Revolutionizing Ferrocenyl Arylamine Production with Metal-Free Catalysis for Global Pharma Supply Chains
Revolutionizing Ferrocenyl Arylamine Production with Metal-Free Catalysis for Global Pharma Supply Chains
The landscape of organic synthesis for high-value intermediates is constantly evolving, driven by the dual demands of sustainability and cost-efficiency. A significant breakthrough in this domain is documented in patent CN111662339A, which details a novel, metal-free methodology for constructing ferrocenyl-containing arylamine compounds. These compounds are not merely academic curiosities; they serve as critical building blocks in the development of advanced pharmaceuticals, agrochemicals, and functional materials. As illustrated in the background art of the patent, aromatic amines are ubiquitous in medicinal chemistry, forming the core structure of various therapeutic agents ranging from anti-Parkinson's medications to local anesthetics. The ability to efficiently introduce a ferrocenyl moiety into these scaffolds opens new avenues for modulating electronic properties and biological activity. This report analyzes the technical merits of this invention and its profound implications for global supply chains.
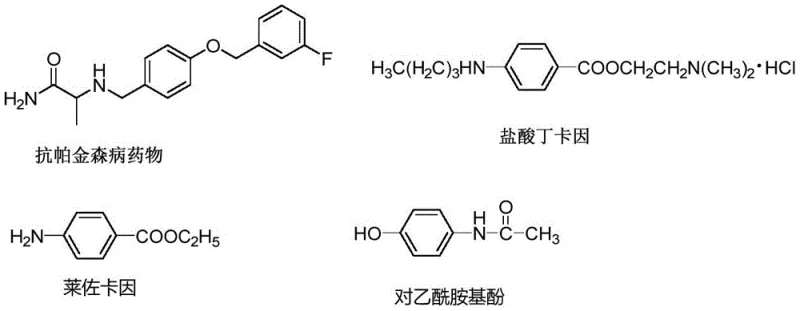
Traditionally, the construction of C-N bonds involving ferrocenyl systems has relied heavily on transition metal catalysis, often involving palladium or copper complexes. While effective, these conventional routes present substantial logistical and economic hurdles for industrial adoption. The reliance on precious metals introduces volatility in raw material pricing and necessitates rigorous downstream processing to meet stringent regulatory limits on heavy metal residues in active pharmaceutical ingredients (APIs). Furthermore, many traditional protocols require harsh reaction conditions, inert atmospheres, and anhydrous solvents, which escalate energy consumption and operational complexity. For procurement managers and supply chain directors, these factors translate into higher cost of goods sold (COGS) and increased risk of production delays due to catalyst availability or purification bottlenecks. The industry urgently requires a paradigm shift towards more robust and economically viable synthetic strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional synthetic pathways for ferrocenyl arylamines are often plagued by inefficiencies that hinder commercial scalability. The primary drawback is the dependency on exogenous transition metal catalysts, which are not only expensive but also pose significant environmental and safety challenges during disposal. The removal of trace metal contaminants from the final product is a resource-intensive process, often requiring specialized scavengers or multiple recrystallization steps, which inevitably erodes overall yield. Additionally, the substrates used in traditional cross-coupling reactions can be sensitive to moisture and oxygen, demanding sophisticated reactor setups and strictly controlled environments. These苛刻 (harsh) conditions limit the substrate scope and increase the barrier to entry for process chemists aiming to diversify their molecular libraries. Consequently, the lead time for developing these intermediates is prolonged, and the final cost remains prohibitively high for many applications.
The Novel Approach
In stark contrast, the technology disclosed in CN111662339A offers a streamlined, "one-pot" solution that elegantly circumvents these historical limitations. By utilizing a benzyne precursor strategy mediated by cesium fluoride and 18-crown-6-ether, the invention achieves the efficient construction of the ferrocenyl arylamine structural unit without any added metal catalyst. This approach leverages the high reactivity of benzyne intermediates generated in situ, which react rapidly with the nucleophilic nitrogen of the ferrocenyl oxazoline derivative. The result is a process that is not only simpler and faster but also exhibits superior atom economy. The reaction conditions are remarkably mild, operating effectively in standard toluene solvent at moderate temperatures. This simplicity translates directly into operational excellence, allowing manufacturers to reduce unit operations and minimize waste generation, thereby aligning with green chemistry principles while enhancing profitability.
