Advanced Metal-Free Synthesis of Ferrocenyl Arylamines for Commercial Pharmaceutical Applications
Advanced Metal-Free Synthesis of Ferrocenyl Arylamines for Commercial Pharmaceutical Applications
The landscape of organic synthesis for functionalized arylamines is undergoing a significant transformation, driven by the urgent need for greener, more efficient, and cost-effective manufacturing processes in the pharmaceutical and fine chemical sectors. A pivotal advancement in this domain is detailed in patent CN111662339B, which discloses a novel, metal-free methodology for constructing ferrocenyl arylamine structural units. This technology represents a paradigm shift from traditional transition-metal catalyzed cross-couplings to a streamlined benzyne-mediated approach that operates under mild conditions. By leveraging trifluoromethanesulfonic acid-2-(trimethylsilyl)phenyl ester compounds as stable benzyne precursors, combined with cesium fluoride and 18-crown-6-ether, this invention achieves efficient bond formation without the burden of expensive palladium or copper catalysts. For R&D directors and procurement strategists, this patent offers a compelling route to high-value intermediates that are critical for the development of next-generation medicinal agents and advanced functional materials.
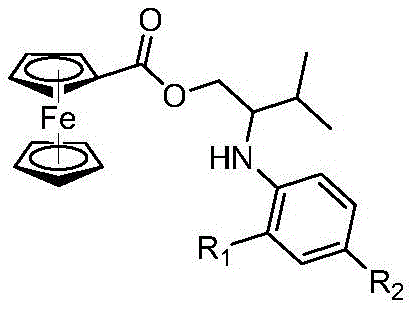
The significance of this technological breakthrough extends beyond mere academic interest; it addresses fundamental supply chain vulnerabilities associated with precious metal dependency. Ferrocenyl arylamines are versatile scaffolds known for their unique electronic properties, finding applications ranging from hole-transport materials in OLEDs to active pharmaceutical ingredients with antioxidant and anticancer potential. The ability to synthesize these complex architectures through a one-pot reaction that tolerates various functional groups opens new avenues for rapid library generation and process optimization. As a reliable ferrocenyl arylamine supplier, understanding the nuances of such proprietary synthetic routes is essential for maintaining a competitive edge in the global market for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-nitrogen bonds in ferrocenyl systems has relied heavily on transition-metal catalyzed cross-coupling reactions, such as Buchwald-Hartwig aminations, which necessitate the use of palladium or copper complexes. While effective, these conventional methodologies suffer from inherent drawbacks that pose significant challenges for industrial scale-up and regulatory compliance. The requirement for expensive noble metal catalysts not only inflates the raw material costs but also introduces severe purification hurdles, as residual metal levels in pharmaceutical intermediates must be reduced to parts-per-million (ppm) levels to meet strict safety standards. Furthermore, these reactions often demand rigorous exclusion of moisture and oxygen, requiring specialized equipment and inert atmosphere handling that complicates the manufacturing workflow. The generation of stoichiometric metal waste also conflicts with modern green chemistry principles, creating environmental liabilities and increasing waste disposal costs for chemical manufacturers striving for sustainability.
The Novel Approach
In stark contrast to the metal-dependent status quo, the method described in patent CN111662339B utilizes a transition-metal-free strategy that capitalizes on the high reactivity of benzyne intermediates generated in situ. By employing trifluoromethanesulfonic acid-2-(trimethylsilyl)phenyl esters as precursors, the reaction proceeds through a fluoride-induced elimination mechanism that generates a highly electrophilic benzyne species without the need for external metal activation. This approach drastically simplifies the reaction setup, allowing the use of common laboratory solvents like toluene and standard heating conditions between 60-80°C. The elimination of transition metals inherently solves the heavy metal residue problem, significantly reducing downstream purification costs and accelerating the time-to-market for new drug candidates. Moreover, the use of cesium fluoride as a mild base coupled with 18-crown-6-ether as a phase transfer catalyst ensures high atom economy and operational simplicity, making this route exceptionally attractive for the commercial scale-up of complex organometallic intermediates.
Mechanistic Insights into Benzyne-Mediated Ferrocenyl Coupling
The mechanistic elegance of this synthesis lies in the generation and trapping of the benzyne intermediate under remarkably mild conditions. Upon addition of cesium fluoride to the reaction mixture containing the trimethylsilyl-substituted triflate precursor, the fluoride ion acts as a trigger to cleave the silicon-carbon bond, simultaneously expelling the triflate group to generate a transient benzyne species. This highly reactive intermediate possesses a strained triple bond within the aromatic ring, rendering the ortho-carbon atoms extremely electrophilic. The nitrogen atom of the (s)-(4-isopropyloxazolin-2-yl)ferrocene, acting as a nucleophile, attacks one of the benzyne carbons to form a new carbon-nitrogen bond. This step is facilitated by the electron-donating nature of the ferrocenyl moiety, which enhances the nucleophilicity of the amine nitrogen, ensuring rapid capture of the fleeting benzyne before it can undergo undesired polymerization or side reactions.
