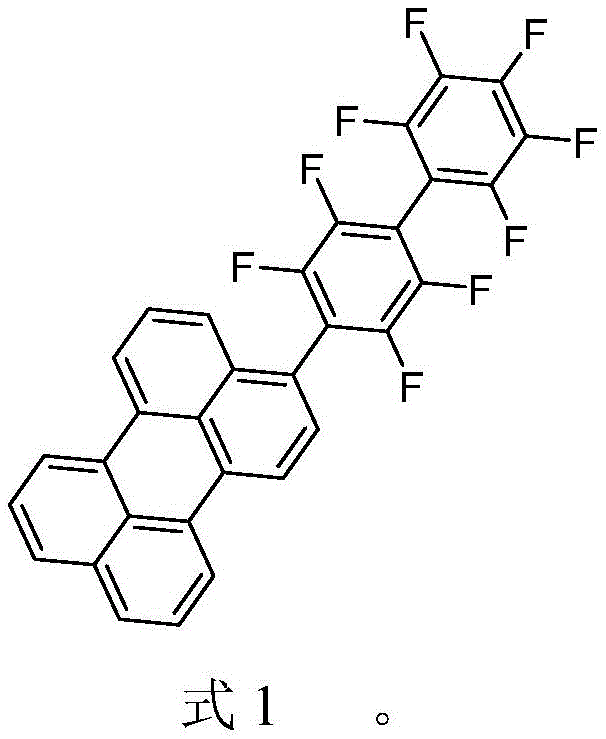
Advanced Synthesis of Fluorinated Perylene Derivatives for Next-Generation Green LED Applications
The rapid evolution of solid-state lighting technologies has driven an urgent demand for high-performance organic photoelectric materials that can surpass the limitations of traditional inorganic phosphors. Patent CN112194563B introduces a groundbreaking advancement in this sector by disclosing a novel compound containing perylene and fluorobenzene, specifically designed to function as a high-color-purity green fluorescent powder for white light LEDs. This innovation addresses critical stability and efficiency challenges inherent in earlier generations of organic emitters by strategically incorporating strong electron-withdrawing fluorine substituents onto the perylene core. For R&D directors and procurement specialists in the electronic materials sector, this patent represents a significant opportunity to access next-generation display and optoelectronic materials that offer superior quantum yields and enhanced environmental stability. The disclosed synthesis route provides a robust framework for the commercial scale-up of complex organic intermediates, ensuring a reliable supply chain for manufacturers aiming to integrate advanced green phosphors into their LED device architectures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to generating green light in LED devices have predominantly relied on inorganic phosphor systems such as sulfides, aluminates, phosphates, and silicates. While these materials have served the industry well, they often suffer from intrinsic drawbacks including complex synthesis requirements, the use of rare earth elements, and potential environmental concerns regarding heavy metal content. Furthermore, achieving high color purity with these inorganic systems can be challenging due to broad emission spectra that limit the color gamut of the final display or lighting product. Organic alternatives have historically struggled with poor air stability and low quantum efficiency in the solid state, primarily due to aggregation-caused quenching and susceptibility to oxidation. These factors have hindered the widespread adoption of pure organic non-nitrogen oxides as viable replacements for inorganic green phosphors in commercial solid-state lighting applications, creating a gap in the market for stable, high-efficiency organic emitters.
The Novel Approach
The methodology outlined in the patent data presents a sophisticated solution by modifying the perylene backbone with multiple fluorine atoms at non-bay positions. This strategic structural modification leverages the high electronegativity of fluorine to lower the Lowest Unoccupied Molecular Orbital (LUMO) energy level of the derivative, effectively preventing oxidation and significantly improving air stability compared to unmodified perylene compounds. Additionally, the introduction of these fluorinated groups enhances the molecular polarity, which improves solubility and processability during device fabrication. The resulting Compound IV demonstrates a narrow emission band with high color purity, directly addressing the spectral limitations of conventional phosphors. By utilizing a stepwise Suzuki-Miyaura cross-coupling strategy, the synthesis allows for precise control over the molecular architecture, ensuring consistent batch-to-bquality essential for industrial LED manufacturing.