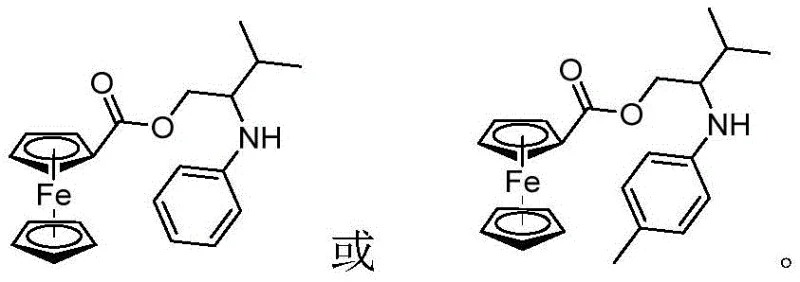
Mechanistic Insights into Benzyne-Mediated C-N Bond Formation
To fully appreciate the innovation of this protocol, one must understand the underlying mechanistic intricacies that drive the reaction efficiency. The process initiates with the fluoride-induced elimination of the trimethylsilyl and triflate groups from the benzyne precursor, generating a highly reactive benzyne intermediate. This species acts as a powerful electrophile. Simultaneously, the lone pair of electrons on the nitrogen atom of the (s)-(4-isopropyloxazolin-2-yl)ferrocene serves as a potent nucleophile. The interaction between these two species is facilitated by the phase transfer catalyst, 18-crown-6-ether, which complexes with the cesium cation to enhance the solubility and reactivity of the fluoride anion in the organic phase. This synergistic effect ensures that the benzyne generation and subsequent nucleophilic attack proceed smoothly without the need for external thermal activation beyond moderate heating.
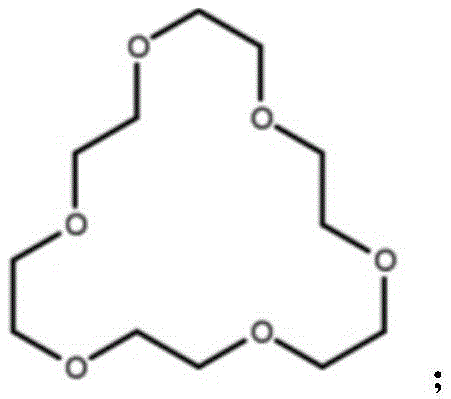
Furthermore, the mechanism elegantly incorporates trace amounts of water present in the system—either from the hygroscopic nature of cesium fluoride or non-distilled toluene—as a proton source for the final aromatization step. After the initial nucleophilic addition, the resulting zwitterionic intermediate undergoes proton transfer and electron rearrangement to yield the stable ferrocenyl arylamine product. This tolerance to moisture is a distinct advantage over many organometallic reactions that require rigorously anhydrous conditions. From an impurity control perspective, the absence of metal catalysts means that the impurity profile is significantly cleaner, dominated primarily by organic by-products that are easier to separate via standard chromatographic techniques. This mechanistic clarity provides R&D directors with confidence in the robustness and reproducibility of the process when transferring from laboratory to pilot plant scales.
How to Synthesize Ferrocenyl Arylamine Efficiently
Implementing this synthesis in a practical setting involves a straightforward sequence of operations that minimizes handling risks and maximizes throughput. The protocol dictates a precise molar ratio of reactants to ensure optimal conversion while minimizing excess reagent waste. The reaction is conducted in toluene, a solvent familiar to most chemical manufacturing facilities, further reducing the need for specialized infrastructure. Following the reaction period, the workup procedure is designed to be scalable, utilizing liquid-liquid extraction and standard column chromatography for purification. This accessibility makes the technology highly attractive for contract development and manufacturing organizations (CDMOs) looking to expand their portfolio of organometallic intermediates. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps below.
