Advanced Synthesis of Asymmetric Spirobifluorene Amines for High-Performance OLED Manufacturing
Advanced Synthesis of Asymmetric Spirobifluorene Amines for High-Performance OLED Manufacturing
The rapid evolution of the organic light-emitting diode (OLED) industry demands intermediates that offer not only superior optoelectronic properties but also manufacturability at scale. Patent CN102126963B introduces a groundbreaking methodology for synthesizing a specific class of 9,9'-spirobifluorene amino compounds, designated generally as structure (I). These compounds serve as pivotal building blocks for next-generation display and optoelectronic materials. The core innovation lies in the strategic introduction of alkyl groups at the 7-position of the spirobifluorene scaffold, which fundamentally alters the molecular symmetry. This structural modification is not merely academic; it directly translates to enhanced amorphous characteristics, improved thermal stability, and superior solubility in common organic solvents. For R&D directors and procurement specialists alike, this patent represents a shift away from reliance on scarce, expensive precursors toward a more robust, commodity-chemical-based supply chain. By leveraging Friedel-Crafts chemistry and classical Grignard protocols, this route circumvents the bottlenecks associated with palladium-catalyzed cross-couplings often found in legacy syntheses.
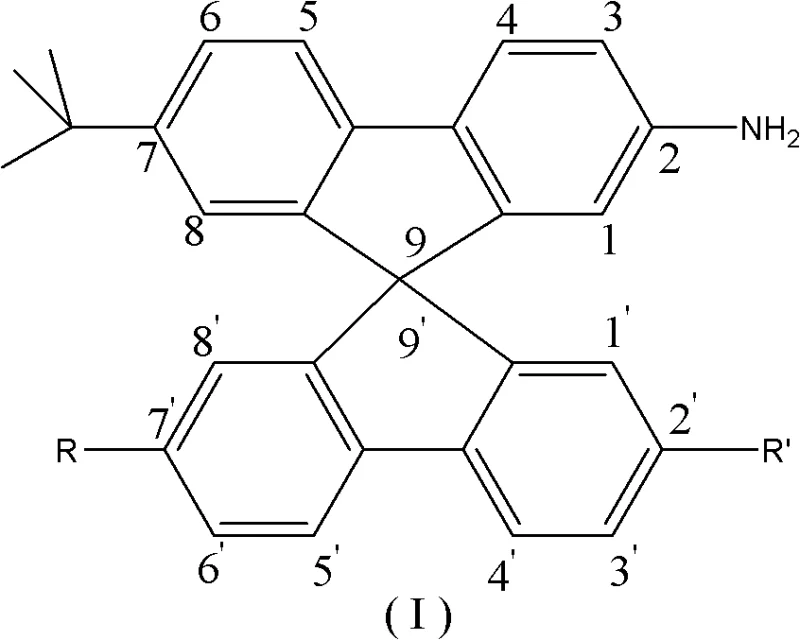
Understanding the limitations of prior art is essential for appreciating the commercial value of this new pathway. Conventional methods for producing spirobifluorene derivatives typically rely on 2-bromobiphenyl as the starting material. Historically, 2-bromobiphenyl is synthesized from o-dibromobenzene via coupling reactions that necessitate expensive palladium catalysts and stringent operational conditions. This dependency creates a fragile supply chain vulnerable to fluctuations in precious metal prices and imposes significant purification burdens to remove trace metal contaminants, which are detrimental to OLED device longevity. Furthermore, existing spirobifluorene amines often lack sufficient structural asymmetry, leading to materials with lower glass transition temperatures and poorer film-forming capabilities. The novel approach detailed in the patent addresses these pain points head-on by utilizing biphenyl, a widely available and inexpensive petrochemical feedstock, as the foundational raw material. Through a sequence of alkylation, bromination, and cyclization, the process constructs the complex spiro architecture without ever requiring a palladium catalyst in the initial stages.
