Advanced Copper-Catalyzed Synthesis of Polysubstituted Pyrroles for Commercial API Manufacturing
Advanced Copper-Catalyzed Synthesis of Polysubstituted Pyrroles for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust and versatile synthetic routes for heterocyclic scaffolds, particularly pyrroles, which serve as critical building blocks for numerous bioactive molecules. Patent CN110746335A introduces a groundbreaking methodology for the construction of polysubstituted pyrrole compounds through a copper-catalyzed cascade reaction involving enaminones and α-diazo esters. This innovation represents a significant leap forward in medicinal chemistry, offering a streamlined pathway to complex architectures that are prevalent in drugs such as atorvastatin and tolmetin. By leveraging a carbene insertion and aryl migration mechanism, this technology enables the precise assembly of highly functionalized pyrrole cores under relatively mild thermal conditions. For R&D directors and process chemists, this patent provides a valuable blueprint for accessing diverse chemical space with high efficiency. The ability to modulate substituents at multiple positions on the pyrrole ring allows for the rapid generation of analog libraries, accelerating the drug discovery pipeline while maintaining strict control over product quality and purity profiles.
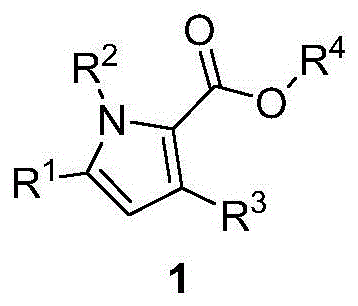
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted pyrroles has relied on classical methodologies such as the Paal-Knorr synthesis, Hantzsch pyrrole synthesis, or cyclization of 1,4-dicarbonyl compounds with amines. While these traditional routes have served the industry for decades, they often suffer from significant drawbacks that hinder modern pharmaceutical manufacturing. Conventional methods frequently require harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to the degradation of sensitive functional groups and limit the scope of compatible substrates. Furthermore, achieving high regioselectivity in the formation of multi-substituted pyrroles remains a persistent challenge, often resulting in complex mixtures of isomers that are difficult and costly to separate. The reliance on less atom-economical reagents and the generation of substantial waste streams also pose environmental and economic burdens. For procurement managers, these inefficiencies translate into higher raw material costs and extended production timelines, while supply chain heads face risks associated with inconsistent yields and purification bottlenecks.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach disclosed in CN110746335A utilizes a transition metal-catalyzed strategy that fundamentally reshapes the synthetic landscape for pyrrole derivatives. By employing enaminones and α-diazo esters as key starting materials, this method bypasses the need for pre-functionalized 1,4-dicarbonyl precursors. The reaction proceeds through a sophisticated cascade involving copper-catalyzed carbene formation, followed by intramolecular cyclization and a subsequent aryl migration. This sequence not only constructs the pyrrole ring with exceptional precision but also installs diverse substituents in a single operational step. The use of copper salts as catalysts offers a cost-effective alternative to precious metals like rhodium or ruthenium, significantly reducing the overall cost of goods. Moreover, the reaction conditions are remarkably mild, typically operating between 10°C and 120°C in common organic solvents, which enhances safety and simplifies process control. This innovative route provides a powerful tool for the cost reduction in pharmaceutical intermediate manufacturing, enabling the production of high-value scaffolds with improved sustainability metrics.
Mechanistic Insights into Copper-Catalyzed Carbene Insertion and Aryl Migration
The core of this technological advancement lies in its unique mechanistic pathway, which orchestrates the formation of the pyrrole ring through a series of well-defined organometallic steps. The reaction initiates with the activation of the α-diazo ester by the copper catalyst, leading to the extrusion of nitrogen gas and the generation of a highly reactive copper-carbene intermediate. This electrophilic species then undergoes a nucleophilic attack by the electron-rich enaminone, forming a new carbon-carbon bond. Subsequent cyclization closes the five-membered ring, setting the stage for the critical aryl migration step. This migration is driven by the restoration of aromaticity and the stabilization of the final pyrrole system. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters, as the choice of copper salt (e.g., CuCl, Cu(OTf)2, [Cu(MeCN)4]PF6) and ligand environment can profoundly influence the rate of carbene formation and the fidelity of the migration. The patent data indicates that the reaction tolerates a wide range of electronic environments, suggesting a robust catalytic cycle that is resilient to steric and electronic variations in the substrate.
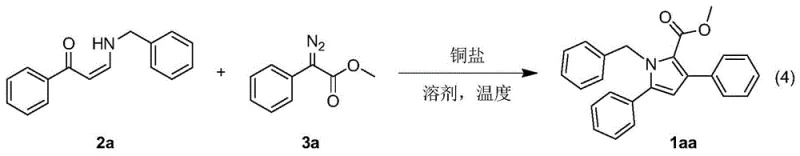
From an impurity control perspective, this mechanism offers distinct advantages over random cyclization processes. The directed nature of the carbene insertion and the specific aryl migration pathway minimize the formation of regioisomeric byproducts, which are common pitfalls in traditional pyrrole synthesis. The high selectivity ensures that the final product profile is clean, simplifying downstream purification and reducing the burden on analytical quality control labs. For process chemists, this means fewer chromatographic steps and higher overall recovery rates. The ability to fine-tune the reaction by adjusting the molar ratio of enaminone to diazo ester (ranging from 1:0.1 to 1:5) further allows for the suppression of side reactions such as dimerization of the diazo compound. This level of mechanistic control is essential for ensuring the consistent quality required for GMP manufacturing of active pharmaceutical ingredients.
