Advanced Palladium-Catalyzed Synthesis of Indole and Benzoxazine Intermediates for Commercial Scale-Up
Introduction to Next-Generation Heterocyclic Synthesis
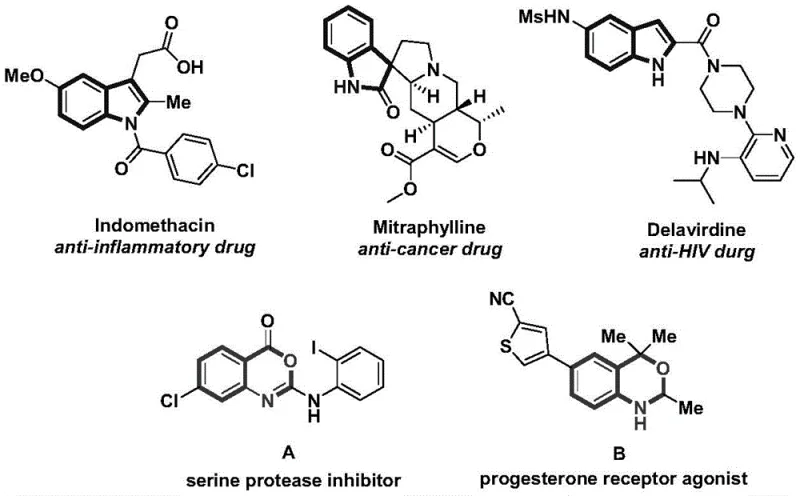
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, selective, and cost-effective synthetic routes. A significant breakthrough in this domain is detailed in patent CN115246786A, which discloses a versatile preparation method for indole and benzoxazine compounds. These nitrogen-containing heterocycles are foundational scaffolds in medicinal chemistry, serving as the core structure for numerous bioactive molecules. As illustrated in the biological context, indole derivatives such as Indomethacin function as potent anti-inflammatory agents, while Mitraphylline exhibits anti-cancer properties, and Delavirdine is a critical anti-HIV drug. Similarly, benzoxazine frameworks are pivotal in the development of serine protease inhibitors and progesterone receptor agonists. The ability to access these privileged structures efficiently is paramount for any reliable pharmaceutical intermediate supplier aiming to support the global drug development pipeline.
The technology described in this patent represents a paradigm shift from conventional multi-step syntheses to a streamlined, transition metal-catalyzed carbonylation approach. By leveraging a palladium-catalyzed system, this method achieves high reaction efficiency and exceptional substrate compatibility. The process is not merely a laboratory curiosity but is designed with scalability in mind, utilizing cheap and readily available starting materials. This aligns perfectly with the industry's demand for cost reduction in API manufacturing, where the complexity of the synthetic route directly correlates with the final cost of goods. The innovation lies in its divergent nature; a single set of initial reactants can be channeled towards two distinct classes of valuable heterocycles simply by modulating the reaction additives, thereby maximizing the utility of the synthetic platform.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of indole and benzoxazine rings has relied on a variety of classical methodologies, many of which suffer from significant drawbacks when applied to industrial settings. Conventional routes often involve harsh reaction conditions, the use of toxic reagents, or multiple protection and deprotection steps that erode overall yield and increase waste generation. Furthermore, many existing carbonylation reactions, while theoretically powerful, have seen limited practical application for these specific skeletons due to issues with catalyst stability, poor regioselectivity, or the requirement for high-pressure carbon monoxide gas, which poses severe safety challenges in a plant environment. The lack of a unified strategy that can access both indole and benzoxazine cores from common precursors has historically forced process chemists to develop entirely separate supply chains for these related structural motifs, leading to inefficiencies in procurement and inventory management.
The Novel Approach
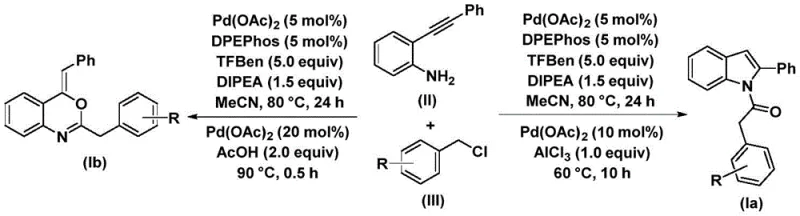
The novel approach presented in the patent data overcomes these hurdles through a sophisticated yet operationally simple palladium-catalyzed carbonylative cyclization. This method utilizes 2-phenylethynylamine and benzyl chloride as the primary building blocks. The reaction proceeds under relatively mild thermal conditions (70~90°C) in a standard organic solvent like acetonitrile. The true brilliance of this methodology is its switchable selectivity. By introducing specific additives in the second stage of the reaction—aluminum chloride for indoles or acetic acid for benzoxazines—the pathway diverges to yield the desired product with high fidelity. This eliminates the need for distinct synthetic lines for these two classes of compounds. The process boasts high reaction efficiency and good functional group tolerance, meaning that complex, substituted derivatives can be synthesized without compromising the integrity of sensitive moieties, a critical factor for the synthesis of high-purity OLED material or advanced pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
To fully appreciate the robustness of this synthesis for commercial scale-up of complex pharmaceutical intermediates, one must understand the underlying catalytic cycle. The reaction initiates with the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. Crucially, the carbon monoxide required for the carbonylation is not supplied as a hazardous gas but is released in situ from phenyl 1,3,5-tricarboxylate (a solid CO source), which inserts into the benzyl-palladium bond to form an acyl-palladium species. This in situ generation of CO significantly enhances safety and operational simplicity. Subsequently, the 2-phenylethynylamine acts as a nucleophile, attacking the acyl-palladium intermediate. Following reductive elimination, an amide compound is formed, which serves as the key precursor for the final cyclization step.