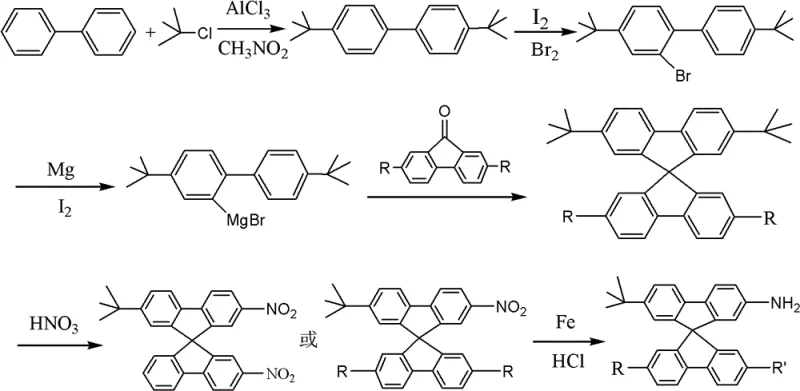
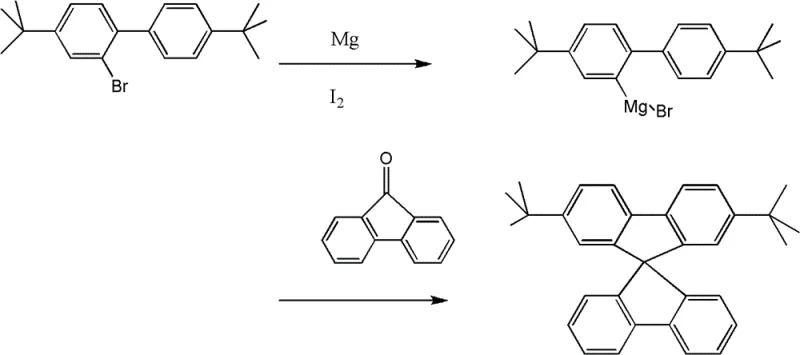
The mechanistic elegance of this synthesis offers profound insights for process chemists aiming to optimize yield and purity. The journey begins with a Lewis acid-catalyzed Friedel-Crafts alkylation, where biphenyl reacts with tert-butyl chloride in the presence of aluminum chloride (AlCl3) within a nitromethane solvent system. This step is critical for installing the solubilizing tert-butyl groups that will later define the material's processing characteristics. Following this, a highly regioselective bromination occurs using liquid bromine catalyzed by elemental iodine. This specific halogenation pattern is prerequisite for the subsequent formation of the Grignard reagent. The heart of the synthesis is the construction of the spiro center. The generated aryl magnesium bromide species undergoes nucleophilic addition to a substituted fluorenone ketone. This is followed by an acid-catalyzed cyclization step, typically performed in acetic acid with hydrochloric acid at elevated temperatures around 125°C. This cascade effectively locks the two fluorene units into an orthogonal arrangement, creating the rigid 9,9'-spirobifluorene core that prevents pi-stacking and aggregation.
A particularly fascinating aspect of the chemical mechanism involves the functionalization of the spiro core via electrophilic aromatic substitution. The patent describes a unique nitration strategy where nitric acid in acetic acid is used to introduce nitro groups. Remarkably, under specific molar ratios, this reaction proceeds via an ipso-substitution mechanism where the tert-butyl group at the 2-position is displaced by the incoming nitro group. This transformation is highly valuable as it allows for the direct conversion of a solubilizing group into a versatile handle for further derivatization or reduction to the final amine functionality. The final reduction step employs iron powder and hydrochloric acid in ethanol, a classic and economically favorable method for converting nitro groups to amines. This choice of reducing agent avoids the use of hydrogen gas and high-pressure equipment, further simplifying the safety profile and capital expenditure requirements for manufacturing facilities. The result is a high-purity amino-spirobifluorene derivative ready for downstream coupling reactions.
How to Synthesize 7-tert-butyl-2-amino-spirobifluorene Efficiently
The synthesis of these high-value electronic intermediates requires precise control over reaction parameters to ensure consistent quality and batch-to-batch reproducibility. The patented procedure outlines a logical progression from simple aromatics to complex heterocyclic systems, emphasizing the importance of temperature control during the exothermic alkylation and bromination phases. Detailed standardized operating procedures regarding reagent addition rates, quenching protocols, and purification methods such as column chromatography or recrystallization are essential for translating this laboratory-scale success into a commercial reality. Manufacturers must pay close attention to the stoichiometry of the nitration step, as the ratio of nitric acid to substrate dictates whether mono- or di-nitrated products are obtained, allowing for tunable functionality depending on the specific end-application requirements.
