Scalable Asymmetric Synthesis of Triptolide: A Breakthrough for Pharmaceutical Manufacturing
Scalable Asymmetric Synthesis of Triptolide: A Breakthrough for Pharmaceutical Manufacturing
The pharmaceutical industry has long recognized the immense therapeutic potential of Triptolide, a bioactive diterpenoid triepoxide isolated from the traditional Chinese medicinal plant Tripterygium wilfordii. Known for its potent anti-tumor, immunosuppressive, and anti-inflammatory properties, the demand for high-purity Triptolide and its advanced intermediates has surged. However, the complex polycyclic structure containing three epoxide rings and a lactone moiety has historically posed significant challenges for total synthesis. Patent CN101638426A introduces a transformative approach to this problem, detailing a novel and effective method for the asymmetric synthesis of Triptolide. This technology leverages chiral induction to construct the molecular framework with high stereocontrol, offering a viable pathway for the mass production of this critical compound. By utilizing 5-methoxy-2-tetralone and 2-(2-iodoethyl)-1,4-butyrolactone as accessible starting materials, the invention provides a route that is not only chemically elegant but also operationally convenient for industrial application.
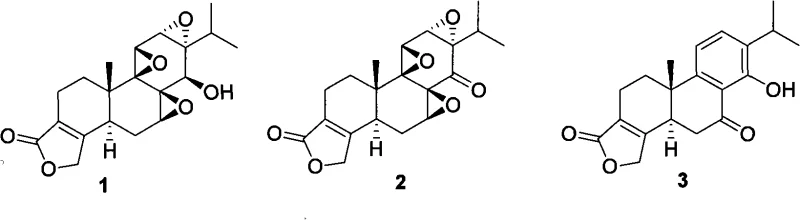
Triptolide, structurally defined as Compound 1 in the patent documentation, represents a pinnacle of structural complexity in natural product synthesis. The molecule's biological activity is strictly dependent on its stereochemistry, making asymmetric synthesis not just a preference but a necessity for clinical efficacy. The disclosed method addresses the critical need for a reliable pharmaceutical intermediate supplier capable of delivering enantiomerically pure materials without the prohibitive costs associated with traditional resolution techniques. This report analyzes the technical merits of this patent, focusing on its mechanistic innovations and the substantial commercial advantages it offers to procurement and supply chain decision-makers in the global fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Triptolide has been fraught with inefficiencies that hinder commercial viability. Early pioneering work by groups such as Berchtold and Van Tamelen established foundational routes, yet these methods were fundamentally limited by their inability to control stereochemistry effectively. As detailed in the background of the patent, these classical approaches typically yielded racemic mixtures, necessitating additional, yield-depleting resolution steps to isolate the biologically active enantiomer. Furthermore, these routes were characterized by excessive linearity, often requiring upwards of twenty steps with poor overall yields. For instance, the method reported by Professor Dan Yang in 1997, while achieving the first asymmetric total synthesis, suffered from low yields in multiple key steps and operational difficulties that rendered it unsuitable for large-scale manufacturing. The reliance on harsh reagents and complex protection-deprotection sequences further exacerbated the cost and environmental footprint of these legacy processes.
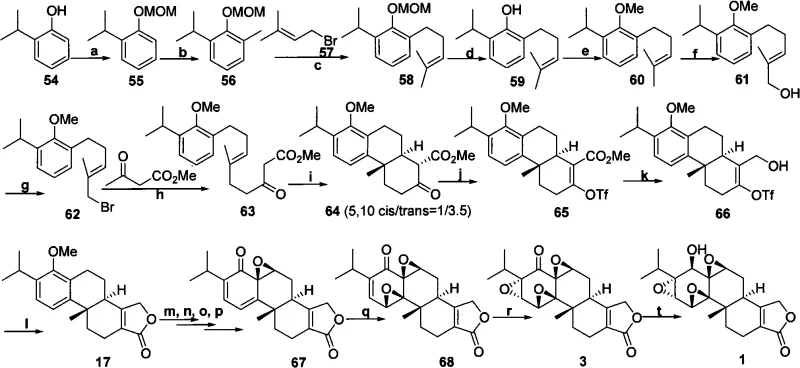
The Novel Approach
In stark contrast to the convoluted pathways of the past, the methodology described in CN101638426A offers a streamlined, convergent strategy that drastically simplifies the construction of the Triptolide skeleton. The core innovation lies in the early introduction of chirality through a catalytic asymmetric alkylation, bypassing the need for late-stage resolution. By employing a chiral phase transfer catalyst, the synthesis establishes the first critical stereocenter directly from achiral starting materials with high fidelity. This strategic shift reduces the step count significantly and improves the overall atom economy of the process. The route proceeds through a series of robust transformations, including intramolecular aldol condensation and selective oxidations, which are optimized for high yield and reproducibility. This modern approach transforms Triptolide from a laboratory curiosity into a feasible target for cost reduction in API manufacturing, aligning perfectly with the needs of modern pharmaceutical development.
