Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to access nitrogen-rich heterocyclic scaffolds, particularly those containing the privileged 1,2,4-triazole motif. As detailed in the recent intellectual property disclosure CN113307790B, a novel preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds has been established, addressing critical bottlenecks in traditional synthetic routes. This technology leverages a metal-free oxidative cyclization strategy that transforms readily available 2-methylquinolines and trifluoroacetimidohydrazides into high-value intermediates under remarkably mild conditions. The significance of this chemical architecture cannot be overstated, as triazole derivatives serve as foundational cores for a vast array of bioactive molecules, including antifungal agents, kinase inhibitors, and functional materials for organic electronics.
For R&D directors and process chemists, the ability to construct these complex bicyclic systems efficiently is paramount. The disclosed methodology not only streamlines the synthetic sequence but also inherently improves the impurity profile by avoiding heavy metal contamination. By utilizing a catalytic system based on tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), the process achieves high atom economy and operational simplicity. This represents a paradigm shift from classical approaches that often rely on precious metal catalysts or harsh stoichiometric oxidants. For procurement and supply chain stakeholders, this innovation translates directly into reduced raw material costs and enhanced supply continuity, as the starting materials are commodity chemicals rather than specialized, scarce reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiencies that hinder commercial viability. Traditional literature methods typically employ quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step synthetic sequence to arrive at the target heterocycle. This multi-step approach suffers from a dismal cumulative total yield of approximately 17%, which is economically unsustainable for large-scale manufacturing. Furthermore, these legacy routes often demand severe reaction conditions, including strict anhydrous and anaerobic environments, which impose significant infrastructure costs on production facilities. The reliance on transition metal catalysts in some variations introduces another layer of complexity, requiring rigorous downstream purification to meet stringent regulatory limits for residual metals in pharmaceutical intermediates. These factors collectively create a high barrier to entry for reliable suppliers attempting to bring these compounds to market at a competitive price point.
The Novel Approach
In stark contrast, the method described in patent CN113307790B offers a direct, one-pot oxidative cyclization that bypasses the need for pre-functionalized carboxylic acid derivatives. By utilizing 2-methylquinoline and trifluoroacetimidohydrazide as the coupling partners, the reaction constructs the triazole ring and installs the quinoline moiety simultaneously. The core transformation is driven by an organic redox system comprising TBAI and TBHP in the presence of diphenylphosphoric acid as an additive. This approach eliminates the need for expensive transition metals and allows the reaction to proceed under ambient air without moisture sensitivity. As illustrated in the reaction scheme below, the process tolerates a wide range of substituents on both the hydrazide and the quinoline ring, enabling the rapid generation of diverse analog libraries for structure-activity relationship (SAR) studies.
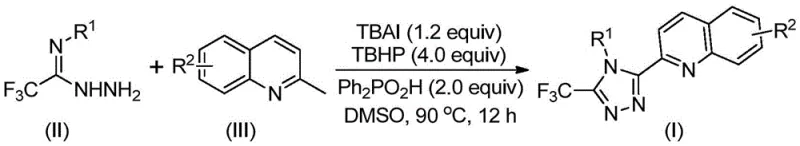
The operational simplicity of this novel route is a major advantage for process development teams. The reaction typically reaches completion within 8 to 14 hours at temperatures between 80°C and 100°C, using dimethyl sulfoxide (DMSO) as the preferred solvent. Post-reaction workup is straightforward, involving filtration and standard silica gel chromatography, which facilitates easy isolation of the pure product. With isolated yields frequently exceeding 80% and reaching as high as 97% for optimized substrates, this method provides a substantial improvement in mass efficiency compared to the prior art. This efficiency gain is critical for reducing the environmental footprint of the manufacturing process and lowering the overall cost of goods sold (COGS) for the final active pharmaceutical ingredient.
Mechanistic Insights into TBAI/TBHP Catalyzed Oxidative Cyclization
The success of this transformation relies on a sophisticated yet elegant radical-mediated mechanism initiated by the TBAI/TBHP system. Mechanistically, the tetrabutylammonium iodide acts as a catalyst that activates the tert-butyl hydroperoxide, generating reactive iodine species in situ. These species are responsible for the initial oxidation of the methyl group on the 2-methylquinoline substrate, effectively converting it into an aldehyde equivalent or a reactive radical intermediate. This activated quinoline species then undergoes a condensation reaction with the trifluoroacetimidohydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination and intramolecular electrophilic substitution trigger the cyclization event, closing the triazole ring. The final aromatization step releases the stable 3-quinolyl-5-trifluoromethyl-1,2,4-triazole product. This cascade sequence avoids the formation of stable off-cycle intermediates that often plague stepwise syntheses, thereby driving the reaction forward with high thermodynamic favorability.
