Advanced Manganese-Mediated Synthesis of High-Purity Fullerene Derivatives for Commercial Scale-Up
Introduction to Advanced Fullerene Functionalization
The landscape of organic electronics and advanced material science is undergoing a significant transformation driven by the need for more efficient and scalable synthesis routes for functional materials. Patent CN110981679A introduces a groundbreaking methodology for producing fullerene derivatives, specifically targeting the fusion of cyclic carbon chains onto the fullerene skeleton. This innovation addresses critical bottlenecks in the manufacturing of acceptor materials for organic thin-film solar cells and biochemical probes. By leveraging a transition metal-mediated reaction in a specialized mixed solvent system, this technology enables the production of high-purity fullerene derivatives under remarkably mild conditions. The strategic shift from hazardous diazo compounds and expensive noble metal catalysts to abundant base metals like manganese represents a paradigm shift in electronic chemical manufacturing. This report analyzes the technical depth of this invention, highlighting its potential to redefine supply chain reliability and cost structures for global buyers seeking a reliable fullerene derivative supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fullerene derivatives fused with cyclic hydrocarbon groups has been plagued by severe operational constraints and safety hazards. Traditional methods often rely on multistage reactions that are not only cumbersome but also inherently dangerous. For instance, the production of the widely used acceptor material PCBM typically involves reacting C60 with diazo compounds, which are thermally unstable and pose significant explosion risks. Furthermore, these conventional pathways frequently require harsh thermal conditions, such as refluxing in o-dichlorobenzene at temperatures reaching 180°C, to facilitate isomerization. Another prevalent approach utilizes cobalt complexes as catalysts; while effective, cobalt is a costly transition metal that complicates the purification process due to the need for rigorous heavy metal removal to meet electronic grade specifications. These factors collectively result in low overall yields, often hovering around 57% for key intermediates, and create substantial barriers to commercial scale-up of complex organic semiconductors.
The Novel Approach
In stark contrast, the method disclosed in the patent utilizes a streamlined, single-step protocol that operates efficiently at room temperature. By employing a mixed solvent system of an aromatic solvent, preferably o-dichlorobenzene (ODCB), and an aprotic polar solvent containing a C=O or S=O bond, such as dimethyl sulfoxide (DMSO) or dimethylformamide (DMF), the solubility of both the fullerene skeleton and the halogenated reactant is maximized. The core innovation lies in the use of inexpensive and abundant metals—specifically manganese, iron, or zinc—to drive the reductive cyclization. This approach eliminates the need for dangerous diazo precursors and expensive cobalt catalysts entirely. Moreover, the reaction conditions are highly generalized, meaning the same protocol can be applied regardless of the ring size being formed, whether it is a cyclopropane, cyclopentane, or cyclohexane structure. This universality simplifies process development and allows for the rapid generation of diverse derivative libraries.
Mechanistic Insights into Metal-Mediated Reductive Cyclization
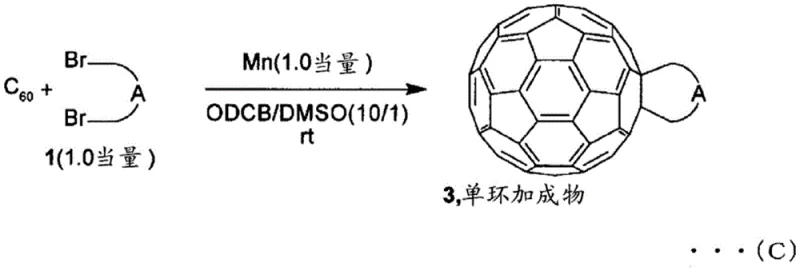
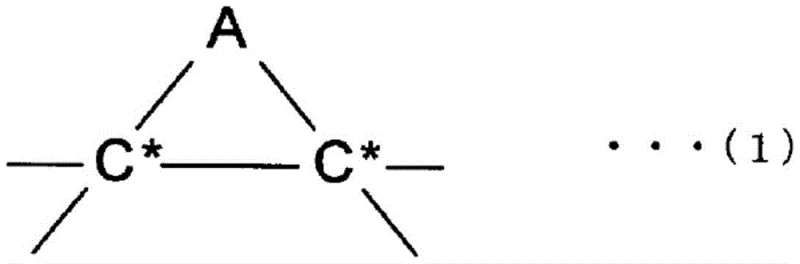
The chemical mechanism underpinning this invention involves the activation of a specific halogenated compound, represented by formula (2), where two halogen atoms (preferably bromine) are connected by a C1-4 linking group. In the presence of the metal reductant, carbon-halogen bonds are cleaved to generate reactive radical or carbanion species that attack adjacent carbon atoms on the fullerene skeleton. The choice of metal and solvent plays a pivotal role in stabilizing these intermediates and directing the reaction pathway. For example, when manganese is used in DMF, the system favors the formation of polycyclic adducts, whereas iron in DMSO selectively produces monocyclic adducts. This level of control is achieved without the need for external light sources or extreme heating, relying instead on the intrinsic redox potential of the metal-solvent combination. The mixed solvent environment is critical; the aromatic component ensures the dissolution of the hydrophobic fullerene cage, while the polar aprotic component facilitates the electron transfer processes necessary for the reductive coupling.
