Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Applications
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazoles, due to their prevalence in bioactive molecular frameworks. Patent CN113307790B introduces a groundbreaking preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current manufacturing processes. This technology leverages a metal-free oxidative cyclization strategy that eliminates the need for expensive transition metal catalysts and严苛 anhydrous conditions. By utilizing readily available starting materials such as 2-methylquinoline and trifluoroacetimidohydrazide, this innovation offers a streamlined pathway to high-value intermediates used in drug discovery and functional material science. The method demonstrates exceptional substrate tolerance, allowing for the introduction of diverse substituents on both the quinoline and phenyl rings, thereby expanding the chemical space available for medicinal chemists designing next-generation therapeutics.
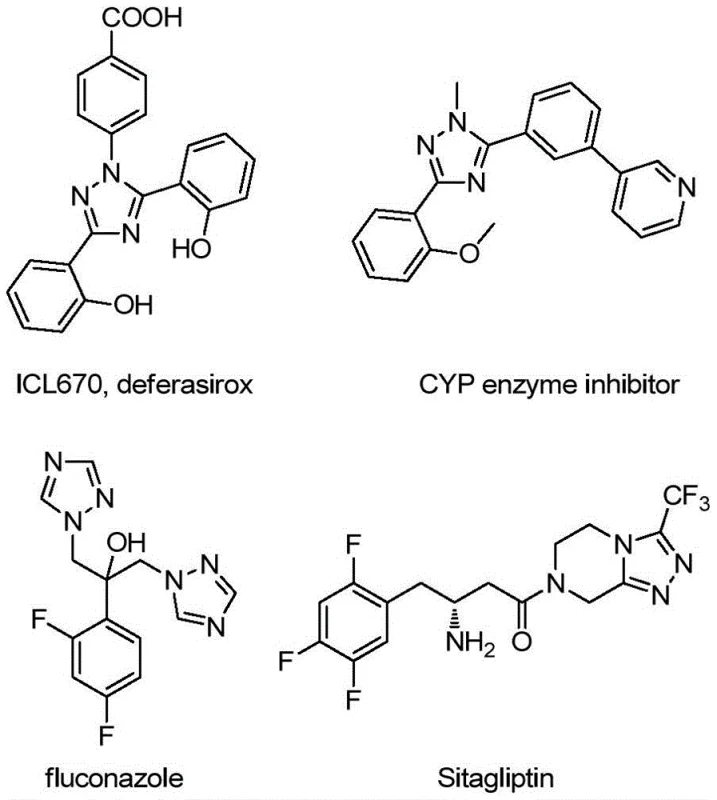
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiency and operational complexity. Traditional protocols often rely on quinoline-2-carboxylic acid as a primary building block, necessitating a tedious five-step reaction sequence to construct the target heterocyclic core. This multi-step approach not only accumulates significant processing costs but also results in a dismal overall yield of approximately 17%, rendering it economically unviable for large-scale commercial applications. Furthermore, conventional methods frequently demand stringent reaction conditions, including strictly anhydrous and oxygen-free environments, which impose heavy burdens on equipment requirements and safety protocols in a manufacturing setting. The reliance on such fragile conditions increases the risk of batch failure and complicates the supply chain continuity for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN113307790B revolutionizes the synthesis landscape by employing a direct oxidative cyclization between 2-methylquinoline and trifluoroacetimidohydrazide. This novel route condenses the synthesis into a single pot, drastically reducing operation time and resource consumption. The reaction proceeds efficiently at moderate temperatures between 80°C and 100°C in common organic solvents like DMSO, eliminating the need for specialized inert atmosphere techniques. By avoiding toxic heavy metal catalysts, this green chemistry approach simplifies downstream purification and ensures the final product meets stringent purity specifications required for API manufacturing. The ability to achieve high conversion rates with simple post-treatment procedures, such as filtration and column chromatography, underscores the practical superiority of this method for industrial adoption.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core of this technological breakthrough lies in the synergistic catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP). Mechanistically, the reaction initiates with the oxidation of the methyl group on the 2-methylquinoline substrate to generate a reactive 2-quinolinecarbaldehyde intermediate in situ. This aldehyde species then undergoes a condensation reaction with the trifluoroacetimidohydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination facilitates an intramolecular electrophilic substitution, followed by aromatization to yield the stable 1,2,4-triazole ring system. The presence of diphenylphosphinic acid as an additive plays a pivotal role in stabilizing reactive intermediates and accelerating the cyclization kinetics, ensuring high selectivity for the desired triazole product over potential side reactions.
