Advanced Atmospheric Pressure Synthesis of Diselenides for Commercial Scale Pharmaceutical Applications
Introduction to Patent CN101619034B: A Paradigm Shift in Organoselenium Chemistry
The chemical industry is constantly seeking more sustainable and economically viable pathways for synthesizing high-value organoselenium compounds, which serve as critical intermediates in the production of pharmaceuticals and advanced functional materials. Patent CN101619034B introduces a groundbreaking methodology for the synthesis of diselenide compounds that fundamentally alters the traditional landscape of selenium chemistry. By utilizing elemental selenium and carbon monoxide as the primary reactants in the presence of water, this invention circumvents the severe safety hazards and complex operational requirements associated with legacy synthetic routes. The core innovation lies in the ability to conduct this transformation under atmospheric pressure at moderate temperatures ranging from 20°C to 100°C, thereby eliminating the need for expensive high-pressure autoclaves or cryogenic conditions. This technical breakthrough not only enhances the safety profile of the manufacturing process but also opens new avenues for cost-effective production of reliable pharmaceutical intermediate suppliers who are looking to optimize their supply chains. The versatility of this method allows for the use of various halogenated hydrocarbons, including benzyl chlorides and bromides, making it a robust platform technology for diverse chemical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diselenides has been plagued by significant technical and safety challenges that hinder efficient commercial scale-up of complex organoselenium compounds. Traditional methodologies often rely on the use of Grignard reagents reacting with selenium or chlorinated hydrocarbons, which necessitates strictly anhydrous conditions and inert atmospheres to prevent premature decomposition of the highly reactive organometallic species. Furthermore, alternative routes involving the direct use of hydrogen selenide gas present extreme toxicity risks, requiring specialized containment equipment and rigorous safety protocols that drastically increase capital expenditure and operational overhead. These conventional processes are frequently characterized by multi-step procedures, low atom economy, and the generation of substantial quantities of hazardous waste, which complicates environmental compliance and waste disposal logistics. The reliance on moisture-sensitive reagents also limits the scalability of these reactions, as even trace amounts of water can lead to catastrophic yield losses and the formation of difficult-to-remove impurities. Consequently, procurement managers have long faced difficulties in securing consistent, high-purity supplies of diselenides due to the inherent instability and danger of the incumbent manufacturing technologies.
The Novel Approach
In stark contrast to these archaic methods, the novel approach detailed in the patent leverages a selenium-catalyzed carbonylation strategy that operates under remarkably mild and forgiving conditions. By employing elemental selenium, a cheap and readily available non-metallic raw material, in conjunction with carbon monoxide and water, the process achieves high conversion rates without the need for exotic catalysts or extreme pressures. The reaction proceeds smoothly in common organic solvents such as DMF or toluene, with the addition of simple inorganic or organic bases acting as promoters to facilitate the transformation. This one-pot synthesis strategy significantly reduces the number of unit operations required, streamlining the workflow from raw material charging to final product isolation. The ability to use industrial grade carbon monoxide tail gas, which may contain impurities like nitrogen or carbon dioxide, further underscores the economic robustness of this method, allowing manufacturers to utilize lower-cost feedstocks without sacrificing product quality. This shift represents a major leap forward in process chemistry, offering a pathway to substantial cost savings and enhanced operational reliability for producers of fine chemical intermediates.
Mechanistic Insights into Selenium-Catalyzed Carbonylation
The mechanistic pathway of this reaction involves a sophisticated interplay between the halogenated hydrocarbon substrate, elemental selenium, and carbon monoxide in an aqueous organic medium. Initially, the elemental selenium acts as a nucleophile or participates in a redox cycle facilitated by the base promoter, activating the carbon-halogen bond of the substrate. The presence of water is critical, likely serving as a proton source or participating in the hydrolysis of intermediate acyl selenium species to drive the reaction forward towards the formation of the diselenide bond. Carbon monoxide serves as the reducing agent and carbon source, inserting into the selenium-carbon framework to generate reactive intermediates that eventually couple to form the R-Se-Se-R structure. The reaction kinetics are highly dependent on the molar ratios of the reactants, with optimal results observed when the halogenated hydrocarbon to selenium ratio is maintained between 2:1 and 2:1.5. The base promoter, whether it be sodium acetate, triethylamine, or DBU, plays a pivotal role in neutralizing the acid byproducts generated during the reaction, thus preventing the accumulation of acidic species that could inhibit the catalytic cycle or degrade the product. Understanding these mechanistic nuances is essential for R&D directors aiming to adapt this chemistry for specific API intermediates, as slight modifications in solvent polarity or base strength can influence the reaction rate and selectivity.