Mechanistic Insights into Suzuki-Miyaura Cross-Coupling Strategy
The core of this synthetic innovation lies in the sequential application of palladium-catalyzed cross-coupling reactions to construct the extended conjugated system. The process begins with the selective bromination of perylene using N-bromosuccinimide (NBS) in DMF, followed by a borylation step to install the pinacol boronate ester functionality. This intermediate then undergoes a critical coupling with 2,3,5,6-tetrafluoro-1,4-dibromobenzene, facilitated by tetrakis(triphenylphosphine)palladium and anhydrous potassium carbonate in DMF at elevated temperatures between 80°C and 120°C. The final step involves a second Suzuki coupling with pentafluorophenylboronic acid, completing the assembly of the highly fluorinated architecture. Each catalytic cycle relies on the oxidative addition of the aryl halide to the Pd(0) species, transmetallation with the boronate, and reductive elimination to forge the carbon-carbon bonds, a mechanism that ensures high regioselectivity and minimizes the formation of homocoupling byproducts.
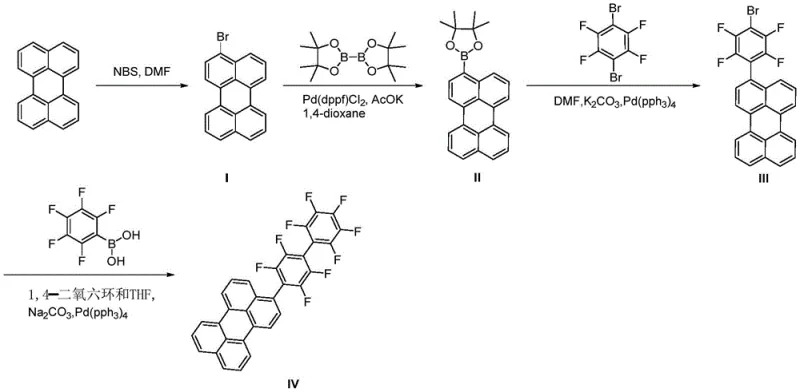
Impurity control in this multi-step synthesis is paramount for achieving the high optical performance required for LED applications. The use of specific ligands such as triphenylphosphine and rigorous purification protocols, including column chromatography with defined eluent systems like petroleum ether and ethyl acetate, are critical for removing residual palladium catalysts and unreacted starting materials. Residual transition metals can act as quenching sites that drastically reduce fluorescence quantum yield, so the detailed workup procedures involving aqueous washes and drying over anhydrous sodium sulfate are essential operational parameters. Furthermore, the choice of solvents like 1,4-dioxane and THF in the final steps optimizes the solubility of the highly fluorinated intermediates, preventing premature precipitation that could trap impurities within the crystal lattice. This meticulous attention to reaction conditions and purification ensures that the final Compound IV meets the stringent purity specifications necessary for high-efficiency optoelectronic devices.
How to Synthesize Fluorinated Perylene Derivative Efficiently
The synthesis of Compound IV requires precise control over stoichiometry and reaction environments to maximize yield and purity across the four distinct transformation stages. The process initiates with the activation of the perylene core, followed by the installation of boron handles and subsequent iterative coupling with fluorinated aromatic building blocks. Maintaining an inert nitrogen atmosphere throughout the palladium-catalyzed steps is crucial to prevent catalyst deactivation and ensure consistent reaction kinetics. Detailed standardized operating procedures for temperature ramping, reagent addition rates, and isolation techniques are vital for reproducibility. For a comprehensive breakdown of the specific molar ratios, solvent volumes, and purification gradients required to execute this synthesis at scale, please refer to the technical guide below.
- Bromination of perylene using N-bromosuccinimide (NBS) in DMF at 20-30°C to yield 3-bromoperylene (Compound I).
- Borylation of Compound I with bis(pinacolato)diboron using Pd(dppf)Cl2 catalyst in 1,4-dioxane at 80-120°C to form Compound II.
- First Suzuki coupling of Compound II with 2,3,5,6-tetrafluoro-1,4-dibromobenzene using Pd(PPh3)4 and K2CO3 in DMF to generate Compound III.