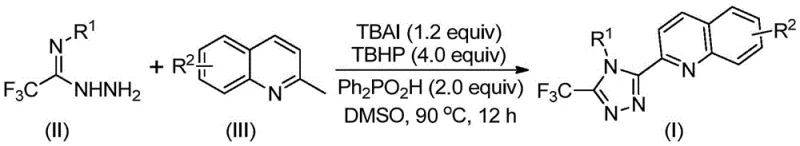
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of complex byproducts often associated with transition metal catalysis. The radical nature of the TBHP-mediated oxidation is well-controlled by the stoichiometric balance of reagents, specifically maintaining a molar ratio of TBAI to TBHP around 1:4. This precise control prevents over-oxidation of the sensitive quinoline moiety or the trifluoromethyl group. Additionally, the absence of metal residues simplifies the impurity profile, reducing the burden on analytical quality control teams who would otherwise need to monitor for trace heavy metals like palladium or copper. The robustness of this free-radical pathway allows for a wide scope of substrates, accommodating electron-donating and electron-withdrawing groups on the aromatic rings without significant loss in efficiency.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent ratios and thermal management to maximize yield. The standard protocol involves charging a reactor with the hydrazide derivative, 2-methylquinoline, TBAI, TBHP aqueous solution, and diphenylphosphinic acid in DMSO. The mixture is then heated to 90°C and maintained for approximately 12 hours to ensure complete conversion. Detailed standardized operating procedures regarding specific molar equivalents and work-up techniques are essential for reproducibility.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphinic acid, trifluoroacetimidohydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and stir for 8-14 hours to facilitate oxidative cyclization and aromatization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-quinolyl-5-trifluoromethyl-1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this metal-free oxidative cyclization represents a significant opportunity for cost optimization and risk mitigation. The elimination of precious metal catalysts removes a major cost driver and supply bottleneck, as the prices of metals like palladium can be volatile and subject to geopolitical instability. Furthermore, the use of commodity chemicals like 2-methylquinoline and TBHP ensures a stable and diversified supply base, reducing the risk of raw material shortages that could disrupt production schedules. The simplified workflow also translates to reduced utility consumption and waste generation, aligning with corporate sustainability goals while lowering the total cost of ownership for the manufacturing process.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the reduction of synthetic steps from five to one dramatically lowers the direct material costs associated with production. By avoiding complex multi-step sequences, manufacturers save significantly on labor, solvent usage, and energy consumption required for intermediate isolations. The high yields reported, reaching up to 97% for certain substrates, mean less raw material is wasted, directly improving the gross margin for the final active pharmaceutical ingredient. Additionally, the simplified purification process reduces the load on chromatography columns and solvent recovery systems, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit greatly from the use of widely available, non-proprietary starting materials that are produced by multiple global suppliers. This redundancy ensures that production lines remain operational even if one vendor faces disruptions, providing a buffer against supply chain shocks. The robustness of the reaction conditions, which do not require specialized anhydrous infrastructure, allows for manufacturing in a broader range of facilities, increasing geographic flexibility. This adaptability is crucial for maintaining continuous supply to downstream customers in the pharmaceutical sector, where interruptions can have severe regulatory and commercial consequences.
- Scalability and Environmental Compliance: The transition from bench-scale to commercial production is facilitated by the straightforward nature of the reaction, which utilizes standard heating and stirring equipment found in most chemical plants. The absence of heavy metals simplifies environmental compliance, as there is no need for costly wastewater treatment processes to remove toxic metal ions before discharge. This eco-friendly profile not only reduces regulatory hurdles but also enhances the marketability of the final product to environmentally conscious partners. The process generates minimal hazardous waste, supporting a cleaner manufacturing footprint and reducing the long-term liability associated with chemical disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is critical for assessing the feasibility of integrating this technology into existing manufacturing portfolios.
Q: What are the key advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring 5 steps and yielding only 17%, this patent describes a one-pot oxidative cyclization achieving yields up to 97% without heavy metal catalysts or anhydrous conditions.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process uses cheap, commercially available starting materials like 2-methylquinoline and avoids sensitive conditions, making it highly scalable for industrial production of pharmaceutical intermediates.
Q: What is the role of diphenylphosphinic acid in the reaction?
A: Diphenylphosphinic acid acts as a crucial additive that promotes the oxidative cyclization efficiency, significantly improving reaction yields compared to systems without this additive.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
As the demand for complex heterocyclic intermediates grows, partnering with an experienced CDMO is essential for navigating the complexities of scale-up. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the lab to the market. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole meets the highest industry standards. We understand the critical nature of timeline adherence in drug development and are committed to delivering consistent quality and reliability.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this metal-free process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
