Advanced Ruthenium-Carborane Catalysis for Efficient Isocoumarin Production and Commercial Scale-Up
Advanced Ruthenium-Carborane Catalysis for Efficient Isocoumarin Production and Commercial Scale-Up
The landscape of heterocyclic synthesis is undergoing a significant transformation driven by the need for greener, more efficient catalytic systems. A pivotal advancement in this domain is detailed in Chinese Patent CN114213383B, which discloses a novel method for synthesizing isocoumarin compounds utilizing a specialized half-sandwich ruthenium complex. This technology represents a paradigm shift from traditional high-energy processes to ambient condition catalysis, offering profound implications for the manufacturing of pharmaceutical intermediates and fine chemicals. The core innovation lies in the deployment of a ruthenium complex featuring an ortho-carborane-based benzoxazole ligand, which acts as a highly robust and active catalyst. Unlike conventional transition metal catalysts that often demand stringent exclusion of air and moisture, this specific ruthenium species demonstrates remarkable stability against environmental factors, thereby simplifying operational protocols and reducing infrastructure costs associated with inert atmosphere handling. For R&D directors and process chemists, this patent offers a compelling route to access bioactive isocoumarin scaffolds, which are prevalent in natural products exhibiting antiviral, antibacterial, and anti-inflammatory properties, with unprecedented ease and efficiency.
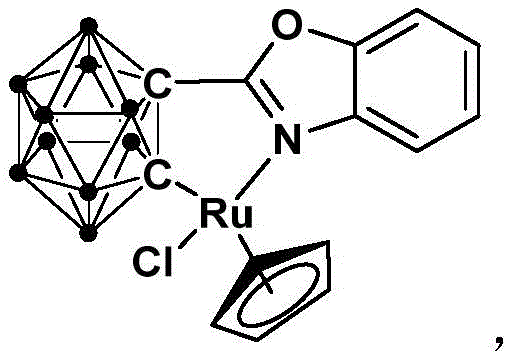
The structural integrity of the catalyst, as depicted in the patent documentation, is fundamental to its performance. The integration of the carborane cluster provides a unique electronic and steric environment around the ruthenium center, facilitating the activation of substrates under mild thermal conditions. This stability is not merely a laboratory curiosity but a critical commercial asset, ensuring consistent catalytic activity over extended periods and reducing the frequency of catalyst replenishment. Furthermore, the ability to operate at room temperature drastically lowers the energy footprint of the synthesis, aligning with modern sustainability goals in chemical manufacturing. As we delve deeper into the technical specifics, it becomes evident that this methodology addresses multiple pain points simultaneously: substrate accessibility, reaction severity, and product isolation complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isocoumarin core has relied on methodologies that are fraught with synthetic inefficiencies and operational hazards. Traditional routes often involve the intramolecular cyclization of o-alkynyl benzoic acid derivatives or cascade reactions between o-halobenzamides and 1,3-diketones. These legacy approaches suffer from significant drawbacks that hinder their scalability and economic viability. Firstly, the preparation of the requisite starting materials, such as o-alkynyl benzoic acids, is often multi-step and labor-intensive, requiring harsh reagents and protecting group strategies that inflate the overall cost of goods. Secondly, these reactions frequently necessitate elevated temperatures and prolonged reaction times, leading to high energy consumption and increased safety risks in a production setting. Moreover, the harsh conditions often promote side reactions, resulting in complex impurity profiles that complicate downstream purification and reduce overall yield. The reliance on sensitive reagents also mandates the use of specialized equipment and rigorous exclusion of moisture, further escalating capital expenditure and operational complexity for manufacturing facilities.
