Revolutionizing Pyridazinone Production: A Green One-Pot Nickel-Catalyzed Strategy for Commercial Scale-up
Introduction to Advanced Pyridazinone Synthesis
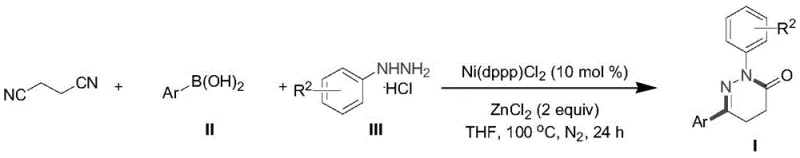
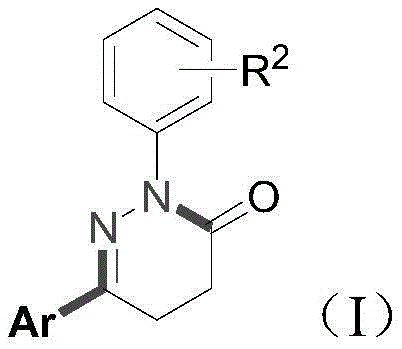
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to construct nitrogen-containing heterocyclic scaffolds, particularly the biologically privileged 4,5-dihydropyridazin-3-one core. Recent advancements disclosed in patent CN114057650A introduce a transformative one-pot methodology that addresses long-standing synthetic bottlenecks. This innovative approach leverages a nickel-catalyzed tandem cyclization strategy, merging succinonitrile, arylboronic acids, and phenylhydrazine hydrochlorides into a unified reaction vessel. For R&D directors and process chemists, this represents a paradigm shift from linear, multi-step sequences to a convergent, high-efficiency protocol. The ability to access diverse substituted pyridazinones through a single operational sequence not only accelerates lead optimization but also lays the groundwork for robust commercial manufacturing. By eliminating the need for pre-functionalized complex substrates, this technology offers a streamlined entry point for producing high-purity pharmaceutical intermediates with superior step economy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 4,5-dihydropyridazin-3-one framework has relied on classical methodologies that are increasingly viewed as inefficient for modern process chemistry. Traditional routes often involve the reaction of Wittig reagents with aryl hydrazones or the condensation of alpha-keto esters with hydrazinocarbonyl acetates. These legacy processes are plagued by significant drawbacks, including the requirement for harsh reaction conditions that can degrade sensitive functional groups. Furthermore, the substrates employed in these conventional methods frequently possess complicated structures that necessitate additional synthetic steps prior to the cyclization event. This results in poor atom economy and low step economy, driving up both the cost of goods sold (COGS) and the environmental footprint of the manufacturing process. The reliance on multiple isolation and purification stages between steps further exacerbates yield losses and extends production lead times, making these methods suboptimal for large-scale supply chains.
The Novel Approach
In stark contrast, the novel one-pot method described in the patent data utilizes a green, low-toxicity transition metal nickel catalyst to drive a cascade reaction directly from simple, commercially available starting materials. By employing succinonitrile—a cheap and abundant commodity chemical—as the foundational building block, the new route bypasses the need for complex substrate preparation. The reaction proceeds through a sophisticated sequence of cyano group addition, intramolecular condensation, and subsequent cyclization, all occurring within a single pot under mild thermal conditions (100°C). This approach not only simplifies the operational workflow but also enhances chemical selectivity and functional group tolerance. The elimination of external oxidants and the use of a robust nickel catalytic system ensure that the process remains environmentally benign while delivering high yields across a broad scope of substrates, including those with electron-withdrawing and electron-donating groups.
Mechanistic Insights into Ni-Catalyzed Tandem Cyclization
The core of this technological breakthrough lies in the unique reactivity of the Ni(dppp)Cl2 catalyst system in conjunction with a Lewis acid additive, specifically zinc chloride. Mechanistically, the reaction initiates with the activation of the nitrile groups in succinonitrile by the nickel center, facilitating a nucleophilic attack or addition process involving the arylboronic acid species. This initial step is critical for establishing the carbon-carbon bond framework necessary for the subsequent ring closure. The presence of zinc chloride plays a pivotal role as a Lewis acid promoter, likely coordinating with the nitrogen atoms or the nitrile groups to enhance their electrophilicity and stabilize key intermediates during the transformation. This synergistic interaction between the transition metal and the Lewis acid allows for the seamless progression from linear precursors to the cyclic hydrazine intermediate without the accumulation of stable byproducts that typically hinder such cascade reactions.
