Scalable Manufacturing of 5-Trifluoromethyl-1,2,4-Triazole Derivatives via Iron Catalysis
Scalable Manufacturing of 5-Trifluoromethyl-1,2,4-Triazole Derivatives via Iron Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which enhance metabolic stability and lipophilicity. A significant breakthrough in this domain is detailed in patent CN111978265B, which discloses a highly efficient preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives. This scaffold is not merely an academic curiosity but a critical structural motif found in numerous blockbuster drugs and functional materials. As illustrated in the provided chemical structures, prominent pharmaceutical agents such as Maraviroc, Triazolam, Sitagliptin, and Deferasirox all incorporate this vital 1,2,4-triazole core, underscoring its immense value in modern medicinal chemistry. The ability to synthesize these derivatives efficiently directly impacts the supply chain reliability for reliable pharmaceutical intermediate suppliers globally.
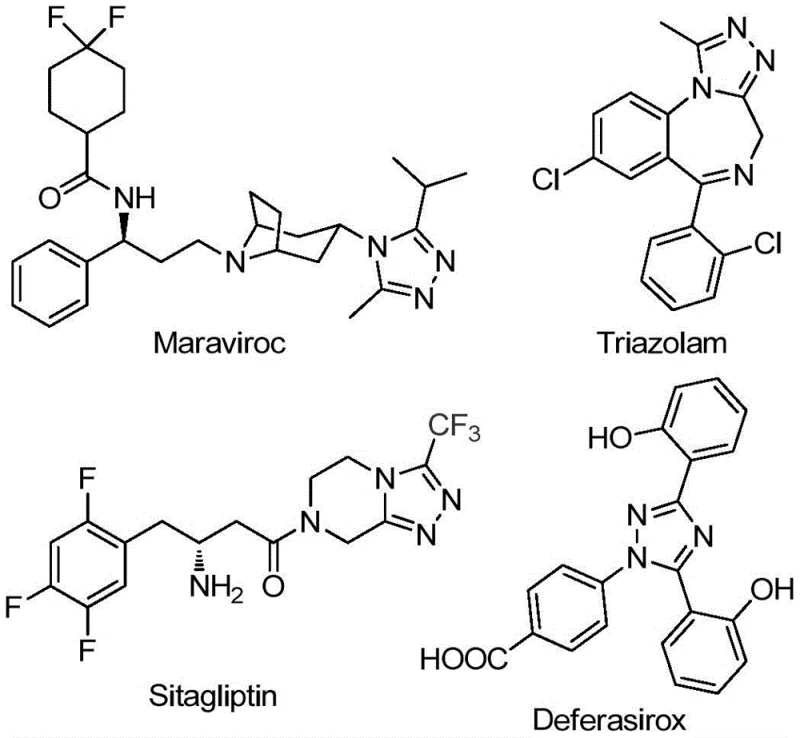
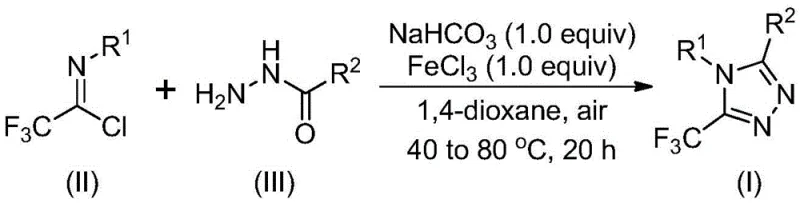
The strategic importance of this patent lies in its potential to streamline the manufacturing of complex APIs. By introducing a trifluoromethyl group into the heterocyclic molecular framework, manufacturers can significantly improve the electronegativity and bioavailability of the final drug product. However, traditional synthetic routes have often been plagued by inefficiencies. The technology described in CN111978265B offers a compelling solution by utilizing inexpensive and readily available starting materials, specifically acyl hydrazides and trifluoroethylimidoyl chlorides. This approach eliminates the dependency on exotic reagents, thereby lowering the barrier to entry for cost reduction in API manufacturing and ensuring a more stable supply of high-purity triazole derivatives for downstream formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the literature reported several traditional methods for synthesizing trifluoromethyl-substituted 1,2,4-triazoles, each carrying significant drawbacks that hindered large-scale adoption. These conventional pathways included the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines, or the cyclization of trifluoromethyl hydrazides with amidines. Other methods involved the reaction of trifluoromethyl-substitured hydrazones with nitriles, or the hydrazinolysis of 1,2,4-oxadiazoles. A critical analysis of these prior art techniques reveals they are generally limited by harsh reaction conditions that demand rigorous safety protocols, lengthy multi-step sequences that reduce overall throughput, and a notably narrow substrate range. Furthermore, these older methods frequently suffer from low reaction yields, making them economically unviable for the commercial scale-up of complex pharmaceutical intermediates required by multinational corporations.