The Novel Approach
In stark contrast, the methodology outlined in CN114213383B introduces a streamlined, direct condensation strategy that circumvents these historical bottlenecks. By employing 2-oxosulfurylidenethanone compounds as readily accessible raw materials, the process eliminates the need for difficult-to-prepare precursors. The use of the ortho-carborane-based ruthenium catalyst enables the reaction to proceed efficiently at room temperature, a condition that is virtually unheard of for such transformative cyclizations. This mildness not only preserves the integrity of sensitive functional groups on the substrate but also significantly reduces the formation of thermal degradation by-products. The reaction system utilizes common inorganic bases like potassium carbonate or sodium carbonate in toluene, avoiding the need for expensive or hazardous strong bases. Consequently, the workup procedure is simplified, often requiring only concentration and standard column chromatography to achieve high-purity products. This approach exemplifies the principles of green chemistry by minimizing waste generation and energy usage while maximizing atom economy and operational safety.
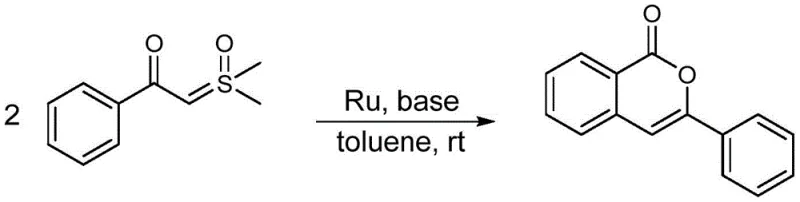
Mechanistic Insights into Ru-Catalyzed Cyclization
The efficacy of this transformation is rooted in the unique mechanistic pathway facilitated by the half-sandwich ruthenium center. The catalytic cycle likely initiates with the coordination of the sulfur ylide substrate to the ruthenium metal, activated by the electron-deficient nature of the carborane-modified ligand sphere. This interaction promotes the deprotonation or activation of the methylene group adjacent to the carbonyl, generating a reactive nucleophilic species in situ. The presence of the mild base assists in this activation step without inducing non-specific decomposition of the substrate. Subsequently, an intramolecular nucleophilic attack occurs, closing the lactone ring to form the isocoumarin skeleton. The robustness of the Ru-C and Ru-N bonds within the catalyst framework ensures that the metal center remains intact throughout the turnover, preventing the leaching of ruthenium into the product stream, which is a critical consideration for pharmaceutical applications where heavy metal limits are strictly regulated. The steric bulk of the carborane cage may also play a role in directing the regioselectivity of the cyclization, ensuring that the desired isocoumarin isomer is formed exclusively.
From an impurity control perspective, the room temperature operation is a decisive factor. Thermal stress is a primary driver of side reactions such as polymerization, over-oxidation, or rearrangement in organic synthesis. By maintaining the reaction at ambient temperatures (typically 20-25°C), the kinetic energy available to drive these parasitic pathways is minimized. The patent data indicates yields reaching as high as 96%, which implies a suppression of by-product formation to negligible levels. This high selectivity simplifies the purification burden, allowing for straightforward crystallization or chromatographic separation. Furthermore, the catalyst's insensitivity to air and water means that trace moisture in the solvent or reagents does not deactivate the catalytic species or generate hydrolysis by-products, ensuring batch-to-batch consistency. This level of control is essential for producing high-purity pharmaceutical intermediates where impurity profiles must be tightly managed to meet regulatory standards.
How to Synthesize Isocoumarin Derivatives Efficiently
Implementing this technology requires a precise understanding of the catalyst preparation and the subsequent coupling reaction. The process is designed to be modular, allowing for the independent optimization of catalyst synthesis and substrate conversion. The following sections outline the procedural logic derived from the patent examples, emphasizing the critical parameters for success. For detailed operational specifics, manufacturers should refer to the standardized protocols below which ensure reproducibility and safety.
- Prepare the half-sandwich ruthenium complex containing ortho-carborane-based benzoxazole by reacting o-carborane with n-BuLi, followed by bromobenzoxazole and [CpRuCl2]2.
- Dissolve the 2-oxosulfurylidenethanone substrate, the ruthenium catalyst (0.2-0.5 mol%), and a base (K2CO3 or Na2CO3) in toluene.
- Stir the reaction mixture at room temperature for 6-10 hours to allow condensation and cyclization to occur.