Furthermore, the impurity profile of the resulting product is significantly improved compared to traditional methods. Because the reaction proceeds under mild conditions without high-temperature isomerization steps, the formation of unwanted regioisomers and open-cage byproducts is minimized. The patent data indicates that by adjusting the molar equivalents of the metal and the halogenated compound, manufacturers can selectively target specific adducts. For instance, using 1.0 equivalent of manganese relative to C60 predominantly yields monocyclic adducts, while increasing the metal loading to 3.0 or 9.0 equivalents shifts the distribution toward bicyclic and polycyclic species. This stoichiometric control provides a robust handle for reducing lead time for high-purity fullerene derivatives by eliminating complex chromatographic separations often required to isolate specific isomers from messy reaction mixtures generated by older technologies.
How to Synthesize High-Purity Fullerene Derivatives Efficiently
The synthesis protocol described in the patent offers a straightforward pathway for R&D teams to access novel fullerene architectures. The process begins with the preparation of the halogenated precursor, which can be synthesized via standard bromination techniques, followed by its immediate reaction with the fullerene source in the optimized solvent blend. The beauty of this method lies in its operational simplicity; reactions are conducted under an inert atmosphere at ambient temperature, removing the need for specialized heating mantles or cryogenic cooling systems. Detailed standardized synthetic steps for implementing this technology in a pilot plant setting are outlined below.
- Prepare a mixed solvent system comprising an aromatic solvent such as o-dichlorobenzene (ODCB) and an aprotic polar solvent like DMSO or DMF.
- Add the fullerene skeleton compound (e.g., C60) and the specific halogenated compound (formula 2) to the solvent mixture.
- Introduce the metal reductant (Mn, Fe, or Zn) at room temperature to initiate the cyclization reaction, adjusting stoichiometry to control mono- or bicyclic adduct formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this manganese-mediated technology translates into tangible strategic benefits beyond mere technical novelty. The most immediate impact is seen in the drastic simplification of the supply chain for raw materials. By replacing scarce and price-volatile cobalt catalysts with commodity-grade manganese powder, the exposure to metal price fluctuations is virtually eliminated. Additionally, the removal of diazo compounds from the process removes a major safety liability, potentially lowering insurance premiums and reducing the regulatory burden associated with handling explosive precursors. The ability to run reactions at room temperature also implies a significant reduction in energy consumption, as no heating or cooling cycles are required, contributing to a lower carbon footprint for the manufacturing facility.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling when analyzing the total cost of ownership. The substitution of expensive cobalt complexes with inexpensive manganese or iron powders results in a direct decrease in catalyst costs. Furthermore, the single-step nature of the synthesis, particularly for high-value targets like PCBM, removes an entire unit operation from the production line. In conventional PCBM synthesis, a two-stage process is required involving an initial addition followed by a thermal isomerization; this new method achieves the final structure in one pot. This consolidation of steps reduces labor hours, solvent usage, and waste disposal costs, leading to substantial overall cost savings without compromising on the quality of the final electronic material.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any reliable electronic chemical supplier. The reliance on specialized substrates or difficult-to-source methylenecyclopropane compounds in prior art methods created fragile supply chains prone to disruption. In contrast, the halogenated compounds used in this invention, such as 1,2-bis(bromomethyl)benzene or various dibromoalkanes, are commercially available bulk chemicals with stable supply lines. The robustness of the reaction conditions—tolerating a range of solvents and functioning effectively at room temperature—also means that production is less susceptible to equipment failures or utility interruptions. This resilience ensures consistent delivery schedules for downstream customers in the photovoltaic and semiconductor industries.
- Scalability and Environmental Compliance: Scaling chemical processes from the gram scale to multi-ton production often reveals hidden inefficiencies, but this method is inherently designed for scalability. The use of o-dichlorobenzene and DMSO, while requiring careful handling, is well-established in industrial chemistry, and the absence of gaseous byproducts or highly exothermic events simplifies reactor design. From an environmental perspective, the higher atom economy and the avoidance of heavy metal catalysts simplify the wastewater treatment process. The ability to recover unreacted C60 with high efficiency, as demonstrated in the patent examples where recovery rates exceed 95%, further enhances the sustainability profile by minimizing the loss of the most expensive starting material. This aligns perfectly with modern green chemistry principles and stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fullerene derivative synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on yield optimization and substrate scope.
Q: What are the primary advantages of this manganese-mediated method over conventional cobalt-catalyzed routes?
A: The primary advantage is the elimination of expensive cobalt complexes and dangerous diazo compounds. This method operates under mild room temperature conditions using abundant metals like manganese, significantly lowering raw material costs and safety risks while maintaining high yields.
Q: How does the process control the formation of monocyclic versus bicyclic adducts?
A: Selectivity is precisely controlled by the stoichiometric ratio of the metal reductant and the halogenated compound. For instance, using 1.0 equivalent of manganese favors monocyclic adducts, while higher equivalents (e.g., 3.0 to 9.0) promote the formation of bicyclic or polycyclic structures.
Q: Is this method suitable for the industrial production of PCBM for solar cells?
A: Yes, the patent demonstrates a single-step synthesis of [60]PCBM with a yield of 98%, which is substantially higher than the conventional two-step method yielding approximately 57%. The mild conditions and simplified workflow make it highly suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fullerene Derivative Supplier
The technological advancements detailed in patent CN110981679A represent a significant opportunity for the industry to produce superior organic electronic materials more efficiently. NINGBO INNO PHARMCHEM stands at the forefront of adopting such innovative methodologies to serve our global clientele. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for high-performance solar cells and semiconductor applications, guaranteeing that every batch of fullerene derivative meets the exacting standards of the market.
We invite potential partners to engage with our technical team to explore how this manganese-mediated synthesis can be tailored to your specific product requirements. By leveraging our expertise, you can achieve a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient route. We encourage you to contact our technical procurement team today to request specific COA data for our fullerene portfolio and to discuss route feasibility assessments for your next-generation organic electronic projects.