Mechanistic Insights into Chiral Phase Transfer Catalyzed Alkylation
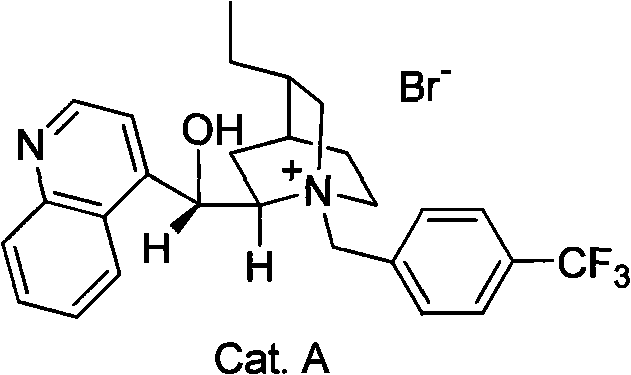
The heart of this synthetic breakthrough is the enantioselective alkylation of 5-methoxy-2-tetralone. In this pivotal step, the ketone substrate reacts with 2-(2-iodoethyl)-1,4-butyrolactone under the influence of a chiral quaternary ammonium salt, specifically a cinchoninium derivative such as Catalyst A. This phase transfer catalyst operates at the interface of the organic and aqueous phases, shuttling the enolate of the tetralone into the organic layer within a chiral environment. The bulky quaternary ammonium cation shields one face of the planar enolate, forcing the electrophilic attack of the iodide to occur from the opposite, unhindered face. This precise spatial control ensures the formation of the desired (R)-configured ketone intermediate (Compound 6) with high enantiomeric excess. The success of this step is paramount, as it sets the stereochemical trajectory for all subsequent ring closures and functional group manipulations in the synthesis.
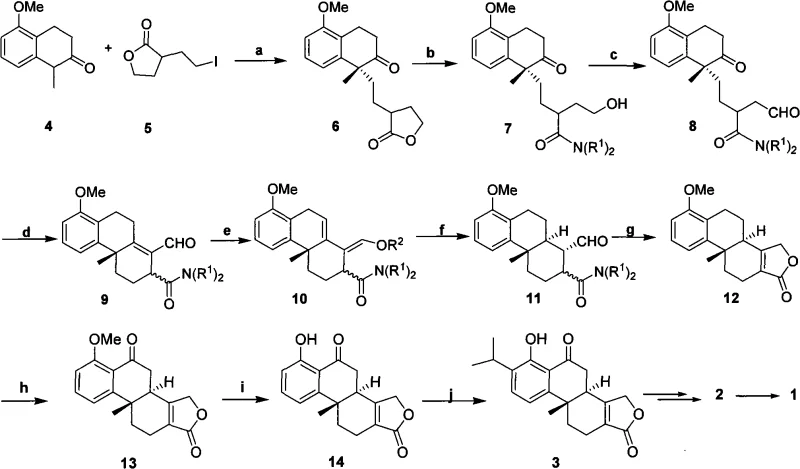
Following the establishment of chirality, the synthesis proceeds through a carefully orchestrated cascade of reactions designed to build complexity while maintaining stereochemical integrity. The conversion of the initial alkylated ketone into the tricyclic core involves an intramolecular aldol condensation, which closes the central ring system. Subsequent steps include enol etherification using silyl chlorides and catalytic hydrogenation to saturate specific double bonds without affecting sensitive epoxide precursors. The mechanism also features a strategic benzylic oxidation using chromium trioxide to install the necessary oxygen functionality, followed by demethylation and acid-catalyzed isopropylation to finalize the aromatic substitution pattern. Each transformation is selected not only for its chemical efficiency but also for its compatibility with the sensitive lactone and future epoxide functionalities, ensuring that the final product retains the rigorous purity specifications required for pharmaceutical applications.