- Prepare the reaction mixture by combining (s)-(4-isopropyloxazolin-2-yl)ferrocene, trifluoromethanesulfonic acid-2-(trimethylsilyl)phenyl ester, cesium fluoride, and 18-crown-6-ether in toluene solvent.
- Heat the reaction mixture to a temperature range of 60-80°C and maintain stirring for approximately 12 hours to facilitate the benzyne intermediate formation and subsequent nucleophilic attack.
- Upon completion, perform workup by washing with water, extracting with ethyl acetate, drying under reduced pressure, and purifying via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and supply continuity, the shift to this metal-free methodology represents a strategic opportunity to optimize the cost structure of ferrocenyl intermediate production. The elimination of precious metal catalysts removes a major variable cost component and mitigates the supply risk associated with geopolitically sensitive materials like palladium. Moreover, the simplified purification process reduces the consumption of solvents and stationary phases, leading to substantial cost savings in waste management and raw materials. The robustness of the reaction conditions also implies a lower rate of batch failures, enhancing overall equipment effectiveness (OEE) and ensuring more reliable delivery schedules to downstream customers. These factors collectively contribute to a more resilient and cost-competitive supply chain.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of expensive transition metal catalysts from the bill of materials. In traditional processes, the cost of the catalyst and the subsequent metal scavenging agents can account for a significant portion of the total production cost. By replacing these with inexpensive inorganic salts like cesium fluoride and macrocyclic ethers, the direct material cost is drastically reduced. Additionally, the "one-pot" nature of the reaction minimizes the number of isolation and purification steps, which lowers labor costs and energy consumption associated with heating, cooling, and solvent recovery. This lean manufacturing approach allows for a more competitive pricing strategy in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals that are readily available from multiple global suppliers. Unlike specialized ligands or proprietary catalyst systems that may have single-source dependencies, reagents such as toluene, cesium fluoride, and 18-crown-6-ether are produced at scale worldwide. This diversity of supply sources reduces the risk of disruption due to logistics issues or manufacturer outages. Furthermore, the mild reaction conditions reduce the stress on reactor equipment, potentially extending asset life and reducing maintenance downtime. For procurement managers, this translates to greater flexibility in sourcing and a more predictable lead time for raw material acquisition, ensuring continuous production flow.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the simplicity of this benzyne-mediated reaction facilitates a smoother transition from gram to kilogram scales. The absence of toxic heavy metals simplifies the environmental compliance landscape, as there is no need for complex wastewater treatment protocols to remove metal ions. This aligns with increasingly stringent global environmental regulations and corporate sustainability goals. The high atom economy of the reaction ensures that a larger proportion of the starting materials end up in the final product, minimizing the generation of hazardous waste. This eco-friendly profile not only reduces disposal costs but also enhances the brand reputation of the manufacturer as a responsible supplier of green chemistry solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a transparent view of the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for their specific project needs. We encourage technical teams to review these points to assess the alignment with their current process development goals.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method (CN111662339A) specifically operates without external transition metal or precious metal catalysts, utilizing cesium fluoride and 18-crown-6-ether instead, which significantly reduces raw material costs and simplifies purification.
Q: What are the key advantages for large-scale manufacturing of these intermediates?
A: The process offers high atom economy and utilizes readily available reagents. The 'one-pot' nature reduces unit operations, and the absence of heavy metals eliminates the need for complex and costly metal scavenging steps typically required in pharmaceutical manufacturing.
Q: Can this method be adapted for different substituents on the aromatic ring?
A: Yes, the patent demonstrates substrate versatility by successfully synthesizing derivatives with both hydrogen and methyl substituents (R1, R2), indicating a broad scope for generating diverse ferrocenyl arylamine libraries for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ferrocenyl Arylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis technology described in CN111662339A for the production of high-value ferrocenyl intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel chemistry can be realized at an industrial level. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of ferrocenyl arylamine meets the exacting standards required by the pharmaceutical and specialty chemical industries.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive efficiency and value for your organization. Let us be your trusted partner in navigating the complexities of modern chemical synthesis.