The Novel Approach
In stark contrast to the limitations of the past, the novel approach disclosed in the patent utilizes a ferric chloride-promoted strategy that is both simple and highly efficient. This method starts from cheap and readily available hydrazides and trifluoroethylimide chlorides, bypassing the need for complex precursor synthesis. The reaction proceeds under relatively mild conditions, avoiding the extreme temperatures or pressures often associated with traditional heterocycle formation. Crucially, this new route does not require anhydrous or oxygen-free conditions, which drastically simplifies the operational complexity and equipment requirements for manufacturing facilities. The process is designed to be easily scalable to the gram level and beyond, providing distinct convenience for industrial scale production applications. This shift represents a paradigm change for procurement managers looking for cost reduction in electronic chemical manufacturing and pharmaceutical sectors alike.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for R&D directors focused on process optimization and impurity control. The reaction mechanism likely involves a sequential pathway initiated by base promotion. Initially, sodium bicarbonate facilitates an intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and the hydrazide, generating a trifluoroacetamidine derivative intermediate. This step is critical as it sets the stage for the subsequent ring closure. Following this initial coupling, the addition of the metal Lewis acid, specifically ferric chloride (FeCl3), promotes an intramolecular dehydration condensation reaction. This cyclization step effectively closes the five-membered triazole ring, yielding the final 5-trifluoromethyl substituted 1,2,4-triazole derivative. The use of FeCl3 is particularly advantageous as it acts as a potent yet inexpensive promoter, driving the reaction to completion without the need for precious metal catalysts.
From an impurity control perspective, the mild nature of this two-stage temperature protocol (initially 30 to 50 °C, followed by 70 to 90 °C) allows for better management of side reactions. Traditional high-energy methods often lead to decomposition or polymerization of sensitive intermediates, resulting in difficult-to-remove impurities. By contrast, this controlled thermal profile ensures that the intermediate trifluoroacetamidine forms cleanly before the cyclization energy is applied. The broad functional group tolerance mentioned in the patent suggests that substituents on the aryl rings (R1 and R2), such as methyl, methoxy, halogens, or trifluoromethyl groups, remain stable throughout the process. This stability is paramount for maintaining high purity specifications, ensuring that the final product meets the stringent quality standards required for reliable agrochemical intermediate supplier networks and pharmaceutical applications.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot plant execution. The process begins by combining the key reagents—sodium bicarbonate, trifluoroethylimidoyl chloride, and the specific acyl hydrazide—in a suitable organic solvent. The choice of solvent is critical; while various solvents can dissolve the raw materials, aprotic solvents like 1,4-dioxane, acetonitrile, or tetrahydrofuran are preferred for their ability to effectively promote the reaction. Specifically, 1,4-dioxane has been identified as optimal, allowing various raw materials to convert into products at a higher rate. The reaction mixture is stirred at a moderate temperature to allow the initial coupling to occur before the Lewis acid is introduced for the final cyclization step. Detailed standardized synthesis steps follow below.
- Mix sodium bicarbonate, trifluoroethylimidoyl chloride, and hydrazide in an aprotic organic solvent such as 1,4-dioxane.