How to Synthesize Triptolide Efficiently
The implementation of this synthesis requires precise control over reaction parameters to maximize yield and stereoselectivity. The process begins with the preparation of the chiral catalyst and the careful drying of solvents to prevent hydrolysis of sensitive intermediates. The alkylation step is conducted under biphasic conditions with vigorous stirring to ensure efficient mass transfer between the aqueous base and the organic substrates. Following the initial coupling, the intermediate undergoes amidation and oxidation to prepare for the crucial ring-closing events. The detailed standardized synthetic steps, including specific molar ratios, temperature profiles, and workup procedures for each transformation from Compound 4 to Compound 1, are outlined in the technical guide below.
- Perform asymmetric alkylation of 5-methoxy-2-tetralone with 2-(2-iodoethyl)-1,4-butyrolactone using a chiral phase transfer catalyst to establish the first stereocenter.
- Convert the resulting ketone to an amide, oxidize to an aldehyde, and undergo intramolecular aldol condensation to form the cyclic enone structure.
- Execute enol etherification, catalytic hydrogenation, and lactonization to construct the core skeleton, followed by benzylic oxidation and isopropylation to yield the key intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing 5-methoxy-2-tetralone and simple lactone derivatives as starting points, manufacturers can source inputs from a broad base of commodity chemical suppliers, reducing dependency on exotic or single-source reagents. This diversification of the supply base significantly enhances supply chain reliability and mitigates the risk of production stoppages due to raw material shortages. Furthermore, the elimination of resolution steps means that the entire batch of synthesized material is potentially usable, effectively doubling the theoretical yield compared to racemic synthesis followed by separation. This efficiency translates directly into substantial cost savings and a reduced environmental footprint, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the removal of inefficient separation processes and the reduction of total step count. Traditional methods often discard half of the produced material during chiral resolution, a waste that is completely eliminated here. Additionally, the use of phase transfer catalysis allows reactions to proceed under milder conditions, reducing energy consumption associated with cryogenic cooling or high-pressure systems. The simplified purification protocols, often relying on standard silica gel chromatography or crystallization rather than preparative HPLC, further lower the operational expenditure per kilogram of product. These factors combine to create a manufacturing process that is inherently more cost-effective, allowing for competitive pricing in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the chemical transformations described in the patent ensures consistent output quality, which is critical for maintaining long-term supply contracts. The reactions are tolerant to minor variations in conditions, making the process scalable from pilot plant to commercial production without significant re-optimization. This scalability reduces lead time for high-purity pharmaceutical intermediates, enabling faster response to market demand fluctuations. Moreover, the stability of the intermediates allows for potential stockpiling at key stages of the synthesis, providing a buffer against unexpected disruptions in the upstream supply of starting materials. This resilience is a key asset for any organization aiming to secure a steady flow of critical API ingredients.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route offers significant advantages over prior art. The avoidance of highly toxic reagents where possible, and the use of catalytic amounts of chiral inducers rather than stoichiometric chiral auxiliaries, reduces the generation of hazardous waste. The process is designed to be easily processed, meaning that waste streams are more manageable and treatment costs are lower. The ability to scale this synthesis to multi-ton levels without compromising safety or purity makes it an ideal candidate for industrial adoption. Companies adopting this technology can demonstrate a commitment to sustainable manufacturing practices, a growing requirement for inclusion in the supply chains of major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Triptolide synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of this supply source.
Q: What distinguishes this Triptolide synthesis from previous methods?
A: Unlike earlier methods that produced racemic mixtures or suffered from low yields and complex operations, this patent utilizes a chiral phase transfer catalyst to achieve high stereoselectivity from simple starting materials, significantly shortening the synthetic route.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process employs mild reaction conditions and avoids extreme cryogenic temperatures or hazardous reagents where possible, making it highly amenable to scale-up for industrial API manufacturing.
Q: What are the primary starting materials for this route?
A: The synthesis begins with commercially available 5-methoxy-2-tetralone and 2-(2-iodoethyl)-1,4-butyrolactone, ensuring a stable and cost-effective supply chain foundation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triptolide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for complex molecules like Triptolide. Our team of expert chemists has extensively evaluated the methodology described in CN101638426A and possesses the technical capability to execute this route with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Triptolide intermediate meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug development programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective, and high-quality supply of Triptolide intermediates, accelerating your path from discovery to market.