How to Synthesize Polysubstituted Pyrrole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the preparation of the two key synthons: the enaminone and the α-diazo ester. The patent outlines a straightforward protocol where the enaminone is generated via refluxing a ketone derivative with an amine in ethanol, while the diazo ester is prepared using DBU-mediated diazo transfer in acetonitrile. Once these precursors are ready, the coupling reaction is initiated by mixing them with a catalytic amount of copper salt in a solvent such as acetonitrile, DCE, or toluene. The detailed standardized synthesis steps provided below offer a comprehensive guide for executing this transformation with high reproducibility. Following these protocols ensures that the delicate balance between carbene generation and capture is maintained, leading to optimal yields.
- Preparation of Enaminone Precursors: Reflux 1-keto-1-aryl-3-(N,N-dimethyl)amino-2-propene with appropriate amines in ethanol to generate the enaminone intermediate.
- Synthesis of Alpha-Diazo Esters: React phenylacetates with p-toluenesulfonyl azide in acetonitrile using DBU as a base under microwave irradiation to form the diazo compound.
- Copper-Catalyzed Cyclization: Combine the enaminone and alpha-diazo ester with a copper salt catalyst in a suitable solvent (e.g., acetonitrile, DCE) and heat to induce carbene insertion and aryl migration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed synthesis offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the accessibility and cost-effectiveness of the raw materials. Enaminones and α-diazo esters are derived from commodity chemicals such as acetophenones, amines, and phenylacetic acid derivatives, which are widely available in the global market. This abundance mitigates supply risk and prevents bottlenecks associated with exotic or proprietary reagents. Furthermore, the elimination of expensive noble metal catalysts in favor of abundant copper salts results in substantial cost savings in raw material expenditure. The simplified workup procedure, which typically involves standard aqueous extraction and silica gel chromatography, reduces the consumption of specialized resins and solvents, thereby lowering the overall environmental footprint and waste disposal costs.
- Cost Reduction in Manufacturing: The shift from precious metal catalysts to copper-based systems drastically lowers the catalyst cost per kilogram of product. Additionally, the high atom economy of the cascade reaction minimizes waste generation, leading to reduced expenses in waste treatment and disposal. The mild reaction conditions also translate to lower energy consumption for heating and cooling, contributing to a more sustainable and economically viable manufacturing process.
- Enhanced Supply Chain Reliability: By utilizing broadly available starting materials, manufacturers can diversify their supplier base and reduce dependency on single-source vendors. The robustness of the reaction conditions ensures consistent batch-to-batch performance, which is critical for maintaining uninterrupted supply lines to downstream API producers. This reliability is further bolstered by the method's tolerance to various functional groups, allowing for flexibility in sourcing substituted precursors without compromising the final product quality.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in batch reactors with standard agitation and temperature control. The use of common organic solvents facilitates solvent recovery and recycling, aligning with green chemistry principles. The absence of highly toxic reagents or extreme pressure conditions simplifies regulatory compliance and safety assessments, accelerating the timeline for technology transfer from lab to commercial scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polysubstituted pyrrole synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on reaction parameters, substrate compatibility, and potential applications. Understanding these nuances is vital for teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this copper-catalyzed pyrrole synthesis over traditional methods?
A: This method offers superior regioselectivity and milder reaction conditions compared to classical Paal-Knorr syntheses. It utilizes readily available enaminones and diazo esters, allowing for diverse structural modifications at the R1, R2, R3, and R4 positions without requiring harsh acidic or basic environments.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the process is designed for scalability. It employs common organic solvents like acetonitrile and toluene, and the catalyst loading can be optimized. The workup involves standard extraction and column chromatography, which can be adapted for large-scale crystallization or distillation processes.
Q: What types of substituents are tolerated in this polysubstituted pyrrole synthesis?
A: The method demonstrates excellent functional group tolerance. It accommodates various aryl groups (phenyl, substituted phenyl), benzyl, allyl, and alkyl groups at the nitrogen position, as well as different ester groups (methyl, ethyl, benzyl, allyl) at the 2-position of the pyrrole ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrrole Supplier
As the demand for complex heterocyclic intermediates continues to grow, partnering with an experienced CDMO is essential for navigating the complexities of process development and scale-up. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our team of expert chemists is adept at optimizing copper-catalyzed reactions to meet stringent purity specifications, utilizing our rigorous QC labs to guarantee that every batch meets the highest industry standards. We understand the critical importance of timeline and cost efficiency in the pharmaceutical sector and are committed to delivering solutions that enhance your competitive edge.
We invite you to collaborate with us to explore the full potential of this innovative pyrrole synthesis technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions. Let us be your trusted partner in bringing high-quality polysubstituted pyrrole intermediates to market faster and more efficiently.