The final transformation is where the selectivity is determined. In the presence of a Lewis acid like aluminum chloride, the system favors an intramolecular cyclization that leads to the formation of the indole ring system (Structure Ia). Conversely, when acetic acid is used as the additive, the reaction pathway shifts to favor the formation of the benzoxazine ring (Structure Ib). This mechanistic divergence allows for precise control over the product outcome without altering the primary feedstock. The catalyst system, comprising palladium acetate and the bidentate ligand DPEPhos, ensures high turnover and stability throughout the prolonged reaction times (24~48 hours for the first stage). This deep understanding of the mechanism allows process engineers to fine-tune parameters for reducing lead time for high-purity intermediates while maintaining strict quality control standards.
How to Synthesize Indole and Benzoxazine Derivatives Efficiently
Implementing this synthesis in a production environment requires careful attention to the sequential addition of reagents and temperature control. The process is divided into two distinct thermal stages to maximize conversion and selectivity. The initial carbonylation and amidation step requires a longer duration to ensure complete consumption of the benzyl chloride and formation of the amide intermediate. The subsequent cyclization step is faster but requires precise dosing of the selectivity-determining additive. The detailed standardized synthetic steps below outline the exact molar ratios and conditions optimized in the patent examples to achieve yields ranging from 45% to 80%, depending on the specific substrate substituents.
- Combine palladium acetate, DPEPhos, phenyl 1,3,5-tricarboxylate, DIPEA, 2-phenylethynylamine, and benzyl chloride in acetonitrile.
- Heat the mixture to 80°C for 24 hours to form the intermediate amide species.
- Add aluminum chloride for indole synthesis or acetic acid for benzoxazine synthesis, then heat at 60-90°C to complete cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. Since both indole and benzoxazine products stem from the same pool of starting materials (benzyl chlorides and ethynyl amines), purchasing teams can consolidate orders, leverage volume discounts, and reduce the complexity of vendor management. This consolidation directly contributes to cost reduction in pharmaceutical intermediate manufacturing by minimizing the overhead associated with sourcing and qualifying multiple distinct sets of reagents. Furthermore, the use of solid CO surrogates eliminates the logistical and safety burdens associated with handling high-pressure carbon monoxide cylinders, thereby lowering insurance and compliance costs.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of commodity chemicals. Benzyl chlorides and simple aniline derivatives are produced on a massive industrial scale globally, ensuring stable pricing and availability. The catalytic system uses palladium, which, while precious, is employed at low loadings (5 mol%) and can potentially be recovered and recycled in a continuous flow setup, further driving down the cost per kilogram. The elimination of hazardous gas handling infrastructure also represents a significant capital expenditure saving for manufacturing sites.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The tolerance for various functional groups means that even if a specific substituted starting material faces a temporary shortage, alternative analogs can often be synthesized using the same protocol without extensive re-optimization. This flexibility ensures continuity of supply for downstream API manufacturers. Additionally, the reaction operates in acetonitrile, a common solvent with a well-established global supply chain, reducing the risk of solvent-related bottlenecks.
- Scalability and Environmental Compliance: From an environmental perspective, the atom economy of this carbonylation reaction is superior to many traditional condensation reactions that generate stoichiometric amounts of salt waste. The ability to run the reaction at moderate temperatures (80°C) reduces energy consumption compared to high-temperature pyrolysis methods. The streamlined workup procedure, involving simple filtration and chromatography, minimizes solvent usage and waste generation, aligning with modern green chemistry principles and facilitating easier regulatory approval for commercial scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing a realistic overview of what process development teams can expect when adopting this route for their specific projects.
Q: What is the key advantage of this carbonylation method over traditional indole synthesis?
A: This method utilizes a divergent palladium-catalyzed carbonylation strategy that allows for the selective synthesis of either indoles or benzoxazines from the same starting materials simply by changing the additive (AlCl3 vs. AcOH), offering superior flexibility and atom economy compared to multi-step traditional routes.
Q: Are the starting materials for this process commercially viable for large-scale production?
A: Yes, the process relies on readily available and inexpensive starting materials such as benzyl chlorides and 2-phenylethynylamines, which significantly reduces the raw material costs and simplifies the supply chain logistics for commercial manufacturing.
Q: What kind of functional group tolerance does this catalytic system exhibit?
A: The protocol demonstrates excellent substrate compatibility, tolerating various substituents on the benzyl chloride including electron-donating groups like methyl and tert-butyl, as well as electron-withdrawing groups like fluoro and chloro, ensuring broad applicability for diverse API intermediate libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess the technical expertise to translate these patented laboratory methods into robust, GMP-compliant manufacturing processes. Our facilities are equipped to handle diverse synthetic pathways, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that consistency is key; therefore, our rigorous QC labs and stringent purity specifications ensure that every batch of indole or benzoxazine intermediate meets the exacting standards required by top-tier pharmaceutical companies.
We invite you to collaborate with us to evaluate the feasibility of this route for your specific pipeline candidates. Our technical team is ready to provide a Customized Cost-Saving Analysis that compares this novel carbonylation method against your current supply chain. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage through superior chemical manufacturing.