Following the nucleophilic attack, the resulting zwitterionic intermediate undergoes a proton transfer process that is critically dependent on the trace amounts of water present in the system, either from the hygroscopic nature of cesium fluoride or the solvent. The negatively charged carbon on the aromatic ring abstracts a proton from water, while the positively charged nitrogen is stabilized, eventually leading to the formation of the final ferrocenyl arylamine product after tautomerization or hydrolysis steps. The presence of 18-crown-6-ether is paramount in this mechanism, as it encapsulates the cesium cation, effectively "naked" the fluoride anion and increasing its basicity and nucleophilicity in the non-polar toluene medium. This intricate interplay of reagents ensures high regioselectivity and minimizes the formation of by-products, providing a robust platform for synthesizing diverse derivatives with varying substituents on the aromatic ring.
How to Synthesize Ferrocenyl Arylamine Efficiently
Implementing this synthesis protocol requires precise control over reagent stoichiometry and reaction parameters to maximize yield and purity. The process begins with the preparation of a homogeneous mixture of the ferrocenyl oxazoline substrate and the benzyne precursor in dry toluene, followed by the sequential addition of cesium fluoride and the phase transfer catalyst. Maintaining the reaction temperature within the optimal window of 60-80°C for a duration of 12 hours is crucial to ensure complete conversion of the starting materials while preventing thermal degradation of the sensitive ferrocenyl scaffold. Detailed standardized operating procedures for this synthesis, including specific workup and purification guidelines, are outlined below to assist process chemists in replicating these results.
- Combine (s)-(4-isopropyloxazolin-2-yl)ferrocene, trifluoromethanesulfonic acid-2-(trimethylsilyl)phenyl ester, cesium fluoride, and 18-crown-6-ether in toluene solvent.
- Heat the reaction mixture to a temperature range of 60-80°C and maintain stirring for approximately 12 hours to facilitate the benzyne-mediated coupling.
- Upon completion, perform aqueous workup followed by ethyl acetate extraction and purify the crude red solid via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route offers tangible strategic benefits that directly impact the bottom line and operational resilience. By shifting away from precious metal catalysts, manufacturers can insulate their production costs from the volatile pricing of palladium and rhodium, which are subject to geopolitical supply constraints and market speculation. The simplified reaction profile, which operates under ambient pressure and moderate temperatures, reduces the energy consumption and capital expenditure required for specialized high-pressure reactors or cryogenic cooling systems. Furthermore, the reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, minimizing the risk of compliance penalties and facilitating smoother audits for international clients who prioritize sustainable sourcing practices in their supply chains.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete elimination of expensive transition metal catalysts and the associated ligands, which often constitute a significant portion of the raw material bill in traditional cross-coupling reactions. Without the need for costly metal scavengers or extensive chromatographic purification to remove trace metals, the downstream processing costs are drastically simplified, leading to substantial overall cost savings per kilogram of produced intermediate. Additionally, the use of commodity chemicals like cesium fluoride and toluene, which are readily available in bulk quantities from multiple global suppliers, ensures a stable and competitive pricing structure that is not vulnerable to the supply bottlenecks often seen with specialty organometallic reagents.
- Enhanced Supply Chain Reliability: Diversifying the synthetic route to exclude critical raw materials that are prone to supply disruptions enhances the overall robustness of the manufacturing supply chain. Since the reagents required for this benzyne-mediated process are standard industrial chemicals with well-established production networks, the risk of production stoppages due to reagent shortages is significantly mitigated. This reliability allows for more accurate lead time forecasting and inventory planning, enabling companies to respond more agilely to fluctuating market demands for ferrocenyl derivatives without the fear of being held hostage by single-source suppliers of exotic catalysts.
- Scalability and Environmental Compliance: The one-pot nature of this reaction, combined with its tolerance for standard solvents and mild thermal conditions, makes it inherently scalable from gram-scale laboratory synthesis to multi-ton commercial production without significant process re-engineering. The absence of heavy metals simplifies the waste stream management, reducing the volume of hazardous waste that requires specialized treatment and disposal, thereby lowering the environmental footprint of the manufacturing facility. This alignment with green chemistry principles not only improves the corporate sustainability profile but also facilitates easier regulatory approval for new drug applications where impurity profiles are scrutinized heavily by health authorities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ferrocenyl arylamine synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters and partners.
Q: What is the primary advantage of this ferrocenyl arylamine synthesis method over traditional routes?
A: The primary advantage is the elimination of transition metal or noble metal catalysts, which simplifies purification, reduces heavy metal contamination risks, and lowers overall production costs while maintaining high atom economy.
Q: What role does 18-crown-6-ether play in this reaction system?
A: 18-crown-6-ether acts as a phase transfer catalyst that complexes with cesium ions, thereby enhancing the solubility and reactivity of the fluoride anion in the organic toluene solvent, which is critical for generating the reactive benzyne intermediate.
Q: Can this method be adapted for large-scale manufacturing of arylamine derivatives?
A: Yes, the method utilizes readily available reagents and mild heating conditions (60-80°C) in a one-pot process, making it highly suitable for commercial scale-up without the need for specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ferrocenyl Arylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in advancing the production of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like patent CN111662339B can be seamlessly translated into robust industrial processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the absence of heavy metal contaminants and ensure batch-to-batch consistency for our global clientele.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this advanced synthesis technology for your specific project needs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments. Let us partner with you to optimize your supply chain, reduce manufacturing costs, and accelerate the development of next-generation ferrocenyl-based therapeutics and materials.