From an impurity control perspective, the metal-free nature of this catalytic cycle is a distinct advantage for quality assurance. Traditional cross-coupling reactions often leave behind traces of palladium, copper, or nickel, which can catalyze degradation pathways in the final drug product or pose toxicity risks. By utilizing an entirely organic catalytic system based on iodine and peroxide, the impurity profile is significantly cleaner, consisting primarily of organic byproducts that are easier to separate via crystallization or chromatography. Furthermore, the use of diphenylphosphoric acid as an additive appears to stabilize the reactive intermediates and promote the cyclization step, minimizing the formation of polymeric side products. This mechanistic understanding allows process chemists to fine-tune reaction parameters, such as the stoichiometry of the oxidant (typically 4.0 equivalents of TBHP) and the loading of the iodide source (1.2 equivalents of TBAI), to maximize yield and minimize waste generation.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators should follow the standardized protocol outlined in the patent examples, which emphasizes precise stoichiometric control and temperature management. The procedure involves charging a reaction vessel with the requisite amounts of tetrabutylammonium iodide, aqueous tert-butyl hydroperoxide, diphenylphosphoric acid, the specific trifluoroacetimidohydrazide derivative, and 2-methylquinoline in DMSO. The mixture is then heated to 90°C and maintained for 12 hours to ensure full conversion. Detailed standard operating procedures (SOPs) regarding safety handling of peroxides and workup protocols are essential for safe execution. For a comprehensive step-by-step guide tailored to your specific substrate, please refer to the technical documentation below.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphoric acid, trifluoroacetimidohydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and stir for 8-14 hours to facilitate oxidative cyclization without requiring inert atmosphere conditions.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of precious metal catalysts removes a significant cost driver from the bill of materials, as there is no longer a need to purchase expensive palladium or rhodium complexes. Moreover, the absence of heavy metals simplifies the purification train, potentially removing entire unit operations dedicated to metal scavenging, which reduces both processing time and consumable costs. The starting materials, specifically 2-methylquinolines and aromatic amines used to prepare the hydrazides, are commodity chemicals available from multiple global suppliers, ensuring a resilient supply chain that is not dependent on single-source vendors. This diversification of the supply base mitigates the risk of production delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound, primarily driven by the drastic simplification of the downstream processing workflow. By avoiding the use of transition metals, manufacturers save significantly on the costs associated with specialized metal scavenger resins and the extensive analytical testing required to certify low residual metal levels. Additionally, the high atom economy of the oxidative cyclization means that less raw material is wasted as byproduct, leading to a lower effective cost per kilogram of the final intermediate. The use of inexpensive oxidants like TBHP and common solvents like DMSO further contributes to a leaner cost structure, making the final API more competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of robust, non-sensitive reagents that do not require specialized storage conditions such as inert gas blanketing or cryogenic temperatures. The reaction tolerance to moisture and oxygen means that production can proceed even if minor deviations in environmental controls occur, reducing the rate of batch failures and reworks. Since the key building blocks are widely produced bulk chemicals, lead times for raw material procurement are typically short, allowing for agile response to fluctuations in market demand. This reliability is crucial for maintaining consistent inventory levels of critical pharmaceutical intermediates and avoiding stockouts that could disrupt downstream drug formulation.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns well with green chemistry principles by reducing the generation of hazardous heavy metal waste streams. The scalability of the reaction has been demonstrated at the gram scale in the patent examples, and the exothermic profile of the oxidation is manageable with standard cooling systems, facilitating safe scale-up to multi-kilogram or tonnage production. The simplified workup procedure generates less solvent waste compared to multi-step sequences, lowering the overall E-factor of the process. This environmental efficiency not only reduces disposal costs but also supports corporate sustainability goals, which are increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps R&D and procurement teams evaluate the feasibility of integrating this route into their existing manufacturing portfolios. For further clarification on specific substrate compatibility or custom synthesis requests, direct consultation with our technical team is recommended.
Q: Does this synthesis method require toxic heavy metal catalysts?
A: No, the patented process (CN113307790B) utilizes an organic catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), completely eliminating the need for transition metals and simplifying downstream purification.
Q: What are the typical reaction yields for this triazole scaffold?
A: The method demonstrates robust efficiency with isolated yields ranging from 51% to 97% across various substrates, with electron-deficient aryl groups often providing superior conversion rates compared to traditional multi-step routes.
Q: Is the process suitable for large-scale industrial production?
A: Yes, the reaction operates under ambient air conditions without strict anhydrous requirements and uses commercially available, inexpensive reagents, making it highly amenable to kilogram-to-ton scale-up for commercial supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN113307790B and is fully equipped to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities feature rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole intermediate meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis route for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this metal-free process for your specific application. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and to discuss route feasibility assessments tailored to your unique molecular targets. Let us help you optimize your supply chain and reduce your time to market with our superior manufacturing capabilities.