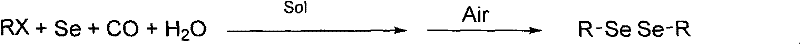
Impurity control in this system is inherently superior to traditional methods due to the simplicity of the reaction matrix and the ease of separating unreacted starting materials. Since elemental selenium is used in a stoichiometric or slightly excess amount, any unreacted selenium can be quantitatively removed by a simple oxidation and filtration step, where switching the atmosphere from CO to air causes the selenium to precipitate out of the solution. This physical separation method ensures that the final filtrate contains minimal inorganic contaminants, leading to products with purity levels exceeding 99% as confirmed by NMR analysis in the patent examples. Furthermore, the absence of transition metal catalysts eliminates the risk of heavy metal residues, a critical quality attribute for pharmaceutical intermediates intended for human consumption. The reaction selectivity is also remarkably high, minimizing the formation of side products such as selenides or oxidized selenium species, which simplifies the downstream purification process and improves overall yield. For quality assurance teams, this translates to a more predictable impurity profile and reduced analytical burden, facilitating faster batch release times and more consistent supply chain performance.
How to Synthesize Dibenzyl Diselenide Efficiently
Implementing this synthesis route requires careful attention to the sequence of reagent addition and gas flow management to maximize yield and safety. The process begins with the preparation of a reaction mixture containing the halogenated hydrocarbon, selenium powder, water, and a chosen base promoter dissolved in a polar aprotic solvent like DMF. It is crucial to maintain a continuous flow of carbon monoxide throughout the heating phase to ensure sufficient concentration of the gas in the liquid phase for the reaction to proceed efficiently. The detailed standardized synthesis steps below outline the precise operational parameters derived from the patent data, providing a clear roadmap for laboratory validation and subsequent pilot plant trials. Operators should monitor the temperature closely to stay within the 20-100°C window, adjusting the heating rate to prevent localized hot spots that could lead to side reactions. Following the reaction period, the switch to an oxidative atmosphere must be performed gradually to allow for the controlled precipitation of selenium, ensuring that the filtration step captures all solid residues effectively before product isolation via water precipitation.
- Charge a reactor with halogenated hydrocarbon, elemental selenium powder, water, and an organic or inorganic base promoter in a suitable polar solvent such as DMF.
- Continuously feed carbon monoxide gas into the reaction mixture while heating to a temperature range of 20-100°C and stirring for 1 to 24 hours under normal pressure.
