Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Scalable Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Scalable Pharmaceutical Manufacturing
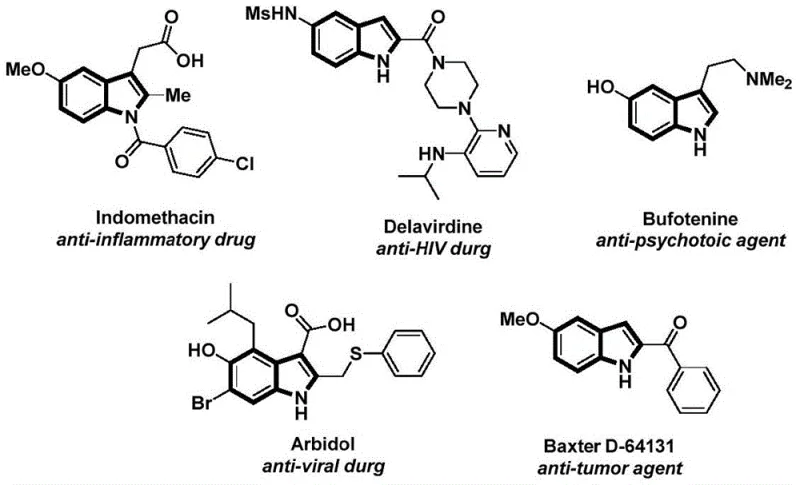
The structural motif of indole represents a cornerstone in modern medicinal chemistry, serving as the fundamental backbone for a vast array of bioactive molecules including anti-inflammatory, anti-tumor, and anti-viral agents. Recent advancements in synthetic methodology have focused on streamlining the construction of these complex heterocycles to meet the rigorous demands of the pharmaceutical industry. A particularly significant development is detailed in Chinese Patent CN112898192B, which discloses a robust and efficient preparation method for N-acyl indole compounds. This technology leverages a palladium-catalyzed carbonylation cyclization strategy that utilizes readily available 2-alkynylanilines and aryl iodides as starting materials. By employing a solid carbon monoxide surrogate rather than hazardous gaseous CO, this innovation addresses critical safety and operational challenges inherent in traditional carbonylation processes, positioning it as a highly attractive route for the industrial production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles has often relied on methodologies that present significant hurdles for large-scale manufacturing and process safety. Traditional carbonylation reactions frequently necessitate the use of carbon monoxide gas, which is not only highly toxic but also requires specialized high-pressure equipment and stringent safety protocols to prevent leaks and exposure. Furthermore, many existing routes involve multi-step sequences with harsh reaction conditions, such as strong acids or elevated temperatures, which can lead to poor functional group tolerance and the generation of complex impurity profiles. These factors collectively increase the cost of goods sold (COGS) and extend the lead time for process development, making it difficult for supply chain managers to ensure consistent quality and availability of these critical building blocks for drug discovery and development pipelines.
The Novel Approach
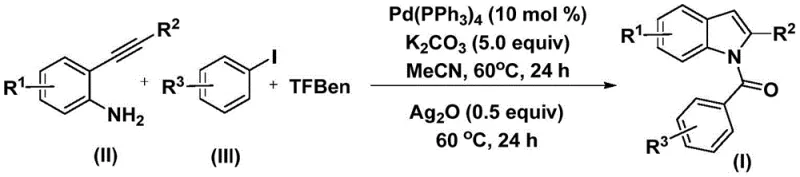
In stark contrast to these legacy methods, the technology described in patent CN112898192B introduces a streamlined, one-pot two-step protocol that operates under remarkably mild conditions. The core innovation lies in the use of 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe, solid carbon monoxide substitute, which releases CO in situ under the reaction conditions. This eliminates the need for gas cylinders and high-pressure reactors, drastically simplifying the engineering requirements for scale-up. The reaction proceeds in acetonitrile at a moderate temperature of 60°C, utilizing a palladium catalyst system that demonstrates excellent compatibility with a wide range of substituents, including halogens, alkyl groups, and alkoxy moieties. This approach not only enhances operational safety but also improves the overall atom economy and step efficiency, providing a direct pathway to diverse N-acyl indole derivatives with high purity.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps orchestrated by the palladium catalyst. Initially, the catalytic cycle commences with the oxidative addition of the aryl iodide to the zero-valent palladium species, generating an aryl-palladium(II) intermediate. Subsequently, carbon monoxide, released from the decomposition of the TFBen surrogate, inserts into the palladium-carbon bond to form an acyl-palladium complex. This electrophilic acyl species then undergoes nucleophilic attack by the amino group of the 2-alkynylaniline, followed by reductive elimination to yield an intermediate amide compound. This precise control over the carbonylation step ensures that the acyl group is installed regioselectively, minimizing the formation of side products that are common in less controlled Friedel-Crafts type acylations.