Following the initial addition, the system undergoes an intramolecular condensation driven by the proximity of the reactive centers generated in the first stage. This is followed by a final cyclization event that aromatizes or stabilizes the six-membered dihydropyridazinone ring. The entire catalytic cycle is designed to be redox-neutral regarding the metal center in many steps, avoiding the need for stoichiometric oxidants which often generate heavy metal waste. This mechanistic elegance ensures that the reaction maintains high fidelity even with sterically demanding substrates, such as ortho-substituted arylboronic acids or heterocyclic variants like thiophene and naphthyl derivatives. The result is a highly controlled synthesis where impurity profiles are minimized, a crucial factor for meeting the stringent purity specifications required in API manufacturing.
How to Synthesize 4,5-Dihydropyridazin-3-one Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires precise control over reaction parameters to maximize yield and reproducibility. The protocol dictates a specific molar ratio of reactants, typically favoring an excess of the boronic acid and succinonitrile relative to the phenylhydrazine hydrochloride to drive the equilibrium forward. The choice of solvent is equally critical, with tetrahydrofuran (THF) identified as the optimal medium for solubilizing the diverse range of organic and inorganic components while maintaining catalyst stability. The detailed standardized synthesis steps, including specific workup procedures and purification techniques like column chromatography, are outlined below to ensure consistent production of high-quality intermediates.
- Combine succinonitrile, arylboronic acid, phenylhydrazine hydrochloride, Ni(dppp)Cl2 catalyst, and ZnCl2 additive in THF solvent.
- Heat the reaction mixture to 100°C under a nitrogen atmosphere and stir for 24 hours to facilitate tandem cyclization.
- Upon completion, perform standard post-treatment including extraction, drying, and column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot nickel-catalyzed methodology translates into tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing succinonitrile and generic arylboronic acids, manufacturers can source inputs from a wide array of global suppliers, mitigating the risk of single-source dependency that often plagues complex custom syntheses. This diversification of the supply base enhances supply continuity and provides significant leverage in price negotiations. Furthermore, the consolidation of multiple synthetic steps into a single reactor run drastically reduces the consumption of solvents, energy, and labor hours associated with intermediate isolations. This operational efficiency directly correlates to a lower cost of production, allowing for more competitive pricing strategies in the final API market.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of expensive reagents and the reduction of unit operations. Traditional methods often require costly protecting group strategies or specialized reagents like Wittig salts, which are expensive to produce and handle. In contrast, this nickel-catalyzed route uses commodity chemicals and a catalytic amount of a relatively inexpensive first-row transition metal. The absence of stoichiometric metal oxidants further reduces waste disposal costs, which are a significant hidden expense in fine chemical manufacturing. By streamlining the process flow, facilities can achieve higher throughput with existing infrastructure, effectively lowering the capital expenditure required per kilogram of output.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain reliability. The tolerance for various functional groups means that the same core process can be adapted to produce a wide library of analogues without needing to requalify entirely new synthetic routes for each derivative. This flexibility allows manufacturers to respond rapidly to changing market demands or clinical trial requirements. Additionally, the use of stable, shelf-stable reagents minimizes the risks associated with the degradation of sensitive starting materials during storage and transport. This stability ensures that production schedules can be maintained consistently, reducing the likelihood of delays caused by material quality issues.
- Scalability and Environmental Compliance: From a sustainability perspective, this method aligns perfectly with modern green chemistry principles, which is increasingly a prerequisite for doing business with top-tier pharmaceutical companies. The reduction in solvent usage and the avoidance of toxic oxidants simplify the waste treatment process, ensuring easier compliance with stringent environmental regulations. The process has been demonstrated to be scalable, moving smoothly from milligram to gram scales without loss of efficiency, which de-risks the transition to multi-ton commercial production. This scalability ensures that as demand for the final drug product grows, the supply of the intermediate can be ramped up seamlessly without requiring major process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential partners and licensees.
Q: What are the key advantages of this nickel-catalyzed method over traditional Wittig reagent approaches?
A: Unlike traditional methods requiring harsh conditions and complex multi-step syntheses, this one-pot protocol utilizes readily available succinonitrile and operates under mild conditions (100°C), significantly improving atom economy and operational simplicity.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly demonstrates good functional group compatibility and successful expansion to gram-scale synthesis, indicating strong potential for commercial scale-up due to the use of stable, commercially available substrates.
Q: Does this process require expensive metal oxidants or specialized ligands?
A: No, the process is designed to be green and efficient, avoiding the use of additional metal oxidants. It relies on a cost-effective nickel catalyst system with zinc chloride as an additive, reducing overall material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-Dihydropyridazin-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed one-pot synthesis for the next generation of therapeutic agents. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this academic innovation into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move from benchtop discovery to full-scale manufacturing without interruption. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced chemistry for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