- Cool the mixture to room temperature, switch the gas feed to air or oxygen to precipitate unreacted selenium, filter, and add water to the filtrate to isolate the pure diselenide product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers transformative benefits that directly address the key pain points of cost, safety, and scalability in the fine chemicals sector. The elimination of expensive and hazardous reagents like Grignard reagents or hydrogen selenide gas results in a drastic simplification of the raw material procurement strategy, allowing purchasing departments to source commodities that are widely available and price-stable. The ability to operate at atmospheric pressure removes the requirement for specialized high-pressure reactors, significantly lowering the barrier to entry for contract manufacturing organizations and reducing the capital intensity of the production facility. This technological shift enables a more agile supply chain capable of responding quickly to market demand fluctuations without the long lead times associated with complex equipment maintenance or safety audits. Moreover, the green chemistry attributes of the process, characterized by low waste generation and the use of benign solvents, align perfectly with the increasing regulatory pressure for sustainable manufacturing practices, thereby future-proofing the supply chain against evolving environmental regulations.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the substitution of high-cost, high-risk reagents with inexpensive elemental selenium and industrial carbon monoxide. By removing the need for cryogenic cooling systems and inert gas blankets required for Grignard chemistry, the utility consumption per kilogram of product is significantly lowered. The high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, improving the overall atom economy and reducing the cost of goods sold. Additionally, the simplified workup procedure, which relies on filtration and precipitation rather than complex chromatographic separations, reduces solvent usage and labor hours, contributing to substantial operational expenditure savings. These cumulative efficiencies create a competitive pricing structure that allows suppliers to offer high-purity intermediates at a more attractive price point without compromising margin.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, which is critical for maintaining uninterrupted supply to downstream pharmaceutical customers. Since the process tolerates a degree of impurity in the carbon monoxide feedstock, manufacturers are not solely dependent on ultra-high-purity gas suppliers, mitigating the risk of supply disruptions due to gas shortages. The use of stable, non-pyrophoric solids like selenium powder simplifies logistics and storage requirements, reducing the complexity of hazardous material handling and transportation. This stability extends to the shelf-life of the intermediates, as the high purity achieved reduces the likelihood of degradation during storage, ensuring that inventory remains viable for longer periods. For supply chain heads, this translates to reduced safety stock requirements and improved cash flow management.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards and the use of standard agitation and heating equipment. The one-pot nature of the synthesis reduces the number of transfer steps, minimizing the potential for material loss and cross-contamination during scale-up. Environmentally, the process generates minimal hazardous waste, as the primary byproduct is recoverable elemental selenium, which can potentially be recycled back into the process. The lack of heavy metal catalysts simplifies wastewater treatment and ensures compliance with strict discharge limits regarding metal content. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturer, making it a preferred partner for environmentally conscious global enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diselenide synthesis technology, drawing directly from the experimental data and claims of the patent. These insights are designed to clarify the operational feasibility and strategic value of adopting this method for large-scale production. Understanding the specific nuances of catalyst recovery and solvent selection is vital for engineering teams planning the technology transfer. The answers provided reflect the proven capabilities of the process as demonstrated in the patent examples, offering a realistic view of its potential impact on manufacturing efficiency and product quality.
Q: What are the primary safety advantages of this diselenide synthesis method compared to traditional routes?
A: Unlike conventional methods that utilize highly toxic hydrogen selenide gas or moisture-sensitive Grignard reagents, this patented process employs elemental selenium and carbon monoxide under atmospheric pressure. This eliminates the need for handling hazardous gases and reduces the risk of exothermic runaways associated with organometallic reagents, significantly enhancing operational safety for large-scale manufacturing.
Q: Can industrial waste carbon monoxide be utilized in this reaction process?
A: Yes, the patent explicitly demonstrates that industrial carbon monoxide tail gas containing impurities such as nitrogen, carbon dioxide, or water vapor can be effectively used without compromising yield. This flexibility allows manufacturers to utilize lower-cost industrial byproduct gases, further driving down raw material costs while maintaining high product purity above 99%.
Q: How does the catalyst separation process work in this selenium-mediated reaction?
A: The process utilizes a unique oxidation step where switching from carbon monoxide to air or oxygen causes unreacted elemental selenium to precipitate out of the solution. This solid selenium can be easily removed via simple filtration before product isolation, ensuring the final diselenide product is free from heavy metal contamination and simplifying the downstream purification workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diselenide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of patent CN101619034B in reshaping the supply landscape for organoselenium intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped to handle the specific requirements of this carbonylation chemistry, including robust gas handling systems and rigorous QC labs capable of verifying stringent purity specifications. We are committed to leveraging this advanced synthetic route to deliver high-purity diselenides that meet the exacting standards of the global pharmaceutical industry, thereby supporting our partners in accelerating their drug development timelines. Our technical team is ready to collaborate on process optimization to further enhance yield and reduce environmental footprint.
We invite procurement leaders and R&D directors to engage with us to explore how this technology can be integrated into your supply chain for maximum efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments tailored to your project needs. Let us partner with you to turn this innovative chemistry into a commercial reality, driving value and reliability in your manufacturing operations.