- Stir the mixture at a moderate temperature range of 30 to 50 °C for 8 to 16 hours to facilitate initial bond formation.
- Add ferric chloride (FeCl3) as a Lewis acid promoter and heat the reaction system to 70 to 90 °C for 6 to 10 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patent are profound. The shift towards this specific synthetic route addresses several traditional pain points associated with heterocycle manufacturing, primarily centering on raw material costs and operational simplicity. By utilizing reagents that are widely available in the industrial marketplace, such as aromatic amines, acid chlorides, and ferric chloride, the method decouples production from volatile supply chains dependent on specialized catalysts. This accessibility translates directly into enhanced supply chain reliability, as sourcing becomes less risky and more predictable. Furthermore, the elimination of strict anhydrous and anaerobic requirements reduces the capital expenditure needed for specialized reactor setups, allowing existing infrastructure to be utilized more effectively for the commercial scale-up of complex polymer additives or pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of expensive catalytic systems with inexpensive iron salts and common bases. Ferric chloride is significantly cheaper than precious metal catalysts like palladium or rhodium, which are often used in cross-coupling reactions. Additionally, the use of sodium bicarbonate as a base avoids the costs associated with strong, hazardous bases that require special handling and disposal. The high conversion rates observed in the patent data, with yields reaching up to 90% for certain substrates, mean that raw material waste is minimized. This efficiency leads to substantial cost savings in the overall production budget, making the final triazole derivatives more competitive in the global market without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized reagents. This method mitigates that risk by relying on commodity chemicals. The starting materials, such as trifluoroethylimidoyl chloride and various hydrazides, can be synthesized rapidly from corresponding aromatic amines and acid chlorides, which are bulk chemicals produced globally. This abundance ensures that production schedules are not disrupted by raw material shortages. Moreover, the reaction's tolerance to air and moisture means that storage and transportation of intermediates are less critical, reducing logistical costs and complexities. This robustness allows for reducing lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands and urgent API production needs.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new environmental and safety challenges, but this methodology is inherently designed for scalability. The patent notes that the method can be easily expanded to the gram level and implies suitability for larger scales due to its simple operation. From an environmental standpoint, the use of iron, a non-toxic metal, aligns with green chemistry principles, reducing the burden of heavy metal waste disposal compared to processes using copper or palladium. The simplified post-processing, involving filtration and standard column chromatography or crystallization, minimizes solvent usage and energy consumption. These factors collectively support a more sustainable manufacturing footprint, which is increasingly a requirement for partnerships with major multinational corporations focused on ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in patent CN111978265B. They are intended to clarify the operational parameters and potential applications for stakeholders evaluating this technology for integration into their existing manufacturing portfolios. Understanding these details is crucial for assessing the feasibility of adopting this route for specific target molecules.
Q: What are the key advantages of the FeCl3-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods that require harsh conditions or expensive reagents, this patent-disclosed route utilizes cheap, commercially available starting materials and operates under air-tolerant conditions without the need for strict anhydrous or oxygen-free environments.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly states that the method can be easily scaled up to the gram level and beyond, providing significant convenience for industrial-scale production applications due to its simple operational steps and robust reaction conditions.
Q: What is the substrate scope for R1 and R2 groups in this triazole synthesis?
A: The method demonstrates broad functional group tolerance, accommodating substituted or unsubstituted aryl groups for R1 and alkyl, alkenyl, or aryl groups for R2, allowing for the design of diverse 3,4-disubstituted triazole derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies disclosed in CN111978265B for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality supply regardless of volume. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 5-trifluoromethyl-1,2,4-triazole derivative meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging this efficient iron-catalyzed chemistry to deliver cost-effective solutions that accelerate your drug development timelines.
We invite you to collaborate with us to optimize your supply chain for these critical building blocks. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this novel synthetic route can improve your margins. Please contact our technical procurement team today to request specific COA data for our available triazole derivatives and to discuss route feasibility assessments for your custom synthesis projects. Let us help you secure a reliable supply of these essential intermediates for your next generation of therapeutic agents.