- Final Suzuki coupling of Compound III with pentafluorophenylboronic acid using Pd(PPh3)4 and Na2CO3 in 1,4-dioxane/THF to obtain Compound IV.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel fluorinated perylene derivative offers substantial strategic benefits regarding cost structure and supply reliability. The synthetic route utilizes readily available commodity chemicals such as perylene, N-bromosuccinimide, and common fluorinated benzenes, which mitigates the risk of raw material shortages often associated with rare-earth-dependent inorganic phosphors. By eliminating the need for complex high-temperature calcination processes typical of ceramic phosphor production, the manufacturing footprint is significantly reduced, leading to lower energy consumption and capital expenditure requirements. This streamlined organic synthesis pathway facilitates faster time-to-market for new LED products and allows for more agile responses to fluctuating market demands without the long lead times associated with mining and refining inorganic precursors.
- Cost Reduction in Manufacturing: The elimination of expensive rare-earth elements and the use of standard palladium catalysis systems significantly lowers the raw material cost base per kilogram of active phosphor. Furthermore, the ability to purify the product using standard chromatographic techniques rather than energy-intensive zone refining or high-temperature sintering reduces overall utility costs. The high quantum yield of the material means less active material is required per LED device to achieve the same luminous output, effectively reducing the bill of materials for the final lighting assembly. These factors combine to create a compelling economic case for switching from traditional inorganic systems to this advanced organic alternative in high-volume manufacturing scenarios.
- Enhanced Supply Chain Reliability: Sourcing fluorinated organic intermediates is generally more stable and predictable than relying on the geopolitical supply chains of rare earth metals. The synthesis relies on established chemical feedstocks that are produced globally in large quantities, ensuring continuity of supply even during regional disruptions. Additionally, the modular nature of the Suzuki coupling chemistry allows for potential process intensification or continuous flow manufacturing adaptations, which can further de-risk production bottlenecks. This robustness ensures that downstream LED manufacturers can maintain consistent production schedules without fear of raw material allocation issues that frequently plague the inorganic phosphor market.
- Scalability and Environmental Compliance: The solution-phase synthesis operates at moderate temperatures and pressures, making it inherently safer and easier to scale from pilot plant to multi-ton production compared to solid-state reactions requiring extreme heat. The waste streams generated are primarily organic solvents which can be recovered and recycled using standard distillation infrastructure, aligning with increasingly strict environmental regulations regarding industrial emissions. Avoiding the use of toxic heavy metals like cadmium or lead, which are sometimes present in alternative semiconductor nanocrystals, simplifies regulatory compliance and end-of-life disposal for consumer electronics, enhancing the overall sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorinated perylene technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and processing requirements. Understanding these details is essential for engineering teams evaluating the integration of Compound IV into existing LED packaging lines.
Q: How does fluorine substitution improve the stability of perylene derivatives?
A: The introduction of multiple fluorine atoms, particularly the pentafluorophenyl group, significantly lowers the LUMO energy level due to the strong electron-withdrawing nature of fluorine. This reduction in LUMO energy makes the compound less susceptible to oxidation, thereby enhancing its air stability compared to traditional non-fluorinated perylene film-forming materials.
Q: What is the primary application potential of Compound IV?
A: Compound IV is designed specifically for use as a green phosphor in white light LEDs. It exhibits high color purity green light emission when excited by blue LED chips (470nm), making it a superior alternative to traditional inorganic sulfide or aluminate phosphors for solid-state lighting applications.
Q: What are the key reaction conditions for the final coupling step?
A: The final synthesis step involves reacting Compound III with pentafluorophenylboronic acid in a solvent mixture of 1,4-dioxane and THF. The reaction utilizes sodium carbonate and tetrakis(triphenylphosphine)palladium as catalysts under nitrogen protection at temperatures ranging from 80°C to 120°C for 16 to 32 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perylene Derivative Supplier
As the global demand for high-color-purity green LED materials continues to surge, partnering with an experienced CDMO capable of navigating complex organic synthesis is critical for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to mass market deployment is seamless. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, essential for maintaining the high quantum efficiency and color stability required in premium optoelectronic applications. We understand the nuances of handling fluorinated intermediates and palladium catalysts, allowing us to optimize yield and minimize impurity profiles effectively.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into the potential economic benefits of adopting this fluorinated perylene derivative in your product line. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower your R&D and supply chain teams to make informed decisions about integrating this next-generation electronic chemical into your manufacturing portfolio.