- Perform Friedel-Crafts alkylation of biphenyl with tert-butyl chloride using AlCl3 catalyst in nitromethane to form 4,4'-di-tert-butylbiphenyl.
- Execute regioselective bromination using liquid bromine and iodine catalyst to generate 4,4'-di-tert-butyl-2-bromobiphenyl.
- Form the spiro core via Grignard reaction with substituted fluorenone followed by acid-catalyzed cyclization at elevated temperatures.
- Conduct ipso-nitration using nitric acid in acetic acid to replace tert-butyl groups with nitro groups, followed by iron-mediated reduction to amines.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The most immediate impact is seen in the raw material cost structure. By replacing specialized, high-cost starting materials like 2-bromobiphenyl with commodity chemicals like biphenyl and tert-butyl chloride, the overall cost of goods sold (COGS) is drastically reduced. This cost efficiency is compounded by the elimination of palladium catalysts, which not only removes a significant line item from the budget but also simplifies the waste management profile. The absence of heavy metals means that downstream purification does not require expensive scavenging resins or complex filtration setups, leading to faster cycle times and higher throughput. Furthermore, the reliance on robust, well-understood chemical transformations such as Grignard reactions and iron reductions ensures that the process is inherently scalable. These reactions are forgiving and can be easily adapted from pilot plant scales to multi-ton production campaigns without the need for exotic reactor configurations or extreme pressure conditions.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with abundant Lewis acids like aluminum chloride and the use of iron for reduction significantly lowers the direct material costs. Additionally, the simplified purification train reduces solvent consumption and energy usage associated with extensive metal removal processes, contributing to substantial operational expenditure savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Sourcing strategies are de-risked by the use of globally available feedstocks. Biphenyl, tert-butyl chloride, and fluorenone are produced by multiple suppliers worldwide, preventing single-source bottlenecks. This diversification ensures continuity of supply even during regional disruptions, providing a stable foundation for long-term production planning and inventory management.
- Scalability and Environmental Compliance: The process utilizes standard solvents like ethanol, acetic acid, and dichloromethane, which have established recovery and recycling protocols in modern chemical plants. The avoidance of hazardous reagents and the generation of manageable waste streams facilitate easier compliance with increasingly stringent environmental regulations, reducing the regulatory burden and permitting timelines for new manufacturing lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from a detailed analysis of the patent specifications and are intended to clarify the operational nuances for potential partners and licensees. Understanding these details is crucial for assessing the fit of this technology within existing manufacturing portfolios and for evaluating the technical risk associated with adoption.
Q: What is the primary advantage of this synthesis route over conventional methods?
A: The primary advantage is the substitution of expensive 2-bromobiphenyl and palladium catalysts with inexpensive biphenyl and aluminum chloride. This significantly lowers raw material costs and eliminates the need for costly heavy metal removal steps, streamlining the purification process for electronic grade materials.
Q: How does the asymmetric structure benefit OLED performance?
A: The introduction of tert-butyl groups at the 7-position increases the structural asymmetry of the spirobifluorene derivative. This asymmetry disrupts molecular packing, thereby enhancing the amorphousness of the material, which improves thermal stability and solubility—critical parameters for solution-processable OLED fabrication.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes standard unit operations such as reflux, filtration, and distillation with commodity reagents like nitromethane, acetic acid, and ethanol. The absence of sensitive transition metal catalysts and the use of robust reaction conditions (e.g., Fe/HCl reduction) make it highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirobifluorene Amino Compounds Supplier
As the demand for high-performance OLED materials continues to surge, the ability to deliver complex intermediates with consistent quality and competitive pricing becomes a decisive factor for market leadership. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in organic synthesis to bring innovative pathways like the one described in CN102126963B to commercial fruition. Our facility is equipped with state-of-the-art reactors and purification systems capable of handling the specific requirements of spirobifluorene chemistry, from moisture-sensitive Grignard formations to high-temperature cyclizations. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of material regardless of their volume requirements. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which employ advanced analytical techniques to verify the structural integrity and impurity profile of every batch.
We invite global innovators in the display and lighting sectors to collaborate with us on optimizing these synthetic routes for your specific applications. Whether you require custom modifications to the R-groups for tailored solubility or need assistance with regulatory documentation for new material filings, our technical team is ready to support your goals. Please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your current supply chain. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with NINGBO INNO PHARMCHEM can accelerate your product development timelines and enhance your bottom line.