The final stage of the synthesis is equally critical, involving an oxidative cyclization mediated by silver oxide (Ag2O). Upon addition of Ag2O in the second step, the intermediate amide undergoes an intramolecular cyclization where the silver species likely facilitates the activation of the alkyne moiety or assists in the deprotonation necessary for ring closure. This results in the formation of the rigid indole core with the N-acyl substituent intact. The use of silver oxide as a distinct additive in the second stage allows for temporal separation of the carbonylation and cyclization events, which is key to suppressing competing polymerization of the alkyne or hydrolysis of the acyl group. This mechanistic understanding underscores the robustness of the method, as the mild thermal conditions (60°C) prevent thermal degradation of sensitive functional groups, ensuring a clean impurity profile suitable for pharmaceutical applications.
How to Synthesize N-Acyl Indole Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling in a standard laboratory or pilot plant setting. The process begins by charging a reaction vessel with the palladium catalyst, specifically tetrakis(triphenylphosphine)palladium, along with potassium carbonate as the base and the solid CO source TFBen. To this mixture, the 2-alkynylaniline and the chosen aryl iodide are added in acetonitrile solvent. The reaction is heated to 60°C and maintained for 24 hours to allow the initial carbonylation and amide formation to reach completion. Following this incubation period, silver oxide is introduced directly into the reaction mixture without the need for intermediate isolation or workup. The heating is continued at 60°C for an additional 24 hours to drive the cyclization to completion. Detailed standardized synthetic steps for specific derivatives are provided in the guide below.
- Combine Pd(PPh3)4 catalyst, K2CO3 base, TFBen CO source, 2-alkynylaniline, and aryl iodide in acetonitrile solvent.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide (Ag2O) to the mixture and continue heating at 60°C for another 24 hours to induce cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage stems from the substitution of gaseous carbon monoxide with a stable solid surrogate, which fundamentally alters the risk profile and infrastructure requirements of the manufacturing process. By removing the need for high-pressure gas handling systems, facilities can reduce capital expenditure on specialized reactor vessels and safety monitoring equipment. This simplification translates directly into lower overhead costs and faster turnaround times for campaign scheduling, as the regulatory burden associated with toxic gas storage is significantly mitigated. Furthermore, the use of commercially available starting materials like aryl iodides and 2-alkynylanilines ensures a stable and diversified supply base, reducing the risk of raw material shortages that can disrupt production timelines.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the high atom efficiency and the elimination of expensive protecting group strategies often required in alternative indole syntheses. Since the reaction tolerates a broad spectrum of functional groups without the need for extensive protection and deprotection sequences, the total number of processing steps is minimized. This reduction in unit operations leads to substantial savings in solvent consumption, energy usage, and labor hours. Additionally, the mild reaction conditions reduce the thermal load on the facility, contributing to lower utility costs. The ability to achieve high conversion rates with relatively low catalyst loading further optimizes the cost structure, making the production of these high-value intermediates more economically sustainable in a competitive market environment.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on solid reagents rather than compressed gases simplifies transportation and storage protocols. Solid CO surrogates like TFBen can be shipped and stored under ambient conditions without the stringent regulations governing hazardous gases, thereby reducing lead times for raw material procurement. The robustness of the reaction conditions also implies a wider operating window, which increases the reliability of batch-to-batch consistency. This predictability is crucial for maintaining continuous supply to downstream API manufacturers, ensuring that production schedules are met without unexpected delays caused by process deviations or safety incidents. The method's compatibility with standard glass-lined or stainless steel reactors further ensures that it can be implemented across a wide range of existing manufacturing assets without major retrofitting.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is favorably aligned with modern green chemistry principles, primarily due to the avoidance of toxic gas emissions and the use of acetonitrile, a solvent that is readily recoverable and recyclable. The high selectivity of the palladium-catalyzed system minimizes the generation of heavy metal waste and organic byproducts, simplifying the wastewater treatment process. As regulatory pressures on pharmaceutical manufacturing continue to intensify, having a process that inherently produces less hazardous waste provides a significant compliance advantage. The scalability of the method is evidenced by its successful application across a diverse library of substrates, suggesting that the kinetics and heat transfer characteristics are well-suited for translation from gram-scale laboratory experiments to multi-kilogram commercial production runs.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this N-acyl indole synthesis technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, aimed at clarifying the operational parameters for potential licensees or contract manufacturing partners. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing process portfolios.
Q: What is the role of Silver Oxide (Ag2O) in this synthesis?
A: Silver oxide acts as a crucial additive in the second stage of the reaction, facilitating the oxidative cyclization of the intermediate amide to form the final N-acyl indole structure efficiently.
Q: Why is 1,3,5-tricarboxylic acid phenol ester (TFBen) used instead of CO gas?
A: TFBen serves as a solid carbon monoxide surrogate, eliminating the safety hazards and specialized equipment required for handling toxic high-pressure CO gas, thereby simplifying the operational protocol.
Q: What is the typical yield range for this N-acyl indole preparation method?
A: According to the patent data, the reaction demonstrates good substrate compatibility with isolated yields ranging generally from moderate to high efficiency, specifically observed between 44% and 82% across various substituted derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the acceleration of drug development programs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering N-acyl indole compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in palladium-catalyzed transformations allows us to optimize this specific carbonylation protocol for maximum yield and minimal impurity formation, providing our clients with a reliable source of material that adheres to the highest industry standards.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic technology for their specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for custom targets, ensuring that your supply chain remains robust, compliant, and cost-effective in an evolving global market.