- Concentrate the reaction mixture and purify the crude product via silica gel column chromatography to isolate the high-purity isocoumarin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this ruthenium-catalyzed process offers tangible economic and logistical benefits that extend beyond simple yield improvements. The shift from high-temperature, multi-step legacy processes to a one-pot, room-temperature condensation fundamentally alters the cost structure of isocoumarin manufacturing. By eliminating the need for energy-intensive heating and cooling cycles, facilities can achieve significant reductions in utility costs. Moreover, the use of stable, non-sensitive catalysts reduces the dependency on specialized storage infrastructure and expensive inert gas supplies, lowering overhead expenses. The simplicity of the workup procedure, which avoids complex quenching or extraction steps, translates to shorter cycle times and higher throughput in existing reactor trains. These factors collectively contribute to a more resilient and cost-effective supply chain for fine chemical intermediates.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the elimination of extreme reaction conditions and the simplification of purification. Since the reaction proceeds at room temperature, there is no requirement for steam heating or cryogenic cooling, leading to substantial energy savings. Additionally, the high catalytic efficiency allows for low catalyst loading (0.2-0.5 mol%), minimizing the consumption of the precious metal ruthenium. The high selectivity reduces the volume of solvent and silica gel required for purification, further driving down variable costs. The stability of the catalyst also implies a longer shelf life, reducing waste associated with expired reagents.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commercially available and stable raw materials. The substrates, 2-oxosulfurylidenethanone compounds, are accessible via established synthetic routes, and the catalyst itself can be prepared in bulk and stored without degradation due to its air and water stability. This robustness mitigates the risk of production delays caused by reagent sensitivity or transportation constraints associated with hazardous materials. The broad substrate scope means that a single catalytic platform can produce a diverse library of isocoumarin derivatives, allowing manufacturers to respond flexibly to changing market demands without retooling entire production lines.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is facilitated by the exothermic nature of the condensation being manageable at room temperature, reducing the risk of thermal runaway incidents. The process generates minimal waste residues, aligning with increasingly stringent environmental regulations regarding solvent discharge and heavy metal contamination. The absence of toxic by-products simplifies waste treatment protocols, lowering compliance costs. Furthermore, the use of toluene, a common industrial solvent, ensures compatibility with existing recovery and recycling systems, promoting a circular economy approach within the manufacturing facility.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries based on the patent specifications. These insights address concerns regarding catalyst handling, substrate compatibility, and process robustness, providing a clear picture of the operational reality.
Q: Is the ruthenium-carborane catalyst sensitive to air or moisture?
A: No, the half-sandwich ruthenium complex described in patent CN114213383B exhibits exceptional stability. It is insensitive to both air and water, allowing for stable storage and handling under ambient conditions without the need for rigorous inert atmosphere techniques typically required for organometallic catalysts.
Q: What are the typical reaction conditions for this isocoumarin synthesis?
A: The process operates under remarkably mild conditions. The reaction proceeds at room temperature in toluene solvent using mild inorganic bases such as potassium carbonate or sodium carbonate. Reaction times typically range from 6 to 10 hours, eliminating the need for high-energy heating protocols.
Q: What is the substrate scope for this catalytic method?
A: The method demonstrates broad universality across various 2-oxosulfurylidenethanone compounds. It successfully tolerates diverse substituents on the aromatic ring, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens (fluorine, chlorine, bromine), yielding products with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isocoumarin Supplier
The technological breakthroughs encapsulated in patent CN114213383B highlight the immense potential of advanced organometallic catalysis in modernizing fine chemical production. At NINGBO INNO PHARMCHEM, we recognize the value of such innovations and possess the technical expertise to translate them into commercial reality. As a seasoned CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly converted into industrial assets. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with state-of-the-art analytical instrumentation, guaranteeing that every batch of isocoumarin intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite forward-thinking organizations to collaborate with us to leverage this efficient synthesis route for their pipeline projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our mastery of ruthenium-catalyzed transformations can optimize your supply chain and accelerate your time to market.
